|
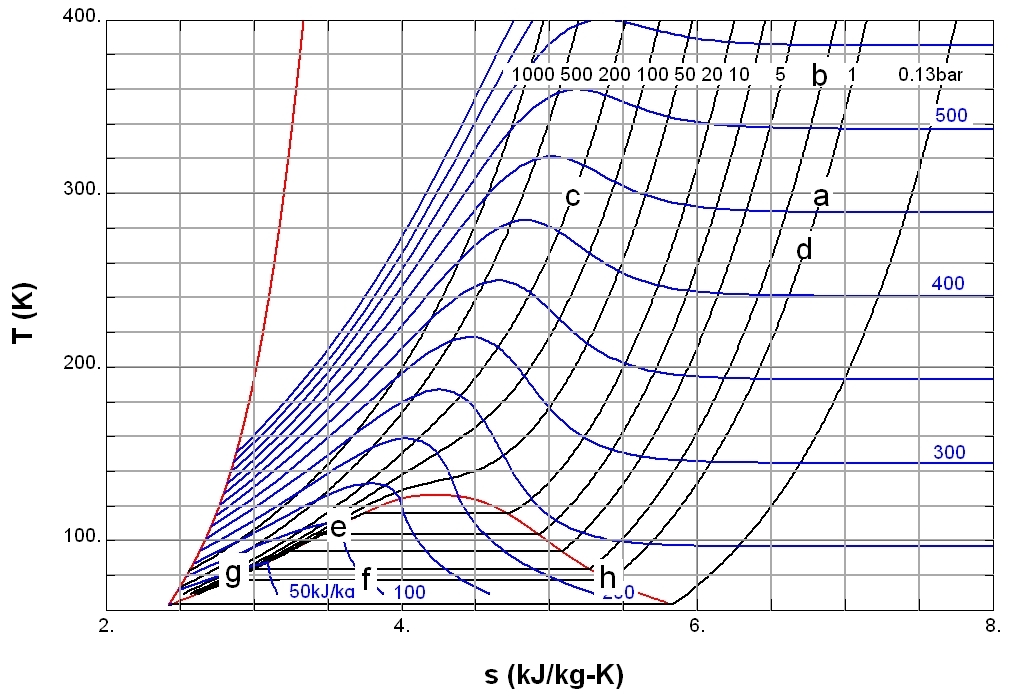
Compressed Liquid
A compressed fluid (also called a compressed or unsaturated liquid, subcooled fluid or liquid) is a fluid under Fluid mechanics, mechanical or thermodynamics, thermodynamic conditions that force it to be a liquid. At a given pressure, a fluid is a compressed fluid if it is at a temperature lower than the saturation temperature. This is the case, for example, for liquid water at atmospheric pressure and room temperature. In a plot that compares absolute pressure, pressure and specific volume (commonly called a Pressure volume diagram, p-v diagram), compressed fluid is the state to the left of the saturation curve. Conditions that cause a fluid to be compressed include: * Specific volume and enthalpy inferior to that of a saturated liquid; * Temperature below the saturation temperature; * Pressure above the saturation pressure. The term ''compressed liquid'' emphasizes that the pressure is greater than the saturation pressure for the given temperature. Compressed liquid propertie ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Room Temperature
Room temperature, colloquially, denotes the range of air temperatures most people find comfortable indoors while dressed in typical clothing. Comfortable temperatures can be extended beyond this range depending on humidity, air circulation, and other factors. In certain fields, like science and engineering, and within a particular context, room temperature can mean different agreed-upon ranges. In contrast, ambient temperature is the actual temperature, as measured by a thermometer, of the air (or other medium and surroundings) in any particular place. The ambient temperature (e.g. an unheated room in winter) may be very different from an ideal ''room temperature''. Food and beverages may be served at "room temperature", meaning neither heated nor cooled. Comfort temperatures Comfort temperature is interchangeable with neutral temperature in the scientific literature, which can be calculated through regression analysis between thermal sensation votes and indoor temperature. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Saturation Pressure
Vapor pressure or equilibrium vapor pressure is the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phases (solid or liquid) at a given temperature in a closed system. The equilibrium vapor pressure is an indication of a liquid's thermodynamic tendency to evaporate. It relates to the balance of particles escaping from the liquid (or solid) in equilibrium with those in a coexisting vapor phase. A substance with a high vapor pressure at normal temperatures is often referred to as '' volatile''. The pressure exhibited by vapor present above a liquid surface is known as vapor pressure. As the temperature of a liquid increases, the attractive interactions between liquid molecules become less significant in comparison to the entropy of those molecules in the gas phase, increasing the vapor pressure. Thus, liquids with strong intermolecular interactions are likely to have smaller vapor pressures, with the reverse true for weaker interactions. The vapor pr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Saturated Liquid
Saturation, saturated, unsaturation or unsaturated may refer to: Chemistry *Saturated and unsaturated compounds, a classification of compounds related to their ability to resist addition reactions ** Degree of unsaturation **Saturated fat or saturated fatty acid **Unsaturated fat or unsaturated fatty acid ** Non-susceptibility of an organometallic compound to oxidative addition * Saturation of protein binding sites * Saturation of enzymes with a substrate * Saturation of a solute in a solution, as related to the solute's maximum solubility at equilibrium ** Supersaturation, where the concentration of a solute exceeds its maximum solubility at equilibrium ** Undersaturation, where the concentration of a solute is less than its maximum solubility at equilibrium Biology * Oxygen saturation, a clinical measure of the amount of oxygen in a patient's blood * Saturation pollination, a pollination technique * Saturated mutagenesis, a form of site-directed mutagenesis * Saturation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enthalpy
Enthalpy () is the sum of a thermodynamic system's internal energy and the product of its pressure and volume. It is a state function in thermodynamics used in many measurements in chemical, biological, and physical systems at a constant external pressure, which is conveniently provided by the large ambient atmosphere. The pressure–volume term expresses the work (physics), work W that was done against constant external pressure P_\text to establish the system's physical dimensions from V_\text=0 to some final volume V_\text (as W=P_\text\Delta V), i.e. to make room for it by displacing its surroundings. The pressure-volume term is very small for solids and liquids at common conditions, and fairly small for gases. Therefore, enthalpy is a stand-in for energy in chemical systems; Bond energy, bond, Lattice energy, lattice, solvation, and other chemical "energies" are actually enthalpy differences. As a state function, enthalpy depends only on the final configuration of internal e ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pressure Volume Diagram
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country and even by industry. Further, both spellings are often used ''within'' a particular industry or country. Industries in British English-speaking countries typically use the "gauge" spelling. is the pressure relative to the ambient pressure. Various units are used to express pressure. Some of these derive from a unit of force divided by a unit of area; the SI unit of pressure, the pascal (Pa), for example, is one newton per square metre (N/m2); similarly, the pound-force per square inch (psi, symbol lbf/in2) is the traditional unit of pressure in the imperial and US customary systems. Pressure may also be expressed in terms of standard atmospheric pressure; the unit atmosphere (atm) is equal to this pressure, and the torr is defined as of th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Specific Volume
In thermodynamics, the specific volume of a substance (symbol: , nu) is the quotient of the substance's volume () to its mass (): :\nu = \frac It is a mass-specific intrinsic property of the substance. It is the reciprocal of density (rho) and it is also related to the molar volume and molar mass: :\nu = \rho^ = \frac The standard unit of specific volume is cubic meters per kilogram (m3/kg), but other units include ft3/lb, ft3/slug, or mL/g. Specific volume for an ideal gas is related to the molar gas constant () and the gas's temperature (), pressure (), and molar mass (): : \nu = \frac It's based on the ideal gas law, PV = , and the amount of substance In chemistry, the amount of substance (symbol ) in a given sample of matter is defined as a ratio () between the particle number, number of elementary entities () and the Avogadro constant (). The unit of amount of substance in the International ..., n = m/M Applications Specific volume is commonly applied to: * ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Absolute Pressure
Pressure measurement is the measurement of an applied force by a fluid (liquid or gas) on a surface. Pressure is typically measured in unit of measurement, units of force per unit of surface area. Many techniques have been developed for the measurement of pressure and vacuum. Instruments used to measure and display pressure mechanically are called pressure gauges, vacuum gauges or compound gauges (vacuum & pressure). The widely used Bourdon gauge is a mechanical device, which both measures and indicates and is probably the best known type of gauge. A vacuum gauge is used to measure pressures lower than the ambient atmospheric pressure, which is set as the zero point, in negative values (for instance, −1 bar or −760 mmHg equals total vacuum). Most gauges measure pressure relative to atmospheric pressure as the zero point, so this form of reading is simply referred to as "gauge pressure". However, anything greater than total vacuum is technically a form of pressure. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Atmospheric Pressure
Atmospheric pressure, also known as air pressure or barometric pressure (after the barometer), is the pressure within the atmosphere of Earth. The standard atmosphere (symbol: atm) is a unit of pressure defined as , which is equivalent to 1,013.25 millibars, 760 mm Hg, 29.9212 inchesHg, or 14.696 psi.International Civil Aviation Organization. ''Manual of the ICAO Standard Atmosphere'', Doc 7488-CD, Third Edition, 1993. . The atm unit is roughly equivalent to the mean sea-level atmospheric pressure on Earth; that is, the Earth's atmospheric pressure at sea level is approximately 1 atm. In most circumstances, atmospheric pressure is closely approximated by the hydrostatic pressure caused by the weight of air above the measurement point. As elevation increases, there is less overlying atmospheric mass, so atmospheric pressure decreases with increasing elevation. Because the atmosphere is thin relative to the Earth's radius—especially the dense atmospheric layer at low altitu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Saturation Temperature
Saturation, saturated, unsaturation or unsaturated may refer to: Chemistry *Saturated and unsaturated compounds, a classification of compounds related to their ability to resist addition reactions ** Degree of unsaturation **Saturated fat or saturated fatty acid **Unsaturated fat or unsaturated fatty acid ** Non-susceptibility of an organometallic compound to oxidative addition * Saturation of protein binding sites * Saturation of enzymes with a substrate * Saturation of a solute in a solution, as related to the solute's maximum solubility at equilibrium ** Supersaturation, where the concentration of a solute exceeds its maximum solubility at equilibrium ** Undersaturation, where the concentration of a solute is less than its maximum solubility at equilibrium Biology * Oxygen saturation, a clinical measure of the amount of oxygen in a patient's blood * Saturation pollination, a pollination technique * Saturated mutagenesis, a form of site-directed mutagenesis * Saturation ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Temperature
Temperature is a physical quantity that quantitatively expresses the attribute of hotness or coldness. Temperature is measurement, measured with a thermometer. It reflects the average kinetic energy of the vibrating and colliding atoms making up a substance. Thermometers are calibrated in various temperature scales that historically have relied on various reference points and thermometric substances for definition. The most common scales are the Celsius scale with the unit symbol °C (formerly called ''centigrade''), the Fahrenheit scale (°F), and the Kelvin scale (K), with the third being used predominantly for scientific purposes. The kelvin is one of the seven base units in the International System of Units (SI). Absolute zero, i.e., zero kelvin or −273.15 °C, is the lowest point in the thermodynamic temperature scale. Experimentally, it can be approached very closely but not actually reached, as recognized in the third law of thermodynamics. It would be impossible ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |