|
Linear Alkylbenzene Sulfonate
Alkylbenzene sulfonates are a class of anionic surfactants, consisting of a hydrophilic sulfonate head-group and a hydrophobic alkylbenzene tail-group. Along with sodium laureth sulfate, they are one of the oldest and most widely used synthetic detergents and may be found in numerous personal-care products (soaps, shampoos, toothpaste etc.) and household-care products (laundry detergent, dishwashing liquid, spray cleaner etc.).Kurt Kosswig,"Surfactants" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, 2005, Weinheim. They were introduced in the 1930s in the form of branched alkylbenzene sulfonates (BAS). However following environmental concerns these were replaced with linear alkylbenzene sulfonates (LAS) during the 1960s. Since then production has increased significantly from about one million tons in 1980, to around 3.5 million tons in 2016, making them most produced anionic surfactant after soaps. Branched alkylbenzene sulfonates Branched alkylbenzene sulfonat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biodegradability
Biodegradation is the breakdown of organic matter by microorganisms, such as bacteria and fungi. It is generally assumed to be a natural process, which differentiates it from composting. Composting is a human-driven process in which biodegradation occurs under a specific set of circumstances. The process of biodegradation is threefold: first an object undergoes biodeterioration, which is the mechanical weakening of its structure; then follows biofragmentation, which is the breakdown of materials by microorganisms; and finally assimilation, which is the incorporation of the old material into new cells. In practice, almost all chemical compounds and materials are subject to biodegradation, the key element being time. Things like vegetables may degrade within days, while glass and some plastics take many millennia to decompose. A standard for biodegradability used by the European Union is that greater than 90% of the original material must be converted into , water and minerals b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1-alkene
In organic chemistry, terminal alkenes (alpha-olefins, α-olefins, or 1-alkenes) are a family of organic compounds which are alkenes (also known as olefins) with a chemical formula , distinguished by having a double bond at the primary, alpha (α), or 1- position.''Petrochemicals in Nontechnical Language'', 3rd Edition, Donald L. Burdick and William L. Leffler, This location of a double bond enhances the reactivity of the compound and makes it useful for a number of applications. Classification There are two types of alpha-olefins, branched and linear (or normal). The chemical properties of branched alpha-olefins with a branch at either the second (vinylidene) or the third carbon number are significantly different from the properties of linear alpha-olefins and those with branches on the fourth carbon number and further from the start of the chain. Examples of linear alpha-olefins are propene, but-1-ene and dec-1-ene. An example of a branched alpha-olefin is isobutylene ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Markovnikov's Rule
In organic chemistry, Markovnikov's rule or Markownikoff's rule describes the outcome of some addition reactions. The rule was formulated by Russian chemist Vladimir Markovnikov in 1870. Explanation The rule states that with the addition of a protic acid HX or other polar reagent to an asymmetric alkene, the acid hydrogen (H) or electropositive part gets attached to the carbon with more hydrogen substituents, and the halide (X) group or electronegative part gets attached to the carbon with more alkyl substituents. This is in contrast to Markovnikov's original definition, in which the rule states that the X component is added to the carbon with the fewest hydrogen atoms while the hydrogen atom is added to the carbon with the greatest number of hydrogen atoms. The same is true when an alkene reacts with water in an additional reaction to form an alcohol that involves carbocation formation. The hydroxyl group (OH) bonds to the carbon that has the greater number of carbon-carbon ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkene
In organic chemistry, an alkene, or olefin, is a hydrocarbon containing a carbon–carbon double bond. The double bond may be internal or at the terminal position. Terminal alkenes are also known as Alpha-olefin, α-olefins. The International Union of Pure and Applied Chemistry (IUPAC) Preferred IUPAC name, recommends using the name "alkene" only for Open-chain compound, acyclic hydrocarbons with just one double bond; alkadiene, alkatriene, etc., or polyene for acyclic hydrocarbons with two or more double bonds; cycloalkene, cycloalkadiene, etc. for Cyclic compound, cyclic ones; and "olefin" for the general class – cyclic or acyclic, with one or more double bonds. Acyclic alkenes, with only one double bond and no other functional groups (also known as mono-enes) form a homologous series of hydrocarbons with the general formula with ''n'' being a >1 natural number (which is two hydrogens less than the corresponding alkane). When ''n'' is four or more, isomers are possible, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sodium Hydroxide
Sodium hydroxide, also known as lye and caustic soda, is an inorganic compound with the formula . It is a white solid ionic compound consisting of sodium cations and hydroxide anions . Sodium hydroxide is a highly corrosive base (chemistry), base and alkali that decomposes lipids and proteins at ambient temperatures and at high concentrations may cause severe chemical burns. It is highly soluble in water, and readily absorbs moisture and carbon dioxide from the air. It forms a series of hydrates . The monohydrate crystallizes from water solutions between 12.3 and 61.8 °C. The commercially available "sodium hydroxide" is often this monohydrate, and published data may refer to it instead of the anhydrous compound. As one of the simplest hydroxides, sodium hydroxide is frequently used alongside neutral water and acidic hydrochloric acid to demonstrate the pH scale to chemistry students. Sodium hydroxide is used in many industries: in the making of wood pulp and paper, tex ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfonic Acid
In organic chemistry, sulfonic acid (or sulphonic acid) refers to a member of the class of organosulfur compounds with the general formula , where R is an organic alkyl or aryl group and the group a sulfonyl hydroxide. As a substituent, it is known as a sulfo group. A sulfonic acid can be thought of as sulfuric acid with one hydroxyl group replaced by an organic substituent. The parent compound (with the organic substituent replaced by hydrogen) is the parent sulfonic acid, , a tautomer of sulfurous acid, . Salt (chemistry), Salts or esters of sulfonic acids are called sulfonates. Preparation Aryl sulfonic acids are produced by the process of sulfonation. Usually the sulfonating agent is sulfur trioxide. A large scale application of this method is the production of alkylbenzenesulfonic acids: : In this reaction, sulfur trioxide is an electrophile and the arene is the nucleophile. The reaction is an example of electrophilic aromatic substitution. In a related process, carboxyli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfur Trioxide
Sulfur trioxide (alternative spelling sulphur trioxide) is the chemical compound with the formula SO3. It has been described as "unquestionably the most conomicallyimportant sulfur oxide". It is prepared on an industrial scale as a precursor to sulfuric acid. Sulfur trioxide exists in several forms: gaseous monomer, crystalline trimer, and solid polymer. Sulfur trioxide is a solid at just below room temperature with a relatively narrow liquid range. Gaseous SO3 is the primary precursor to acid rain. Molecular structure and bonding Monomer The molecule SO3 is trigonal planar. As predicted by VSEPR theory, its structure belongs to the D3h point group. The sulfur atom has an oxidation state of +6 and may be assigned a formal charge value as low as 0 (if all three sulfur-oxygen bonds are assumed to be double bonds) or as high as +2 (if the Octet Rule is assumed). When the formal charge is non-zero, the S-O bonding is assumed to be delocalized. In any case the three S-O bond leng ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dodecylbenzene
Dodecylbenzene is an organic compound with the formula . Dodecylbenzene is a colorless liquid with a weak odor and floats on water. This colorless waxy solid consists of a dodecyl group () attached to a phenyl group (). Dodecylbenzene is a precursor to sodium dodecylbenzenesulfonate, a surfactant that is a key ingredient of household laundry detergents, such as detergent powder.Kurt Kosswig,"Surfactants" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, 2005, Weinheim. Production This compound and some related alkylbenzenes are produced industrially by alkylation of benzene with the corresponding alkenes in the presence of hydrogen fluoride or related acid catalysts. The resulting linear alkylbenzene compounds are sulfonated to give the corresponding sulfonic acid In organic chemistry, sulfonic acid (or sulphonic acid) refers to a member of the class of organosulfur compounds with the general formula , where R is an organic alkyl or aryl group and the group a s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
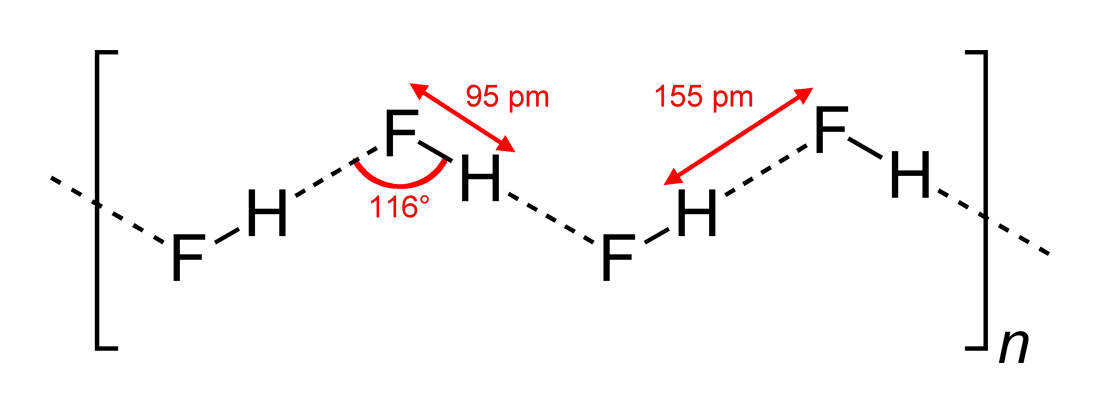
Hydrogen Fluoride
Hydrogen fluoride (fluorane) is an Inorganic chemistry, inorganic compound with chemical formula . It is a very poisonous, colorless gas or liquid that dissolves in water to yield hydrofluoric acid. It is the principal industrial source of fluorine, often in the form of hydrofluoric acid, and is an important feedstock in the preparation of many important compounds including pharmaceuticals and polymers such as polytetrafluoroethylene (PTFE). HF is also widely used in the petrochemical industry as a component of superacids. Due to strong and extensive hydrogen bonding, it boils near room temperature, a much higher temperature than other hydrogen halides. Hydrogen fluoride is an extremely dangerous gas, forming corrosive and penetrating hydrofluoric acid upon contact with moisture. The gas can also cause blindness by rapid destruction of the corneas. History In 1771 Carl Wilhelm Scheele prepared the aqueous solution, hydrofluoric acid in large quantities, although hydrofluoric acid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Linear Alkylbenzene
Linear alkylbenzenes (sometimes also known as LABs) are a family of organic compounds with the formula C6H5CnH2n+1. Typically, ''n'' lies between 10 and 16, although generally supplied as a tighter cut, such as C12-C15, C12-C13 and C10-C13, for detergent use. The CnH2n+1 chain is unbranched. They are mainly produced as intermediate in the production of surfactants, for use in detergent. Since the 1960s, LABs have emerged as the dominant precursor of biodegradable detergents. Production Hydrotreated kerosene is a typical feedstock for high purity linear paraffins (n-paraffins), which are subsequently dehydrogenated to linear olefins: :CnH2n+2 → CnH2n + H2 Alternatively, ethylene can be oligomerized (partially polymerized) to produce linear alkenes. The resulting linear mono-olefins react with benzene in the presence of a catalyst to produce the LABs. Hydrogen fluoride (HF) and aluminium chloride (AlCl3) are the two major catalysts for the alkylation of benzene with linea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |