|
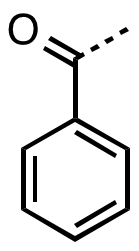
Benzoyl
In organic chemistry, benzoyl (, ) is the functional group with the formula and structure . It can be viewed as benzaldehyde missing one hydrogen. The benzoyl group has a mass of 105 amu. The term "benzoyl" should not be confused with benzyl, which has the formula . The benzoyl group is given the symbol "Bz" whereas benzyl is commonly abbreviated "Bn". Sources Benzoyl chloride is a favored source of benzoyl groups, being used to prepare benzoyl ketones, benzamides (benzoyl amides), and benzoate esters. The source of many naturally occurring benzoyl compounds is the thioester benzoyl-CoA. Irradiation of benzil generates benzoyl radicals, which have the formula PhCO. Benzoyl compounds Many ketones contain the benzoyl group. They have the formula C6H5CO–R, an important example being benzophenone. Benzoyl esters and amides are common in organic chemistry. The esters are used as a protecting groups in organic synthesis, which can be easily removed by hydrolysis in dilute ba ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzoyl Chloride
Benzoyl chloride, also known as benzenecarbonyl chloride, is an organochlorine compound with the formula . It is a colourless, fuming liquid with an irritating odour, and consists of a benzene ring () with an acyl chloride () substituent. It is mainly useful for the production of peroxides but is generally useful in other areas such as in the preparation of dyes, perfumes, pharmaceuticals, and resins. Preparation Benzoyl chloride is produced from benzotrichloride using either water or benzoic acid: : : As with other acyl chlorides, it can be generated from the parent acid and standard chlorinating agents such as phosphorus pentachloride, thionyl chloride, and oxalyl chloride. It was first prepared by treatment of benzaldehyde with chlorine. An early method for production of benzoyl chloride involved chlorination of benzyl alcohol. Reactions It reacts with water to produce hydrochloric acid and benzoic acid: : Benzoyl chloride is a typical acyl chloride. It reacts with alco ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzoyl-CoA
Benzoyl-CoA is the thioester derived from benzoic acid and coenzyme A. The term benzoyl-CoA also include diverse conjugates of coenzyme A and aromatic carboxylic acids. Benzoate, vanillin, anthranilic acid, 4-ethylphenol, p-cresol, phenol, aniline, terephthalic acid, phenylalanine.html" ;"title="-hydroxybenzoic acid, and phenylalanine">-hydroxybenzoic acid, and phenylalanine are all metabolized to benzoyl-CoA. Additionally, cinnamic acid, p-coumaric acid, ferulic acid, toluene, caffeic acid, benzyl alcohol, and mandelic acid are suspected to be processed similarly. As substrate for reductases Benzoyl-CoA is a substrate for diverse reductases: 4-hydroxybenzoyl-CoA reductase, benzoyl-CoA reductase, benzoyl-CoA 3-monooxygenase, benzoate-CoA ligase, 2alpha-hydroxytaxane 2-O-benzoyltransferase, anthranilate N-benzoyltransferase, biphenyl synthase, glycine N-benzoyltransferase, ornithine N-benzoyltransferase and phenylglyoxylate dehydrogenase (acylating). Benzoyl-Co ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzophenone
Benzophenone is a naturally occurring organic compound with the formula (C6H5)2CO, generally abbreviated Ph2CO. Benzophenone has been found in some fungi, fruits and plants, including grapes. It is a white solid with a low melting point and rose-like odor that is soluble in organic solvents. Benzophenone is the simplest diaromatic ketone. It is a widely used building block in organic chemistry, being the parent diarylketone. History Carl Graebe of the University of Königsberg, in an early literature report from 1874, described working with benzophenone. Uses Benzophenone can be used as a photo initiator in ultraviolet (UV)-curing applications such as inks, imaging, and clear coatings in the printing industry. Benzophenone prevents UV light from damaging scents and colors in products such as perfumes and soaps. Benzophenone can also be added to plastic packaging as a UV blocker to prevent photo-degradation of the packaging polymers or its contents. Its use allows manufactur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pteris Ensiformis
''Pteris ensiformis'', the slender brake, silver lace fern, sword brake fern, or slender brake fern, is a plant species of the genus Pteris in the family Pteridaceae. It is found in Asia and the Pacific. Uses Beverages It is the most common ingredient of traditional herbal drinks in Taiwan containing different phenolic compounds : kaempferol 3-O-α-l-rhamnopyranoside-7-O-[α-d-apiofuranosyl-(1-2)-β-d-glucopyranoside], 7-O-caffeic acid, caffeoylhydroxymaltol 3-O-β-d-glucopyranoside, hispidin 4-O-β-d-glucopyranoside, kaempferol 3-O-α-l-rhamnopyranoside-7-O-β-d-glucopyranoside, caffeic acid, 5-O-caffeoylquinic acid, 3,5-di-O-caffeoylquinic acid and 4,5-di-O-caffeoylquinic acid. This plant is resistant to arsenic-induced oxidative stress. Benzoyl-beta-D-glucoside, as well as pterosin sesquiterpenes can be found in ''P. ensiformis''. Cultivation ''Pteris ensiformis'' is cultivated as an ornamental plant for tropical and subtropical climate gardens, and as a house plant. ;Culti ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protecting Group
A protecting group or protective group is introduced into a molecule by chemical modification of a functional group to obtain chemoselectivity in a subsequent chemical reaction. It plays an important role in multistep organic synthesis. In many preparations of delicate organic compounds, specific parts of the molecules cannot survive the required reagents or chemical environments. These parts (functional groups) must be protected. For example, lithium aluminium hydride is a highly reactive reagent that usefully reduces esters to alcohols. It always reacts with carbonyl groups, and cannot be discouraged by any means. When an ester must be reduced in the presence of a carbonyl, hydride attack on the carbonyl must be prevented. One way to do so converts the carbonyl into an acetal, which does not react with hydrides. The acetal is then called a protecting group for the carbonyl. After the hydride step is complete, aqueous acid removes the acetal, restoring the carbonyl. This step ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Basic (chemistry)
In chemistry, there are three definitions in common use of the word "base": '' Arrhenius bases'', '' Brønsted bases'', and ''Lewis bases''. All definitions agree that bases are substances that react with acids, as originally proposed by G.-F. Rouelle in the mid-18th century. In 1884, Svante Arrhenius proposed that a base is a substance which dissociates in aqueous solution to form hydroxide ions OH−. These ions can react with hydrogen ions (H+ according to Arrhenius) from the dissociation of acids to form water in an acid–base reaction. A base was therefore a metal hydroxide such as NaOH or Ca(OH)2. Such aqueous hydroxide solutions were also described by certain characteristic properties. They are slippery to the touch, can taste bitter and change the color of pH indicators (e.g., turn red litmus paper blue). In water, by altering the autoionization equilibrium, bases yield solutions in which the hydrogen ion activity is lower than it is in pure water, i.e., the w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrolysis
Hydrolysis (; ) is any chemical reaction in which a molecule of water breaks one or more chemical bonds. The term is used broadly for substitution reaction, substitution, elimination reaction, elimination, and solvation reactions in which water is the nucleophile. Biological hydrolysis is the cleavage of Biomolecule, biomolecules where a water molecule is consumed to effect the separation of a larger molecule into component parts. When a carbohydrate is broken into its component sugar molecules by hydrolysis (e.g., sucrose being broken down into glucose and fructose), this is recognized as saccharification. Hydrolysis reactions can be the reverse of a condensation reaction in which two molecules join into a larger one and eject a water molecule. Thus hydrolysis adds water to break down, whereas condensation builds up by removing water. Types Usually hydrolysis is a chemical process in which a molecule of water is added to a substance. Sometimes this addition causes both the su ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Synthesis
Organic synthesis is a branch of chemical synthesis concerned with the construction of organic compounds. Organic compounds are molecules consisting of combinations of covalently-linked hydrogen, carbon, oxygen, and nitrogen atoms. Within the general subject of organic synthesis, there are many different types of synthetic routes that can be completed including total synthesis, Enantioselective synthesis, stereoselective synthesis, automated synthesis, and many more. Additionally, in understanding organic synthesis it is necessary to be familiar with the methodology, techniques, and applications of the subject. Total synthesis A total synthesis refers to the complete chemical synthesis of molecules from simple, Precursor (chemistry), natural precursors. Total synthesis is accomplished either via a linear or convergent approach. In a Linear synthesis, ''linear'' synthesis—often adequate for simple structures—several steps are performed sequentially until the molecule is com ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amide
In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a chemical compound, compound with the general formula , where R, R', and R″ represent any group, typically organyl functional group, groups or hydrogen atoms. The amide group is called a peptide bond when it is part of the Polymer backbone, main chain of a protein, and an isopeptide bond when it occurs in a side chain, as in asparagine and glutamine. It can be viewed as a Derivative (chemistry), derivative of a carboxylic acid () with the hydroxyl group () replaced by an amino group (); or, equivalently, an acyl group, acyl (alkanoyl) group () joined to an amino group. Common amides are formamide (), acetamide (), benzamide (), and dimethylformamide (). Some uncommon examples of amides are ''N''-chloroacetamide () and chloroformamide (). Amides are qualified as primary (chemistry), primary, secondary (chemistry), secondary, and tertiary (chemistry), tertiary according to the number of acyl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ester
In chemistry, an ester is a compound derived from an acid (either organic or inorganic) in which the hydrogen atom (H) of at least one acidic hydroxyl group () of that acid is replaced by an organyl group (R). These compounds contain a distinctive functional group. Analogues derived from oxygen replaced by other chalcogens belong to the ester category as well. According to some authors, organyl derivatives of acidic hydrogen of other acids are esters as well (e.g. amides), but not according to the IUPAC. Glycerides are fatty acid esters of glycerol; they are important in biology, being one of the main classes of lipids and comprising the bulk of animal fats and vegetable oils. Lactones are cyclic carboxylic esters; naturally occurring lactones are mainly 5- and 6-membered ring lactones. Lactones contribute to the aroma of fruits, butter, cheese, vegetables like celery and other foods. Esters can be formed from oxoacids (e.g. esters of acetic acid, carbonic acid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |