|
Alpha-olefin
In organic chemistry, terminal alkenes (alpha-olefins, α-olefins, or 1-alkenes) are a family of organic compounds which are alkenes (also known as olefins) with a chemical formula , distinguished by having a double bond at the primary, Alpha and beta carbon, alpha (α), or Locant#Numeric locants, 1- position.''Petrochemicals in Nontechnical Language'', 3rd Edition, Donald L. Burdick and William L. Leffler, This location of a double bond enhances the reactivity of the compound and makes it useful for a number of applications. Classification There are two types of alpha-olefins, branched alpha olefin, branched and linear alpha olefin, linear (or normal). The chemical properties of branched alpha-olefins with a branch at either the second (vinylidene) or the third carbon number are significantly different from the properties of linear alpha-olefins and those with branches on the fourth carbon number and further from the start of the chain. Examples of linear alpha-olefins are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Linear Alpha Olefin
Straight-chain terminal alkenes, also called linear alpha olefins (LAO) or normal alpha olefins (NAO), are alkenes (olefins) having a chemical formula , distinguished from other alkenes with a similar molecular formula by being terminal alkenes, in which the double bond occurs at the alpha (α-, 1- or primary) position, and by having a linear (unbranched) hydrocarbon chain. Linear alpha olefins are a range of industrially important alpha-olefins, including 1-butene, 1-hexene, 1-octene, 1-decene, 1-dodecene, 1-tetradecene, 1-hexadecene, 1-octadecene and higher olefin blends of C20-C24, C24-C30, and C20-C30 ranges. Synthesis Overview Industrially, linear alpha olefins are commonly manufactured by two main routes: oligomerization of ethylene and by Fischer–Tropsch synthesis followed by purification. Another route to linear alpha olefins which has been used commercially on small scale is dehydration of alcohols. Prior to about the 1970s, linear alpha olefins wer ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
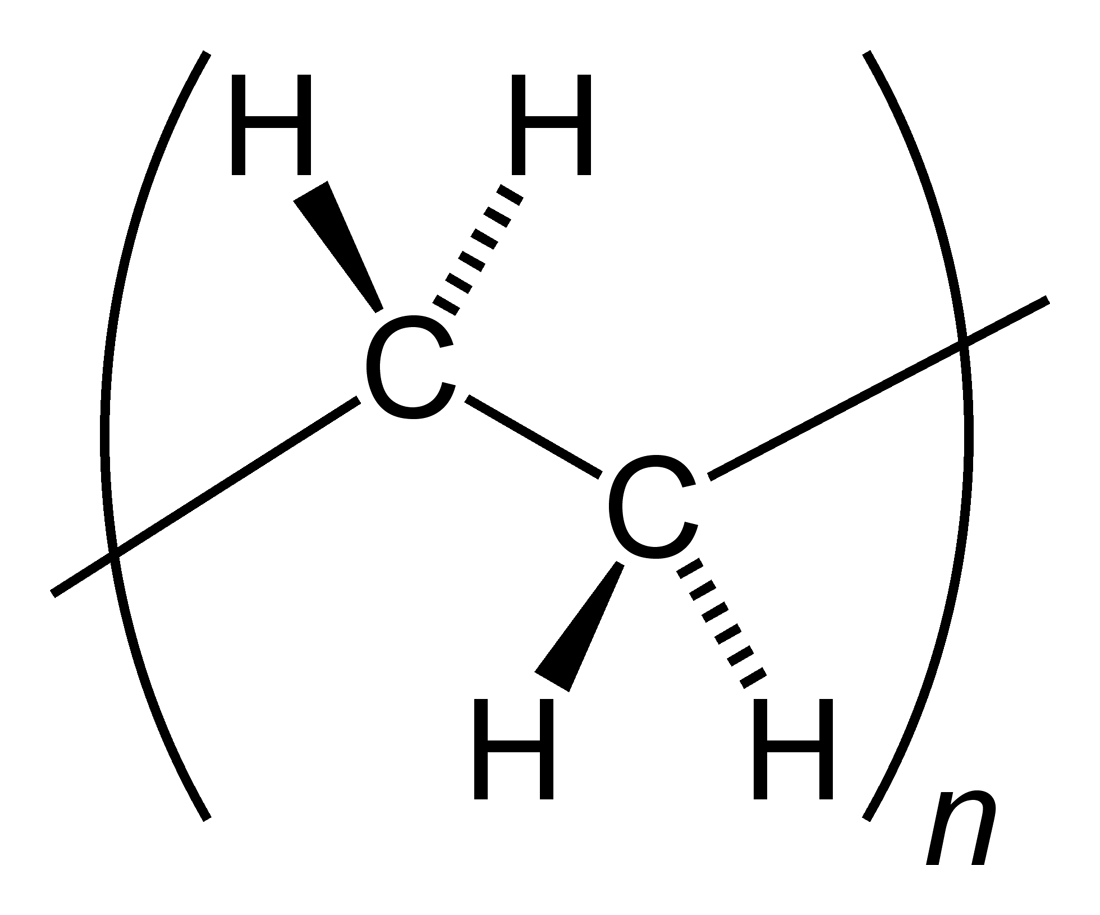
Polyethylene
Polyethylene or polythene (abbreviated PE; IUPAC name polyethene or poly(methylene)) is the most commonly produced plastic. It is a polymer, primarily used for packaging (plastic bags, plastic films, geomembranes and containers including bottles, cups, jars, etc.). , over 100 million tonnes of polyethylene resins are being produced annually, accounting for 34% of the total plastics market. Many kinds of polyethylene are known, with most having the chemical formula (C2H4)''n''. PE is usually a mixture of similar polymers of ethylene, with various values of ''n''. It can be ''low-density'' or ''high-density'' and many variations thereof. Its properties can be modified further by crosslinking or copolymerization. All forms are nontoxic as well as chemically resilient, contributing to polyethylene's popularity as a multi-use plastic. However, polyethylene's chemical resilience also makes it a long-lived and decomposition-resistant pollutant when disposed of improperly. Being a h ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
But-1-ene
1-Butene (IUPAC name: But-1-ene, also known as 1-butylene) is the organic compound with the formula CH3CH2CH=CH2. It is a colorless gas, but easily condensed to give a colorless liquid. It is classified as a linear alpha-olefin (terminal alkene). It is one of the isomers of butene (butylene). It is a precursor to diverse products. Reactions Polymerization of 1-butene gives polybutylene, which is used to make piping for domestic plumbing. Another application is as a comonomer in the production of certain kinds of polyethylene, such as linear low-density polyethylene (LLDPE). It has also been used as a precursor to polypropylene resins, butylene oxide, and butanone. Manufacturing 1-Butene is produced by separation from crude C4 refinery streams and by ethylene dimerization In organic chemistry, hydrovinylation is the formal insertion of an alkene into the C-H bond of ethylene (): : The more general reaction, hydroalkenylation, is the formal insertion of an alkene into the C-H b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Titanium
Titanium is a chemical element; it has symbol Ti and atomic number 22. Found in nature only as an oxide, it can be reduced to produce a lustrous transition metal with a silver color, low density, and high strength, resistant to corrosion in sea water, aqua regia, and chlorine. Titanium was discovered in Cornwall, Great Britain, by William Gregor in 1791 and was named by Martin Heinrich Klaproth after the Titans of Greek mythology. The element occurs within a number of minerals, principally rutile and ilmenite, which are widely distributed in the Earth's crust and lithosphere; it is found in almost all living things, as well as bodies of water, rocks, and soils. The metal is extracted from its principal mineral ores by the Kroll and Hunter processes. The most common compound, titanium dioxide (TiO2), is a popular photocatalyst and is used in the manufacture of white pigments. Other compounds include titanium tetrachloride (TiCl4), a component of smoke screens and cata ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Comonomer
In polymer chemistry, a comonomer refers to a polymerizable precursor to a copolymer aside from the principal monomer. In some cases, only small amounts of a comonomer are employed, in other cases substantial amounts of comonomers are used. Furthermore, in some cases, the comonomers are statistically incorporated within the polymer chain, whereas in other cases, they aggregate. The distribution of comonomers is referred to as the " blockiness" of a copolymer. Polyolefins 1-Octene, 1-hexene, and 1-butene are used comonomers in the manufacture of polyethylene Polyethylene or polythene (abbreviated PE; IUPAC name polyethene or poly(methylene)) is the most commonly produced plastic. It is a polymer, primarily used for packaging (plastic bags, plastic films, geomembranes and containers including bott ...s. The advantages to such copolymers has led to a focus on catalysts that facilitate the incorporation of these comonomers, e.g., constrained geometry complexes. Comonome ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fatty Amine
In chemistry, a fatty amine is loosely defined as any amine possessing a mostly linear hydrocarbon chain of eight or more carbon atoms. They are typically prepared from the more abundant fatty acids, with vegetable or seed-oils being the ultimate starting material. As such they are often mixtures of chain lengths, ranging up to about C22. They can be classified as oleochemicals. Commercially important members include coco amine, oleylamine, tallow amine, and soya amine. These compounds and their derivatives are used as fabric softeners, froth flotation agents (purification of ores), corrosion inhibitors, lubricants and friction modifiers. They are also the basis for a variety of cosmetic formulations. Production and reactions Fatty amines are commonly prepared from fatty acids; which are themselves obtained from natural sources, typically seed-oils. The overall reaction is sometimes referred to as the Nitrile Process and begins with a reaction between the fatty acid and ammonia ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fatty Alcohol
Fatty alcohols (or long-chain alcohols) are usually high-molecular mass, straight-chain primary alcohols, but can also range from as few as 4–6 carbon atoms to as many as 22–26, derived from natural fats and oils. The precise chain length varies with the source. Some commercially important fatty alcohols are Lauryl alcohol, lauryl, Stearyl alcohol, stearyl, and oleyl alcohol. They are colourless oily liquids (for smaller carbon numbers) or waxy solids, although impure samples may appear yellow. Fatty alcohols usually have an even number of carbon atoms and a single alcohol group (–OH) attached to the terminal carbon. Some are unsaturated and some are branched. They are widely used in industry. As with fatty acids, they are often referred to generically by the number of carbon atoms in the molecule, such as "a C12 alcohol", that is an alcohol having 12 carbon atoms, for example dodecanol. Production and occurrence Fatty alcohols became commercially available in the early 1900s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fatty Acid
In chemistry, in particular in biochemistry, a fatty acid is a carboxylic acid with an aliphatic chain, which is either saturated and unsaturated compounds#Organic chemistry, saturated or unsaturated. Most naturally occurring fatty acids have an Branched chain fatty acids, unbranched chain of an even number of carbon atoms, from 4 to 28. Fatty acids are a major component of the lipids (up to 70% by weight) in some species such as microalgae but in some other organisms are not found in their standalone form, but instead exist as three main classes of esters: triglycerides, phospholipids, and cholesteryl esters. In any of these forms, fatty acids are both important diet (nutrition), dietary sources of fuel for animals and important structural components for cell (biology), cells. History The concept of fatty acid (''acide gras'') was introduced in 1813 by Michel Eugène Chevreul, though he initially used some variant terms: ''graisse acide'' and ''acide huileux'' ("acid fat" and "oi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Linear Alkylbenzene Sulfonate
Alkylbenzene sulfonates are a class of anionic surfactants, consisting of a hydrophilic sulfonate head-group and a hydrophobic alkylbenzene tail-group. Along with sodium laureth sulfate, they are one of the oldest and most widely used synthetic detergents and may be found in numerous personal-care products (soaps, shampoos, toothpaste etc.) and household-care products (laundry detergent, dishwashing liquid, spray cleaner etc.).Kurt Kosswig,"Surfactants" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, 2005, Weinheim. They were introduced in the 1930s in the form of branched alkylbenzene sulfonates (BAS). However following environmental concerns these were replaced with linear alkylbenzene sulfonates (LAS) during the 1960s. Since then production has increased significantly from about one million tons in 1980, to around 3.5 million tons in 2016, making them most produced anionic surfactant after soaps. Branched alkylbenzene sulfonates Branched alkylbenzene sulfonat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfonation
In organic chemistry, aromatic sulfonation is a reaction in which a hydrogen atom on an arene is replaced by a sulfonic acid () group. Together with nitration and chlorination, aromatic sulfonation is a widely used electrophilic aromatic substitutions. Aryl sulfonic acids are used as detergents, dye, and drugs. : Stoichiometry and mechanism Typical conditions involve heating the aromatic compound with sulfuric acid: : Sulfur trioxide or its protonated derivative is the actual electrophile in this electrophilic aromatic substitution. To drive the equilibrium, dehydrating agents such as thionyl chloride can be added: : Historically, mercurous sulfate has been used to catalyze the reaction. Chlorosulfuric acid is also an effective agent: : In contrast to aromatic nitration and most other electrophilic aromatic substitutions this reaction is reversible. Sulfonation takes place in concentrated acidic conditions and desulfonation is the mode of action in a dilute hot aqueou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Aldehyde
In organic chemistry, an aldehyde () (lat. ''al''cohol ''dehyd''rogenatum, dehydrogenated alcohol) is an organic compound containing a functional group with the structure . The functional group itself (without the "R" side chain) can be referred to as an aldehyde but can also be classified as a formyl group. Aldehydes are a common motif in many chemicals important in technology and biology. Structure and bonding Aldehyde molecules have a central carbon atom that is connected by a double bond to oxygen, a single bond to hydrogen and another single bond to a third substituent, which is carbon or, in the case of formaldehyde, hydrogen. The central carbon is often described as being sp2- hybridized. The aldehyde group is somewhat polar. The bond length is about 120–122 picometers. Physical properties and characterization Aldehydes have properties that are diverse and that depend on the remainder of the molecule. Smaller aldehydes such as formaldehyde and acetaldehyde are solubl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroformylation
In organic chemistry, hydroformylation, also known as oxo synthesis or oxo process, is an industrial process for the production of aldehydes () from alkenes (). This chemical reaction entails the net addition of a formyl group () and a hydrogen atom to a carbon-carbon double bond. This process has undergone continuous growth since its invention: production capacity reached 6.6 tons in 1995. It is important because aldehydes are easily converted into many secondary products. For example, the resultant aldehydes are hydrogenated to Alcohol (chemistry), alcohols that are converted to detergents. Hydroformylation is also used in speciality chemicals, relevant to the organic synthesis of fragrances and pharmaceuticals. The development of hydroformylation is one of the premier achievements of 20th-century Chemical industry, industrial chemistry. The process entails treatment of an alkene typically with high pressures (between 10 and 100 Atmosphere (unit), atmospheres) of carbon monoxi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |