|
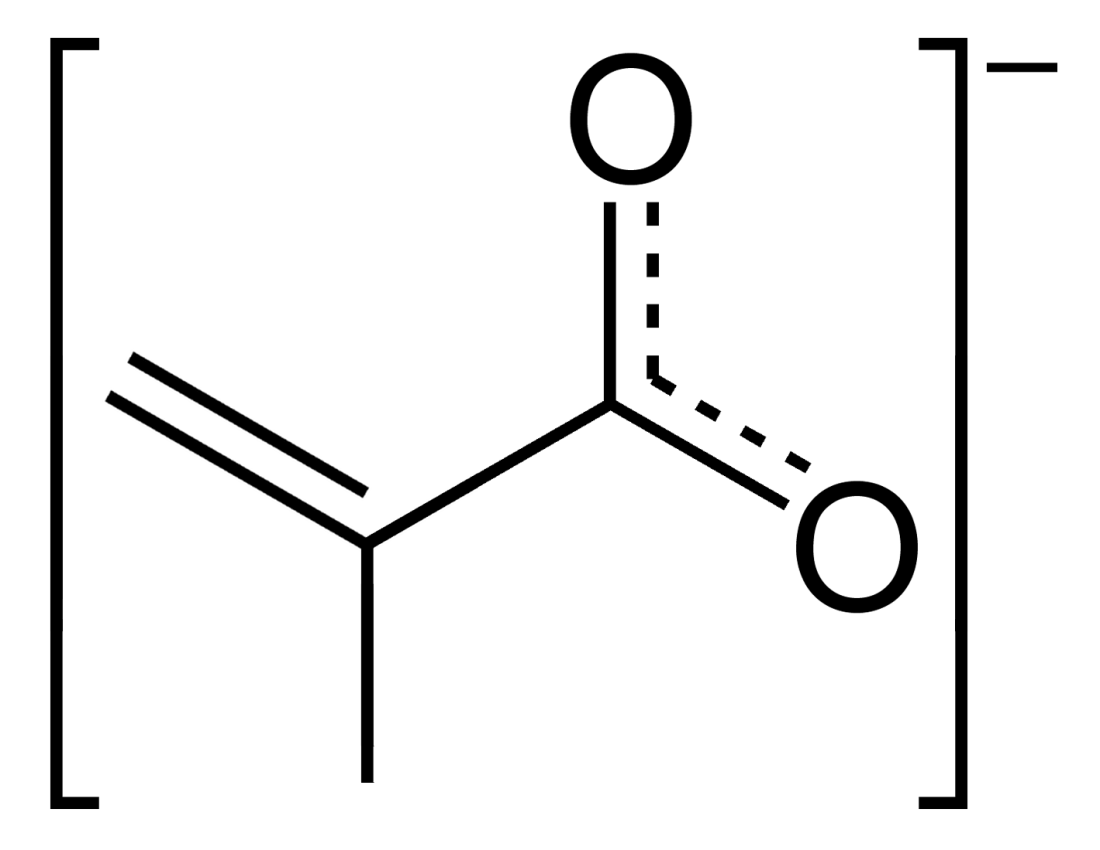
Methacrylate
Methacrylates are derivatives of methacrylic acid. These derivatives are mainly used to make poly(methyl methacrylate) and related polymers. *Monomers ** Methyl methacrylate ** Ethyl methacrylate ** Butyl methacrylate ** Hydroxyethyl methacrylate ** Glycidyl methacrylate Glycidyl methacrylate (GMA) is an ester of methacrylic acid and glycidol. Containing both an epoxide and an acrylate group, the molecule is bifunctional. It is a common monomer A monomer ( ; ''mono-'', "one" + '' -mer'', "part") is a molecu ... {{set index Carboxylate anions Monomers Methacrylate esters ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Poly(methyl Methacrylate)
Poly(methyl methacrylate) (PMMA) is a synthetic polymer derived from methyl methacrylate. It is a transparent thermoplastic, used as an engineering plastic. PMMA is also known as acrylic, acrylic glass, as well as by the trade names and brands Crylux, Walcast, Hesalite, Plexiglas, Acrylite, Lucite, PerClax, and Perspex, among several others ( see below). This plastic is often used in sheet form as a lightweight or shatter-resistant alternative to glass. It can also be used as a casting resin, in inks and coatings, and for many other purposes. It is often technically classified as a type of glass, in that it is a non-crystalline vitreous substance—hence its occasional historic designation as ''acrylic glass''. History The first acrylic acid was created in 1843. Methacrylic acid, derived from acrylic acid, was formulated in 1865. The reaction between methacrylic acid and methanol results in the ester methyl methacrylate. It was developed in 1928 in several different la ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methyl Methacrylate
Methyl methacrylate (MMA) is an organic compound with the formula . This colorless liquid, the methyl ester of methacrylic acid (MAA), is a monomer produced on a large scale for the production of poly(methyl methacrylate) (PMMA). History MMA was discovered by Bernhard Tollens and his student W. A. Caspary in 1873, who noticed and described its tendency to change into a clear, hard, transparent substance especially in sunlight. Studies on acrylic esters slowly developed until the Staudinger's theory of macromolecules and his research into the nature of polyacrylates allowed control over polymerization. Company Rohm and Haas founded by German chemist Otto Röhm, who investigated the topic for three decades, was finally able to start its industrial production in 1931. Production and properties Given the scale of production, many methods have been developed starting from diverse two- to four-carbon precursors. Two principal routes appear to be commonly practiced. Cyanohydrin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroxyethyl Methacrylate
Hydroxyethylmethacrylate (also known as glycol methacrylate) is the organic compound with the chemical formula . It is a colorless viscous liquid that readily polymerizes, i.e. it is a monomer that is used to make various polymers. Synthesis Hydroxyethylmethacrylate was first synthesized around 1925. Common methods of synthesis are: * reaction of methacrylic acid with ethylene oxide: : * esterification of methacrylic acid with a large excess of ethylene glycol. : Both these methods give also some amount of ethylene glycol dimethacrylate. During polymerization of hydroxyethylmethacrylate, it works as crosslinking agent. Properties Hydroxyethylmethacrylate is completely miscible with water and ethanol, but its polymer is practically insoluble in common solvents. Its viscosity is 0.0701 Pa⋅s at 20°C and 0.005 Pa⋅s at 30°C. During polymerization, it shrinks by approximately 6%. Applications Contact lenses In 1960, O. Wichterle and D. Lím described its use in synthesis of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycidyl Methacrylate
Glycidyl methacrylate (GMA) is an ester of methacrylic acid and glycidol. Containing both an epoxide and an acrylate group, the molecule is bifunctional. It is a common monomer A monomer ( ; ''mono-'', "one" + '' -mer'', "part") is a molecule that can react together with other monomer molecules to form a larger polymer chain or two- or three-dimensional network in a process called polymerization. Classification Chemis ... used in the production of epoxy resins. While typical home epoxies contain diglycidyl ether of bisphenol A (DGEBA), glycidyl methacrylate is instead used to provide epoxy functionalization to polyolefins and other acrylate resins. Glycidyl methacrylate is produced by several companies worldwide, including Dow Chemical. It is used to prepare a range of composites.{{cite journal , doi=10.1038/nmat1890, title=Multifunctional chondroitin sulphate for cartilage tissue–biomaterial integration , year=2007 , last1=Wang , first1=Dong-An , last2=Varghese , ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methacrylic Acid
Methacrylic acid, abbreviated MAA, is an organic compound with the formula CH2=C(CH3)CO2H. This colorless, viscous liquid is a carboxylic acid with an acrid unpleasant odor. It is soluble in warm water and miscible with most organic solvents. Methacrylic acid is produced industrially on a large scale as a precursor to its esters, especially methyl methacrylate (MMA), and to poly(methyl methacrylate) (PMMA). Production In the most common route, methacrylic acid is prepared from acetone cyanohydrin, which is converted to methacrylamide sulfate using sulfuric acid. This derivative in turn is hydrolyzed to methacrylic acid, or esterified to methyl methacrylate in one step. Another route to methacrylic acid starts with isobutylene, which obtainable by dehydration of ''tert''-butanol. Isobutylene is oxidized sequentially to methacrolein and then methacrylic acid. Methacrolein for this purpose can also be obtained from formaldehyde and ethylene. Yet a third route involves the dehy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethyl Methacrylate
Ethyl methacrylate is the organic compound with the formula C2H5O2CC(CH3)=CH2. A colorless liquid, it is a common monomer for the preparation of acrylate polymer An acrylate polymer (also known as acrylic or polyacrylate) is any of a group of polymers prepared from acrylate monomers. These plastics are noted for their transparency, resistance to breakage, and elasticity. Acrylate polymer is commonly used ...s.. It is typically polymerized under free-radical conditions. Ethyl methacrylate was first obtained by treating ethyl 2-hydroxyisobutyrate with phosphorus pentachloride in a dehydration reaction. Environmental issues and health hazards The related methyl and butyl methacrylates have respective acute {{LD50s of 10 and 20 g/kg (oral, rat); a linear extrapolation suggests that ethyl methacrylate would have an LD50 of approximately 13 g/kg. Acrylate esters irritate the eyes and can cause blindness. See also * Methyl methacrylate * Butyl methacrylate Ref ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Butyl Methacrylate
Butyl methacrylate is the organic compound with the formula C4H9O2CC(CH3)=CH2. A colorless liquid, it is a common monomer for the preparation of methacrylate polymers.. It is typically polymerized under free-radical In chemistry, a radical, also known as a free radical, is an atom, molecule, or ion that has at least one unpaired valence electron. With some exceptions, these unpaired electrons make radicals highly chemically reactive. Many radicals spont ... conditions.{{cite journal, title=Controlled Radical Polymerization of Methacrylic Monomers in the Presence of a Bis(ortho-chelated) Arylnickel(II) Complex and Different Activated Alkyl Halides, last1=Granel, C., last2=Dubois , first2=Ph., last3=Jérôme , first3=R., last4=Teyssié , first4=Ph., journal=Macromolecules, year=1996, volume=29, issue=27, pages=8576–8582, doi=10.1021/ma9608380, bibcode=1996MaMol..29.8576G Health hazards In terms of the acute toxicity of butyl methacrylate, the LD50 is 20 g/kg (oral, rat). I ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Monomers
A monomer ( ; ''wikt:mono-, mono-'', "one" + ''wikt:-mer, -mer'', "part") is a molecule that can chemical reaction, react together with other monomer molecules to form a larger polymer chain or two- or three-dimensional network in a process called polymerization. Classification Chemistry classifies monomers by type, and two broad classes based on the type of polymer they form. By type: * natural vs synthetic, e.g. glycine vs caprolactam, respectively * polar vs nonpolar, e.g. vinyl acetate vs ethylene, respectively * cyclic vs linear, e.g. ethylene oxide vs ethylene glycol, respectively By type of polymer they form: * those that participate in condensation polymerization * those that participate in addition polymerization Differing stoichiometry causes each class to create its respective form of polymer. : The polymerization of one kind of monomer gives a polymer#Monomers and repeat units, homopolymer. Many polymers are copolymers, meaning that they are derived from two diff ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carboxylate Anions
In organic chemistry, a carboxylate is the conjugate base of a carboxylic acid, (or ). It is an anion, an ion with negative charge. Carboxylate salts are salts that have the general formula , where M is a metal and ''n'' is 1, 2,.... Carboxylate esters have the general formula (also written as ), where R and R′ are organic groups. Synthesis Carboxylate ions can be formed by deprotonation of carboxylic acids. Such acids typically have p''K''a of less than 5, meaning that they can be deprotonated by many bases, such as sodium hydroxide or sodium bicarbonate. : Resonance stabilization of the carboxylate ion Carboxylic acids easily dissociate into a carboxylate anion and a positively charged hydrogen ion (proton), much more readily than alcohols do (into an alkoxide ion and a proton), because the carboxylate ion is stabilized by resonance. The negative charge that is left after deprotonation of the carboxyl group is delocalized between the two electronegative o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |