|
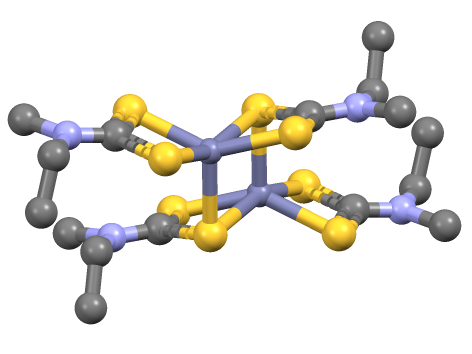
Ferbam
Iron tris(dimethyldithiocarbamate) is the coordination complex of iron with dimethyldithiocarbamate with the formula Fe(S2CNMe2)3 (Me = methyl). It is marketed as a fungicide. Synthesis, structure, bonding Iron tris(dithiocarbamate)s are typically are prepared by salt metathesis reactions. Iron tris(dimethyldithiocarbamate) is an octahedral coordination complex of iron(III) with D3 symmetry. Spin crossover (SCO) was first observed in 1931 by Cambi ''et al.'' who discovered anomalous magnetic behavior for the tris(N,N-dialkyldithiocarbamatoiron(III) complexes. The spin states of these complexes are sensitive to the nature of the amine substituents. Reactions Iron tris(dithiocarbamate)s react with nitric oxide to give a nitrosyl complex: : This efficient chemical trapping reaction provides a means to detect NO. Reflecting the strongly donating properties of dithiocarbamate ligands, iron tris(dithiocarbamate)s oxidize at relatively mild potentials to give isolable iron(IV) d ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fungicides
Fungicides are pesticides used to kill parasitic fungi or their spores. Fungi can cause serious damage in agriculture, resulting in losses of yield and quality. Fungicides are used both in agriculture and to fight fungal infections in animals, including humans. Fungicides are also used to control oomycetes, which are not taxonomically/genetically fungi, although sharing similar methods of infecting plants. Fungicides can either be contact, translaminar or systemic. Contact fungicides are not taken up into the plant tissue and protect only the plant where the spray is deposited. Translaminar fungicides redistribute the fungicide from the upper, sprayed leaf surface to the lower, unsprayed surface. Systemic fungicides are taken up and redistributed through the xylem vessels. Few fungicides move to all parts of a plant. Some are locally systemic, and some move upward. Most fungicides that can be bought retail are sold in liquid form, the active ingredient being present at 0.08% i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron Tris(diethyldithiocarbamate)
Iron tris(diethyldithiocarbamate) is the coordination complex of iron with diethyldithiocarbamate with the formula Fe(S2CNEt2)3 (Et = ethyl). It is a black solid that is soluble in organic solvents. Synthesis, structure, bonding Iron tris(dithiocarbamate)s are typically are prepared by salt metathesis reactions. Iron tris(diethyldithiocarbamate) is an octahedral coordination complex of iron(III) with idealized D3 symmetry. The phenomenon of spin crossover (SCO) was first reported in 1931 by Cambi ''et al.'' who observed anomalous magnetic behavior for the tris(N,N-dialkyldithiocarbamatoiron(III) complexes. The magnetism of these complexes are sensitive to the nature of the amine substituents as well as to temperature. This behavior is consistent with an equilibrium between two or more spin states. In the case of Fe(Et2dtc)3, X-ray crystallography reveals that the Fe-S bonds are 231 pm at 79K but 356 pm at 297 K. These data indicate a low-spin configuration at low temperatures a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coordination Complex
A coordination complex is a chemical compound consisting of a central atom or ion, which is usually metallic and is called the ''coordination centre'', and a surrounding array of chemical bond, bound molecules or ions, that are in turn known as ''ligands'' or complexing agents. Many metal-containing chemical compound, compounds, especially those that include transition metals (elements like titanium that belong to the periodic table's d-block), are coordination complexes. Nomenclature and terminology Coordination complexes are so pervasive that their structures and reactions are described in many ways, sometimes confusingly. The atom within a ligand that is bonded to the central metal atom or ion is called the donor atom. In a typical complex, a metal ion is bonded to several donor atoms, which can be the same or different. A Ligand#Polydentate and polyhapto ligand motifs and nomenclature, polydentate (multiple bonded) ligand is a molecule or ion that bonds to the central atom ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Permissible Exposure Limit
The permissible exposure limit (PEL or OSHA PEL) is a legal limit in the United States for exposure of an employee to a chemical substance or physical agents such as high level noise. Permissible exposure limits were established by the Occupational Safety and Health Administration (OSHA). Most of OSHA's PELs were issued shortly after the adoption of the Occupational Safety and Health (OSH) Act in 1970. Chemical regulation is sometimes expressed in parts per million (ppm), but often in milligrams per cubic meter (mg/m3). Units of measure for physical agents such as noise are specific to the agent. A PEL is usually given as a time-weighted average (TWA), although some are short-term exposure limits (STEL) or ceiling limits. A TWA is the average exposure over a specified period, usually a nominal eight hours. This means that for limited periods, a worker may be exposed to concentration excursions higher than the PEL as long as the TWA is not exceeded and any applicable excursion l ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dithiocarbamates
In organic chemistry, a dithiocarbamate is a chemical compound with the general formula . It contains the functional group with the Chemical structure, structure . It is the analog of a carbamate in which both oxygen atoms are replaced by sulfur atoms (when only one oxygen is replaced the result is thiocarbamate). Dithiocarbamate also refers to the dithiocarbamate ion and its salts. A common example is sodium diethyldithiocarbamate . Dithiocarbamates and their derivatives are widely used in the vulcanization of rubber. Formation Many secondary amines react with carbon disulfide and sodium hydroxide to form dithiocarbamate salts: : Ammonia reacts with carbon disulfide, similarly, to give ammonium dithiocarbamate: : Dithiocarbamate salts are pale colored solids that are soluble in water and Solvent, polar organic solvents. Dithiocarbamic acid A primary amine and carbon disulfide react to give a dithiocarbamic acid: : In the presence of diimides or pyridine, these acids convert t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nickel Bis(dimethyldithiocarbamate)
Nickel bis(dimethyldithiocarbamate) is the coordination complex on nickel and dimethyldithiocarbamate, with the formula Ni(S2CNMe2)2 (Me = methyl). It is the prototype for a large number of square planar bis(dialkhyldithiocarbamate)s of nickel(II), which feature diverse organic substituents. Nickel bis(dimethyldithiocarbamate) has been marketed as a fungicide, and related complexes are used as stabilizers in polymers. Preparation and structure The compound precipitates as a light green solid upon combining aqueous solutions of nickel(II) salts and sodium dimethyldithiocarbamate. In terms of structure and bonding, the nickel is square planar, and the complex is diamagnetic. The structure of the closely related nickel bis(diethyldithiocarbamate) has been determined by X-ray crystallography. Oxidation of nickel bis(dieethyldithiocarbamate) gives the red-brown nickel(IV) complex .{{cite journal , last1=Mazumder , first1=Md. Motiur R. , last2=Dalpati , first2=Niharika , last3=Pokk ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Dimethyldithiocarbamate
Zinc dimethyldithiocarbamate is a coordination complex of zinc with dimethyldithiocarbamate. It is a pale yellow solid that is used as a fungicide, the sulfur vulcanization of rubber, and other industrial applications. Applications Known as ziram in agriculture, it was introduced in the United States in 1960 as a broad-spectrum fungicide. It was used to address scab on apples and pears, leaf curl in peaches, and anthracnose and blight in tomatoes. In 1981, additional uses for ziram were approved, including the prevention of leaf blight and scab on almonds, shot-hole in apricots, brown rot and leaf spot in cherries, and scab and anthracnose in pecans. Ziram also began to be used on residential ornaments as a bird and mammal repellent. As a protectant fungicide, it is active on the plant’s surface where it forms a chemical barrier between the plant and a fungus. A protectant fungicide is not absorbed into the plant and must be applied prior to infection. Ziram can either be direct ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
National Institute For Occupational Safety And Health
The National Institute for Occupational Safety and Health (NIOSH, ) is the List of United States federal agencies, United States federal agency responsible for conducting research and making recommendations for the prevention of work-related occupational injury, injury, occupational disease, illness, disability, and occupational fatality, death. Its functions include gathering information, conducting scientific research both in the laboratory and in the field, and translating the knowledge gained into products and services.About NIOSH National Institute for Occupational Safety and Health. Among NIOSH's programs are determination of recommended exposure limits for toxic chemicals and other hazards, field research such as the Health Hazard Evaluation Program, epidemiology and health surveillance programs such as the National Firefighter Re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Occupational Safety And Health Administration
The Occupational Safety and Health Administration (OSHA; ) is a regulatory agency of the United States Department of Labor that originally had federal visitorial powers to inspect and examine workplaces. The United States Congress established the agency under the Occupational Safety and Health Act (OSH Act), which President Richard Nixon, Richard M. Nixon signed into law on December 29, 1970. OSHA's mission is to "assure safe and healthy working conditions for working men and women by setting and enforcing standards and by providing training, outreach, education, and assistance." The agency is also charged with enforcing a variety of whistleblower statutes and regulations. OSHA's workplace safety inspections have been shown to reduce injury rates and injury costs without adverse effects on employment, sales, credit ratings, or firm survival. History The Bureau of Labor Standards of the Department of Labor has worked on some work safety issues since its creation in 1934. Economi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dimethyldithiocarbamate
Sodium dimethyldithiocarbamate is the organosulfur compound with the formula . It is one of the simplest organic dithiocarbamates. It is a white or pale yellow, water soluble solid. The compound is a precursor to fungicides and rubber chemicals. Preparation Sodium dimethyldithiocarbamate typically crystallizes from water as the dihydrate NaS2CN(CH3)2.2H2O. The anhydrous salt and the trihydrate are often used interchangeably. Sodium dimethyldithiocarbamate is obtained by treating carbon disulfide with dimethylamine in the presence of sodium hydroxide: :CS2 + HN(CH3)2 + NaOH → NaS2CN(CH3)2 + H2O Other dithiocarbamates can be prepared similarly from secondary amines and carbon disulfide. They are used as chelating agents for transition metal ions and as precursors to herbicides and vulcanization reagents. Uses left, 222px, Iron tris(dimethyldithiocarbamate) (Fe(S2CNMe2)3) is illustrative of hundreds of known dithiocarbamate complexes. It is a component of various pe ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrochloric Acid
Hydrochloric acid, also known as muriatic acid or spirits of salt, is an aqueous solution of hydrogen chloride (HCl). It is a colorless solution with a distinctive pungency, pungent smell. It is classified as a acid strength, strong acid. It is a component of the gastric acid in the digestive systems of most animal species, including humans. Hydrochloric acid is an important laboratory reagent and industrial chemical. Etymology Because it was produced from halite, rock salt according to the methods of Johann Rudolph Glauber, hydrochloric acid was historically called by European alchemists ''spirits of salt'' or ''acidum salis'' (salt acid). Both names are still used, especially in other languages, such as , , , , , , , , , , (''ensan''), zh, 盐酸 (''yánsuān''), and (''yeomsan''). Gaseous HCl was called ''marine acid air''. The name ''muriatic acid'' has the same origin (''muriatic'' means "pertaining to brine or salt", hence ''muriate'' means hydrochloride), and this ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Trap
In chemistry, a chemical trap is a chemical compound that is used to detect unstable compounds. The method relies on efficiency of bimolecular reactions with reagents to produce a more easily characterize trapped product. In some cases, the trapping agent is used in large excess. Case studies Cyclobutadiene A famous example is the detection of cyclobutadiene released upon oxidation of cyclobutadieneiron tricarbonyl. When this degradation is conducted in the presence of an alkyne, the cyclobutadiene is trapped as a bicyclohexadiene. The requirement for this trapping experiment is that the oxidant (ceric ammonium nitrate) and the trapping agent be mutually compatible. : Diphosphorus Diphosphorus is an old target of chemists since it is the heavy analogue of N2. Its fleeting existence is inferred by the controlled degradation of certain niobium complexes in the presence of trapping agents. Again, a Diels-Alder strategy is employed in the trapping: : Silylene Another classic but e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |