|
Photoelectric
The photoelectric effect is the emission of electrons from a material caused by electromagnetic radiation such as ultraviolet light. Electrons emitted in this manner are called photoelectrons. The phenomenon is studied in condensed matter physics, solid state, and quantum chemistry to draw inferences about the properties of atoms, molecules and solids. The effect has found use in electronic devices specialized for light detection and precisely timed electron emission. The experimental results disagree with classical electromagnetism, which predicts that continuous light waves transfer energy to electrons, which would then be emitted when they accumulate enough energy. An alteration in the intensity of light would theoretically change the kinetic energy of the emitted electrons, with sufficiently dim light resulting in a delayed emission. The experimental results instead show that electrons are dislodged only when the light exceeds a certain frequency—regardless of the light's ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Albert Einstein
Albert Einstein (14 March 187918 April 1955) was a German-born theoretical physicist who is best known for developing the theory of relativity. Einstein also made important contributions to quantum mechanics. His mass–energy equivalence formula , which arises from special relativity, has been called "the world's most famous equation". He received the 1921 Nobel Prize in Physics for . Born in the German Empire, Einstein moved to Switzerland in 1895, forsaking his German citizenship (as a subject of the Kingdom of Württemberg) the following year. In 1897, at the age of seventeen, he enrolled in the mathematics and physics teaching diploma program at the Swiss ETH Zurich, federal polytechnic school in Zurich, graduating in 1900. He acquired Swiss citizenship a year later, which he kept for the rest of his life, and afterwards secured a permanent position at the Swiss Patent Office in Bern. In 1905, he submitted a successful PhD dissertation to the University of Zurich. In 19 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless particles that can move no faster than the speed of light measured in vacuum. The photon belongs to the class of boson particles. As with other elementary particles, photons are best explained by quantum mechanics and exhibit wave–particle duality, their behavior featuring properties of both waves and particles. The modern photon concept originated during the first two decades of the 20th century with the work of Albert Einstein, who built upon the research of Max Planck. While Planck was trying to explain how matter and electromagnetic radiation could be in thermal equilibrium with one another, he proposed that the energy stored within a material object should be regarded as composed of an integer number of discrete, equal-sized parts. To explain the pho ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Compton Scattering
Compton scattering (or the Compton effect) is the quantum theory of high frequency photons scattering following an interaction with a charged particle, usually an electron. Specifically, when the photon hits electrons, it releases loosely bound electrons from the outer valence shells of atoms or molecules. The effect was discovered in 1923 by Arthur Holly Compton while researching the scattering of X-rays by light elements, and earned him the Nobel Prize in Physics in 1927. The Compton effect significantly deviated from dominating classical theories, using both special relativity and quantum mechanics to explain the interaction between high frequency photons and charged particles. Photons can interact with matter at the atomic level (e.g. photoelectric effect and Rayleigh scattering), at the nucleus, or with just an electron. Pair production and the Compton effect occur at the level of the electron. When a high frequency photon scatters due to an interaction with a charged part ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Photovoltaic Effect
The photovoltaic effect is the generation of voltage and electric current in a material upon exposure to light. It is a physical phenomenon. The photovoltaic effect is closely related to the photoelectric effect. For both phenomena, light is absorbed, causing excitation of an electron or other charge carrier to a higher-energy state. The main distinction is that the term ''photoelectric effect'' is now usually used when the electron is ejected out of the material (usually into a vacuum) and ''photovoltaic effect'' used when the excited charge carrier is still contained within the material. In either case, an electric potential (or voltage) is produced by the separation of charges, and the light has to have a sufficient energy to overcome the potential barrier for excitation. The physical essence of the difference is usually that photoelectric emission separates the charges by ballistic conduction and photovoltaic emission separates them by diffusion, but some "hot carrier" phot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electron
The electron (, or in nuclear reactions) is a subatomic particle with a negative one elementary charge, elementary electric charge. It is a fundamental particle that comprises the ordinary matter that makes up the universe, along with up quark, up and down quark, down quarks. Electrons are extremely lightweight particles that orbit the positively charged atomic nucleus, nucleus of atoms. Their negative charge is balanced by the positive charge of protons in the nucleus, giving atoms their overall electric charge#Charge neutrality, neutral charge. Ordinary matter is composed of atoms, each consisting of a positively charged nucleus surrounded by a number of orbiting electrons equal to the number of protons. The configuration and energy levels of these orbiting electrons determine the chemical properties of an atom. Electrons are bound to the nucleus to different degrees. The outermost or valence electron, valence electrons are the least tightly bound and are responsible for th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Introduction To Quantum Mechanics
Quantum mechanics is the study of matter and its interactions with energy on the orders of magnitude (length), scale of atomic and elementary particle, subatomic particles. By contrast, classical physics explains matter and energy only on a scale familiar to human experience, including the behavior of astronomical bodies such as the Moon. Classical physics is still used in much of modern science and technology. However, towards the end of the 19th century, scientists discovered phenomena in both the large (Macroscopic scale, macro) and the small (Microscopic scale, micro) worlds that classical physics could not explain. The desire to resolve inconsistencies between observed phenomena and classical theory led to a revolution in physics, a shift in the original scientific paradigm: the development of quantum mechanics. Many aspects of quantum mechanics are counterintuitive and can seem paradoxical because they describe behavior quite different from that seen at larger scales. In ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electromagnetic Radiation
In physics, electromagnetic radiation (EMR) is a self-propagating wave of the electromagnetic field that carries momentum and radiant energy through space. It encompasses a broad spectrum, classified by frequency or its inverse, wavelength, ranging from radio waves, microwaves, infrared, visible light, ultraviolet, X-rays, and gamma rays. All forms of EMR travel at the speed of light in a vacuum and exhibit wave–particle duality, behaving both as waves and as discrete particles called photons. Electromagnetic radiation is produced by accelerating charged particles such as from the Sun and other celestial bodies or artificially generated for various applications. Its interaction with matter depends on wavelength, influencing its uses in communication, medicine, industry, and scientific research. Radio waves enable broadcasting and wireless communication, infrared is used in thermal imaging, visible light is essential for vision, and higher-energy radiation, such ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Wave–particle Duality
Wave–particle duality is the concept in quantum mechanics that fundamental entities of the universe, like photons and electrons, exhibit particle or wave (physics), wave properties according to the experimental circumstances. It expresses the inability of the Classical physics, classical concepts such as particle or wave to fully describe the behavior of quantum objects. During the 19th and early 20th centuries, light was found to behave as a wave then later was discovered to have a particle-like behavior, whereas electrons behaved like particles in early experiments then were later discovered to have wave-like behavior. The concept of duality arose to name these seeming contradictions. History Wave-particle duality of light In the late 17th century, Sir Isaac Newton had advocated that light was Corpuscular theory of light, corpuscular (particulate), but Christiaan Huygens took an opposing wave description. While Newton had favored a particle approach, he was the first to at ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Core Electron
Core electrons are the electrons in an atom that are not valence electrons and do not participate as directly in chemical bonding. The nucleus and the core electrons of an atom form the atomic core. Core electrons are tightly bound to the nucleus. Therefore, unlike valence electrons, core electrons play a secondary role in chemical bonding and reactions by screening the positive charge of the atomic nucleus from the valence electrons. The number of valence electrons of an element can be determined by the periodic table group of the element (see valence electron): *For main-group elements, the number of valence electrons ranges from 1 to 8 (''n''s and ''n''p orbitals). *For transition metals, the number of valence electrons ranges from 3 to 12 (''n''s and (''n''−1)d orbitals). *For lanthanides and actinides, the number of valence electrons ranges from 3 to 16 (''n''s, (''n''−2)f and (''n''−1)d orbitals). All other non-valence electrons for an atom of that element are considere ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
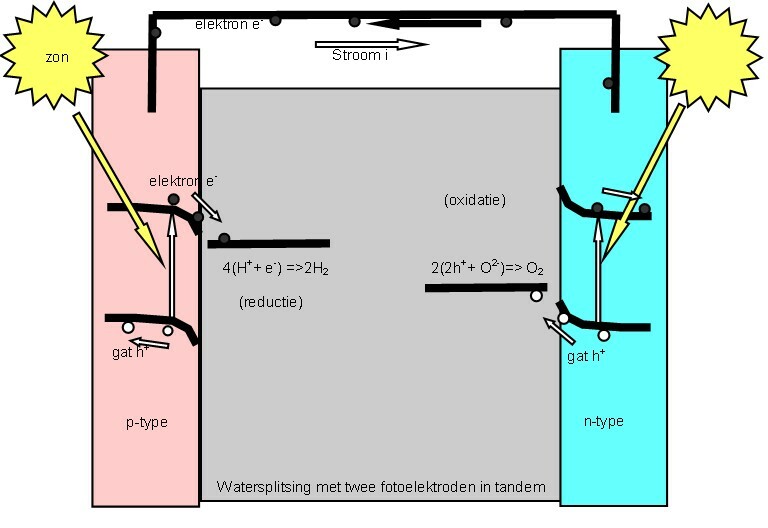
Photoelectrochemical Cell
A "photoelectrochemical cell" is one of two distinct classes of device. The first produces electrical energy similarly to a dye-sensitized photovoltaic cell, which meets the standard definition of a photovoltaic cell. The second is a photoelectrolytic cell, that is, a device which uses light incident on a photosensitizer, semiconductor, or aqueous metal immersed in an electrolytic solution to directly cause a chemical reaction, for example to produce hydrogen via the electrolysis of water. Both types of device are varieties of solar cell, in that a photoelectrochemical cell's function is to use the photoelectric effect (or, very similarly, the photovoltaic effect) to convert electromagnetic radiation (typically sunlight) either directly into electrical power, or into something which can itself be easily used to produce electrical power (hydrogen, for example, can be burned to create electrical power, see photohydrogen). Two principles The standard photovoltaic effect, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Light
Light, visible light, or visible radiation is electromagnetic radiation that can be visual perception, perceived by the human eye. Visible light spans the visible spectrum and is usually defined as having wavelengths in the range of 400–700 nanometres (nm), corresponding to frequency, frequencies of 750–420 terahertz (unit), terahertz. The visible band sits adjacent to the infrared (with longer wavelengths and lower frequencies) and the ultraviolet (with shorter wavelengths and higher frequencies), called collectively ''optical radiation''. In physics, the term "light" may refer more broadly to electromagnetic radiation of any wavelength, whether visible or not. In this sense, gamma rays, X-rays, microwaves and radio waves are also light. The primary properties of light are intensity (physics), intensity, propagation direction, frequency or wavelength spectrum, and polarization (waves), polarization. Its speed of light, speed in vacuum, , is one of the fundamental physi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |