|
Tetrahedrane
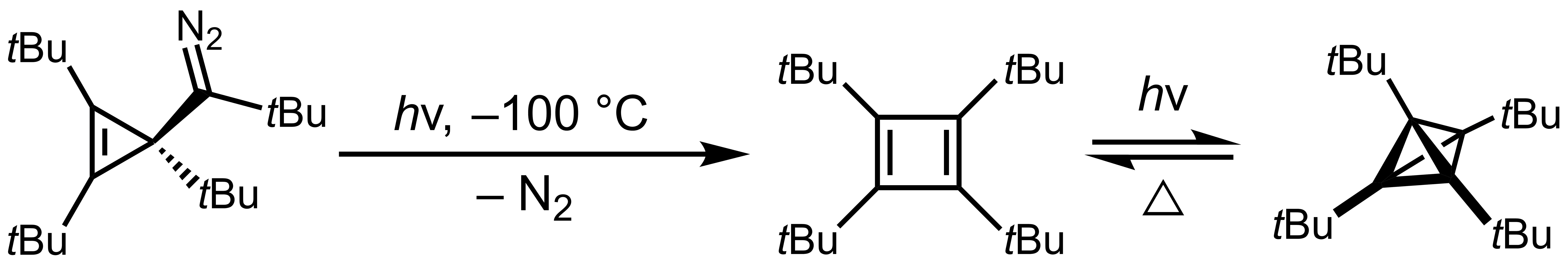
Tetrahedrane is a hypothetical platonic hydrocarbon with chemical formula and a tetrahedral structure. The molecule would be subject to considerable angle strain and has not been synthesized . However, a number of derivatives have been prepared. In a more general sense, the term ''tetrahedranes'' is used to describe a class of molecules and ions with related structure, e.g. white phosphorus. C4 tetrahedranes Tetrahedrane () is one of the possible platonic hydrocarbons and has the IUPAC name tricyclo .1.0.02,4utane. Unsubstituted tetrahedrane remains elusive, although predicted kinetically stable. One strategy that has been explored (but thus far failed) is reaction of propene with atomic carbon. Contrariwise, several organic compounds with the tetrahedrane core are known. All have multiply bulky substituents, ''tert''-butyl (''t''-Bu) or larger. Locking a tetrahedrane molecule inside a fullerene has only been attempted ''in silico''. All known syntheses have ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetrahedrane From Diazo
Tetrahedrane is a hypothetical platonic hydrocarbon with chemical formula and a tetrahedral structure. The molecule would be subject to considerable angle strain and has not been synthesized . However, a number of derivatives have been prepared. In a more general sense, the term ''tetrahedranes'' is used to describe a class of molecules and ions with related structure, e.g. white phosphorus. C4 tetrahedranes Tetrahedrane () is one of the possible platonic hydrocarbons and has the IUPAC name tricyclo .1.0.02,4utane. Unsubstituted tetrahedrane remains elusive, although predicted kinetically stable. One strategy that has been explored (but thus far failed) is reaction of propene with atomic carbon. Contrariwise, several organic compounds with the tetrahedrane core are known. All have multiply bulky substituents, ''tert''-butyl (''t''-Bu) or larger. Locking a tetrahedrane molecule inside a fullerene has only been attempted ''in silico''. All known syntheses have relie ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Platonic Hydrocarbon
In organic chemistry, a Platonic hydrocarbon is a hydrocarbon whose structure matches one of the five Platonic solids, with carbon atoms replacing its vertices, carbon–carbon bonds replacing its edges, and hydrogen atoms as needed. Not all Platonic solids have molecular hydrocarbon counterparts; those that do are the tetrahedron (tetrahedrane), the cube (cubane), and the dodecahedron (dodecahedrane). The possibility and existence of each platonic hydrocarbon is affected by the number of bonds to each carbon vertex and the angle strain between the bonds at each vertex. Tetrahedrane Tetrahedrane (C4H4) is a hypothetical compound. It has not yet been synthesized without substituents, but it is predicted to be kinetically stable in spite of its angle strain. Some stable derivatives, including tetra( ''tert''-butyl)tetrahedrane and tetra(trimethylsilyl)tetrahedrane, have been produced. Cubane Cubane (C8H8) has been synthesized. Although it has high angle strain, cubane is kinet ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Quantum Chemistry
Quantum chemistry, also called molecular quantum mechanics, is a branch of physical chemistry focused on the application of quantum mechanics to chemical systems, particularly towards the quantum-mechanical calculation of electronic contributions to physical and chemical properties of Molecule, molecules, Material, materials, and solutions at the atomic level. These calculations include systematically applied approximations intended to make calculations computationally feasible while still capturing as much information about important contributions to the computed Wave function, wave functions as well as to observable properties such as structures, spectra, and Thermodynamics, thermodynamic properties. Quantum chemistry is also concerned with the computation of quantum effects on molecular dynamics and chemical kinetics. Chemists rely heavily on spectroscopy through which information regarding the Quantization (physics), quantization of energy on a molecular scale can be obtained ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tert-butyllithium
''tert''-Butyllithium is a chemical compound with the Chemical formula, formula (CH3)3CLi. As an organolithium compound, it has applications in organic synthesis since it is a strong base (chemistry), base, capable of deprotonation, deprotonating many carbon molecules, including benzene. ''tert''-Butyllithium is available commercially as solutions in hydrocarbons (such as pentane); it is not usually prepared in the laboratory. Preparation ''tert''-Butyllithium is produced commercially by treating tert-butyl chloride with lithium. Its synthesis was first reported by R. B. Woodward in 1941. Structure and bonding Like other organolithium compounds, ''tert''-butyllithium is a cluster compound. Whereas n-Butyllithium, ''n''-butyllithium exists both as a hexamer and a tetramer, ''tert''-butyllithium exists exclusively as a tetramer with a cubane-type cluster, cubane structure. Bonding in organolithium clusters involves three-center two-electron bond, sigma delocalization and significan ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bromination
In chemistry, halogenation is a chemical reaction which introduces one or more halogens into a chemical compound. Halide-containing compounds are pervasive, making this type of transformation important, e.g. in the production of polymers, drugs. This kind of conversion is in fact so common that a comprehensive overview is challenging. This article mainly deals with halogenation using elemental halogens (). Halides are also commonly introduced using salts of the halides and halogen acids. Many specialized reagents exist for introducing halogens into diverse substrates, e.g. thionyl chloride. Organic chemistry Several pathways exist for the halogenation of organic compounds, including free radical halogenation, ketone halogenation, electrophilic halogenation, and halogen addition reaction. The nature of the substrate determines the pathway. The facility of halogenation is influenced by the halogen. Fluorine and chlorine are more electrophilic and are more aggressive halogena ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cyclopentadienone
Cyclopentadienone is an organic compound with molecular formula C5H4O. The parent cyclopentadienone is rarely encountered, because it rapidly dimerizes. Many substituted derivatives are known, notably tetraphenylcyclopentadienone. Such compounds are used as ligands in organometallic chemistry Organometallic chemistry is the study of organometallic compounds, chemical compounds containing at least one chemical bond between a carbon atom of an organic molecule and a metal, including alkali, alkaline earth, and transition metals, and so .... left, The Knölker complex, derived from a substituted cyclopentadienone, is a catalyst for transfer hydrogenation. Preparation Cyclopentadienone can be generated by the photolysis or pyrolysis of various substances (e.g. 1,2-benzoquinone), and then isolated in an matrix isolation, argon matrix at . It dimerizes readily upon thawing the matrix at . See also * Dienone References {{Cyclopentadienide complexes Ketones Fully conjugat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Decarboxylation
Decarboxylation is a chemical reaction that removes a carboxyl group and releases carbon dioxide (CO2). Usually, decarboxylation refers to a reaction of carboxylic acids, removing a carbon atom from a carbon chain. The reverse process, which is the first chemical step in photosynthesis, is called carboxylation, the addition of CO2 to a compound. Enzymes that catalyze decarboxylations are called decarboxylases or, the more formal term, carboxy-lyases (Enzyme Commission number, EC number 4.1.1). In organic chemistry The term "decarboxylation" usually means replacement of a carboxyl group () with a hydrogen atom: : Decarboxylation is one of the oldest known organic reactions. It is one of the processes assumed to accompany pyrolysis and destructive distillation. Overall, decarboxylation depends upon stability of the carbanion synthon , although the anion may not be a true chemical intermediate. Typically, carboxylic acids decarboxylate slowly, but carboxylic acids with an α el ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Maleic Anhydride
Maleic anhydride is an organic compound with the formula . It is the acid anhydride of maleic acid. It is a colorless or white solid with an acrid odor. It is produced industrially on a large scale for applications in coatings and polymers. Structure and bonding Maleic anhydride is a planar molecule. By virtue of the acid anhydride group, the alkene is electrophilic. On account of its cycle of 4 π electrons in an array of 5 atoms with p orbitals, maleic anhydride was long thought to exhibit antiaromaticity. However, a thermochemical study concluded that only 8 kJ/mol of destabilization energy can be ascribed to this effect, making it weakly antiaromatic at best. Production Maleic anhydride is produced by vapor-phase oxidation of butane, ''n''-butane. The overall process converts the methyl groups to carboxylate and dehydrogenation, dehydrogenates the backbone. The selectivity of the process reflects the robustness of maleic anhydride, with its conjugated double-bond system. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkyne
\ce \ce Acetylene \ce \ce \ce Propyne \ce \ce \ce \ce 1-Butyne In organic chemistry, an alkyne is an unsaturated hydrocarbon containing at least one carbon—carbon triple bond. The simplest acyclic alkynes with only one triple bond and no other functional groups form a homologous series with the general chemical formula . Alkynes are traditionally known as acetylenes, although the name ''acetylene'' also refers specifically to , known formally as ethyne using IUPAC nomenclature. Like other hydrocarbons, alkynes are generally hydrophobic. Structure and bonding In acetylene, the H–C≡C bond angles are 180°. By virtue of this bond angle, alkynes are rod-like. Correspondingly, cyclic alkynes are rare. Benzyne cannot be isolated. The C≡C bond distance of 118 picometers (for C2H2) is much shorter than the C=C distance in alkenes (132 pm, for C2H4) or the C–C bond in alkanes (153 pm). : The triple bond is very strong with a bond strength of 839 kJ/mol. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cycloaddition
In organic chemistry, a cycloaddition is a chemical reaction in which "two or more Unsaturated hydrocarbon, unsaturated molecules (or parts of the same molecule) combine with the formation of a cyclic adduct in which there is a net reduction of the Multiplicity (chemistry)#Molecules, bond multiplicity". The resulting reaction is a cyclization reaction. Many but not all cycloadditions are Concerted reaction, concerted and thus pericyclic. Nonconcerted cycloadditions are not pericyclic. As a class of addition reaction, cycloadditions permit carbon–carbon bond formation without the use of a nucleophile or electrophile. Cycloadditions can be described using two systems of notation. An older but still common notation is based on the size of linear arrangements of atoms in the reactants. It uses parentheses: where the variables are the numbers of linear atoms in each reactant. The product is a cycle of size . In this system, the standard Diels-Alder reaction is a (4 + 2)-cyc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |