|
Halo Nuclei
In nuclear physics, an atomic nucleus is called a halo nucleus or is said to have a nuclear halo when it has a core nucleus surrounded by a "halo" of orbiting protons or neutrons, which makes the radius of the nucleus appreciably larger than that predicted by the liquid drop model. Halo nuclei form at the extreme edges of the table of nuclides — the neutron drip line and proton drip line — and have short half-lives, measured in milliseconds. These nuclei are studied shortly after their formation in an ion beam. Typically, an atomic nucleus is a tightly bound group of protons and neutrons. However, in some nuclides, there is an overabundance of one species of nucleon. In some of these cases, a nuclear core and a halo will form. Often, this property may be detected in scattering experiments, which show the nucleus to be much larger than the otherwise expected value. Normally, the cross-section (corresponding to the classical radius) of the nucleus is proportional to the cube ro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Helium-6 Nucleus
Helium (He) (standard atomic weight: ) has nine known isotopes, but only helium-3 (He) and helium-4 (He) are stable isotope, stable. All radioisotopes are short-lived; the longest-lived is He with half-life . The least stable is He, with half-life (), though He may have an even shorter half-life. In Earth's atmosphere, the ratio of He to He is . However, the isotopic abundance of helium varies greatly depending on its origin. In the Local Interstellar Cloud, the proportion of He to He is , which is ~121 times higher than in Earth's atmosphere. Rocks from Earth's crust have isotope ratios varying by as much as a factor of ten; this is used in geology to investigate the origin of rocks and the composition of the Earth's mantle (geology), mantle. The different formation processes of the two stable isotopes of helium produce the differing isotope abundances. Equal mixtures of liquid He and He below separate into two immiscible phases due to differences in quantum statistics: He atom ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Helium
Helium (from ) is a chemical element; it has chemical symbol, symbol He and atomic number 2. It is a colorless, odorless, non-toxic, inert gas, inert, monatomic gas and the first in the noble gas group in the periodic table. Its boiling point is the lowest among all the Chemical element, elements, and it does not have a melting point at standard pressures. It is the second-lightest and second-most Abundance of the chemical elements, abundant element in the observable universe, after hydrogen. It is present at about 24% of the total elemental mass, which is more than 12 times the mass of all the heavier elements combined. Its abundance is similar to this in both the Sun and Jupiter, because of the very high nuclear binding energy (per nucleon) of helium-4 with respect to the next three elements after helium. This helium-4 binding energy also accounts for why it is a product of both nuclear fusion and radioactive decay. The most common isotope of helium in the universe is helium-4, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon-19
Carbon (6C) has 14 known isotopes, from to as well as , of which only and are stable. The longest-lived radioisotope is , with a half-life of years. This is also the only carbon radioisotope found in nature, as trace quantities are formed cosmogenically by the reaction + → + . The most stable artificial radioisotope is , which has a half-life of . All other radioisotopes have half-lives under 20 seconds, most less than 200 milliseconds. The least stable isotope is , with a half-life of . Light isotopes tend to decay into isotopes of boron and heavy ones tend to decay into isotopes of nitrogen. List of isotopes , -id=Carbon-8 , , style="text-align:right" , 6 , style="text-align:right" , 2 , , [] , proton emission, 2p , Also immediately emits two protons for the net reaction of → + 4 , 0+ , , , -id=Carbon-9 , rowspan=3, , rowspan=3 style="text-align:right" , 6 , rowspan=3 style="text-align:right" , 3 , rowspan=3, , rowspan=3, , β+ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon
Carbon () is a chemical element; it has chemical symbol, symbol C and atomic number 6. It is nonmetallic and tetravalence, tetravalent—meaning that its atoms are able to form up to four covalent bonds due to its valence shell exhibiting 4 electrons. It belongs to group 14 of the periodic table. Carbon makes up about 0.025 percent of Earth's crust. Three Isotopes of carbon, isotopes occur naturally, carbon-12, C and carbon-13, C being stable, while carbon-14, C is a radionuclide, decaying with a half-life of 5,700 years. Carbon is one of the timeline of chemical element discoveries#Pre-modern and early modern discoveries, few elements known since antiquity. Carbon is the 15th abundance of elements in Earth's crust, most abundant element in the Earth's crust, and the abundance of the chemical elements, fourth most abundant element in the universe by mass after hydrogen, helium, and oxygen. Carbon's abundance, its unique diversity of organic compounds, and its unusual abi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boron-19
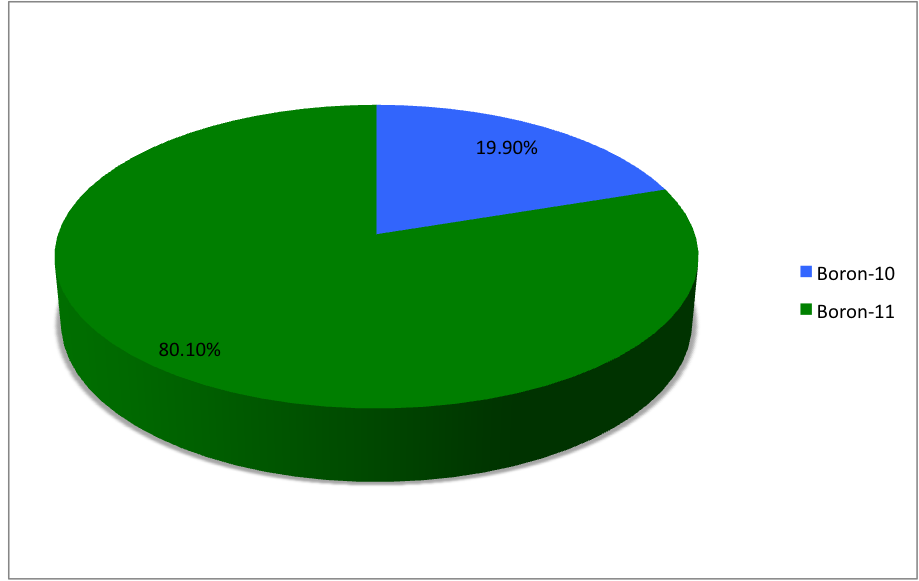
Boron (5B) naturally occurs as isotopes and , the latter of which makes up about 80% of natural boron. There are 13 radioisotopes that have been discovered, with mass numbers from 7 to 21, all with short half-lives, the longest being that of , with a half-life of only and with a half-life of . All other isotopes have half-lives shorter than . Those isotopes with mass below 10 decay into helium (via short-lived isotopes of beryllium for and ) while those with mass above 11 mostly become carbon. List of isotopes , -id=Boron-7 , , style="text-align:center" , 5 , style="text-align:center" , 2 , , [] , p , Subsequently decays by double proton emission to for a net reaction of → + 3 , (3/2−) , , , - , Has 1 halo nucleus, halo protonIntermediate product of Proton–proton chain#The p–p III branch, a branch of proton–proton chain in stellar nucleosynthesis as part of the process converting hydrogen to helium , style="text-align:center" , 5 , style ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boron-8
Boron (5B) naturally occurs as isotopes and , the latter of which makes up about 80% of natural boron. There are 13 radioisotopes that have been discovered, with mass numbers from 7 to 21, all with short half-lives, the longest being that of , with a half-life of only and with a half-life of . All other isotopes have half-lives shorter than . Those isotopes with mass below 10 decay into helium (via short-lived isotopes of beryllium for and ) while those with mass above 11 mostly become carbon. List of isotopes , -id=Boron-7 , , style="text-align:center" , 5 , style="text-align:center" , 2 , , [] , p , Subsequently decays by double proton emission to for a net reaction of → + 3 , (3/2−) , , , - , Has 1 halo nucleus, halo protonIntermediate product of Proton–proton chain#The p–p III branch, a branch of proton–proton chain in stellar nucleosynthesis as part of the process converting hydrogen to helium , style="text-align:center" , 5 , styl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boron
Boron is a chemical element; it has symbol B and atomic number 5. In its crystalline form it is a brittle, dark, lustrous metalloid; in its amorphous form it is a brown powder. As the lightest element of the boron group it has three valence electrons for forming covalent bonds, resulting in many compounds such as boric acid, the mineral sodium borate, and the ultra-hard crystals of boron carbide and boron nitride. Boron is synthesized entirely by cosmic ray spallation and supernovas and not by stellar nucleosynthesis, so it is a low-abundance element in the Solar System and in the Earth's crust. It constitutes about 0.001 percent by weight of Earth's crust. It is concentrated on Earth by the water-solubility of its more common naturally occurring compounds, the borate minerals. These are mined industrially as evaporites, such as borax and kernite. The largest known deposits are in Turkey, the largest producer of boron minerals. Elemental boron is found in smal ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Beryllium-14
Beryllium (4Be) has 11 known isotopes and 3 known isomers, but only one of these isotopes () is stable and a primordial nuclide. As such, beryllium is considered a monoisotopic element. It is also a mononuclidic element, because its other isotopes have such short half-lives that none are primordial and their abundance is very low (standard atomic weight is ). Beryllium is unique as being the only monoisotopic element with both an even number of protons and an odd number of neutrons. There are 25 other monoisotopic elements but all have odd atomic numbers, and even numbers of neutrons. Of the 10 radioisotopes of beryllium, the most stable are with a half-life of million years and with a half-life of . All other radioisotopes have half-lives under , most under . The least stable isotope is , with a half-life of . The 1:1 neutron–proton ratio seen in stable isotopes of many light elements (up to oxygen, and in elements with even atomic number up to calcium) is prevented in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Beryllium
Beryllium is a chemical element; it has Symbol (chemistry), symbol Be and atomic number 4. It is a steel-gray, hard, strong, lightweight and brittle alkaline earth metal. It is a divalent element that occurs naturally only in combination with other elements to form minerals. Gemstones high in beryllium include beryl (Aquamarine (gemstone), aquamarine, emerald, red beryl) and chrysoberyl. It is a Abundance of the chemical elements#Universe, relatively rare element in the universe, usually occurring as a product of the spallation of larger atomic nuclei that have collided with cosmic rays. Within the cores of stars, beryllium is depleted as it is fused into heavier elements. Beryllium constitutes about 0.0004 percent by mass of Earth's crust. The world's annual beryllium production of 220 tons is usually manufactured by extraction from the mineral beryl, a difficult process because beryllium bonds strongly to oxygen. In structural applications, the combination of high flexural ri ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lithium-11
Naturally occurring lithium (3Li) is composed of two stable isotopes, lithium-6 (6Li) and lithium-7 (7Li), with the latter being far more abundant on Earth. Both of the natural isotopes have an unexpectedly low nuclear binding energy per nucleon ( for 6Li and for 7Li) when compared with the adjacent lighter and heavier elements, helium ( for helium-4) and beryllium ( for beryllium-9). The longest-lived radioisotope of lithium is 8Li, which has a half-life of just . 9Li has a half-life of , and 11Li has a half-life of . All of the remaining isotopes of lithium have half-lives that are shorter than 10 nanoseconds. The shortest-lived known isotope of lithium is 4Li, which decays by proton emission with a half-life of about (), although the half-life of 3Li is yet to be determined, and is likely to be much shorter, like 2He (helium-2, diproton) which undergoes proton emission within s. Both 7Li and 6Li are two of the primordial nuclides that were produced in the Big Bang, with 7 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |