|
Tetramer
A tetramer () (''tetra-'', "four" + '' -mer'', "parts") is an oligomer formed from four monomers or subunits. The associated property is called ''tetramery''. An example from inorganic chemistry is titanium methoxide with the empirical formula Ti(OCH3)4, which is tetrameric in solid state and has the molecular formula Ti4(OCH3)16. An example from organic chemistry is kobophenol A, a substance that is formed by combining four molecules of resveratrol. In biochemistry, it similarly refers to a biomolecule formed of four units, that are the same ( homotetramer), i.e. as in Concanavalin A or different ( heterotetramer), i.e. as in hemoglobin. Hemoglobin has 4 similar sub-units while immunoglobulins have 2 very different sub-units. The different sub-units may have each their own activity, such as binding biotin in avidin tetramers, or have a common biological property, such as the allosteric binding of oxygen Oxygen is a chemical element; it has chemical symbol, symbo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Homotetramer
A tetrameric protein is a protein with a quaternary structure of four subunits (tetrameric). Homotetramers have four identical subunits (such as glutathione S-transferase), and heterotetramers are complexes of different subunits. A tetramer can be assembled as dimer of dimers with two homodimer subunits (such as sorbitol dehydrogenase), or two heterodimer subunits (such as hemoglobin). Subunit interactions in tetramers The interactions between subunits forming a tetramer is primarily determined by non covalent interaction. Hydrophobic effects, hydrogen bonds and electrostatic interactions are the primary sources for this binding process between subunits. For homotetrameric proteins such as sorbitol dehydrogenase (SDH), the structure is believed to have evolved going from a monomeric to a dimeric and finally a tetrameric structure in evolution. The binding process in SDH and many other tetrameric enzymes can be described by the gain in free energy which can be determined ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heterotetramer
A tetrameric protein is a protein with a quaternary structure of four subunits (tetrameric). Homotetramers have four identical subunits (such as glutathione S-transferase), and heterotetramers are complexes of different subunits. A tetramer can be assembled as dimer of dimers with two homodimer subunits (such as sorbitol dehydrogenase), or two heterodimer subunits (such as hemoglobin). Subunit interactions in tetramers The interactions between subunits forming a tetramer is primarily determined by non covalent interaction. Hydrophobic effects, hydrogen bonds and electrostatic interactions are the primary sources for this binding process between subunits. For homotetrameric proteins such as sorbitol dehydrogenase (SDH), the structure is believed to have evolved going from a monomeric to a dimeric and finally a tetrameric structure in evolution. The binding process in SDH and many other tetrameric enzymes can be described by the gain in free energy which can be det ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
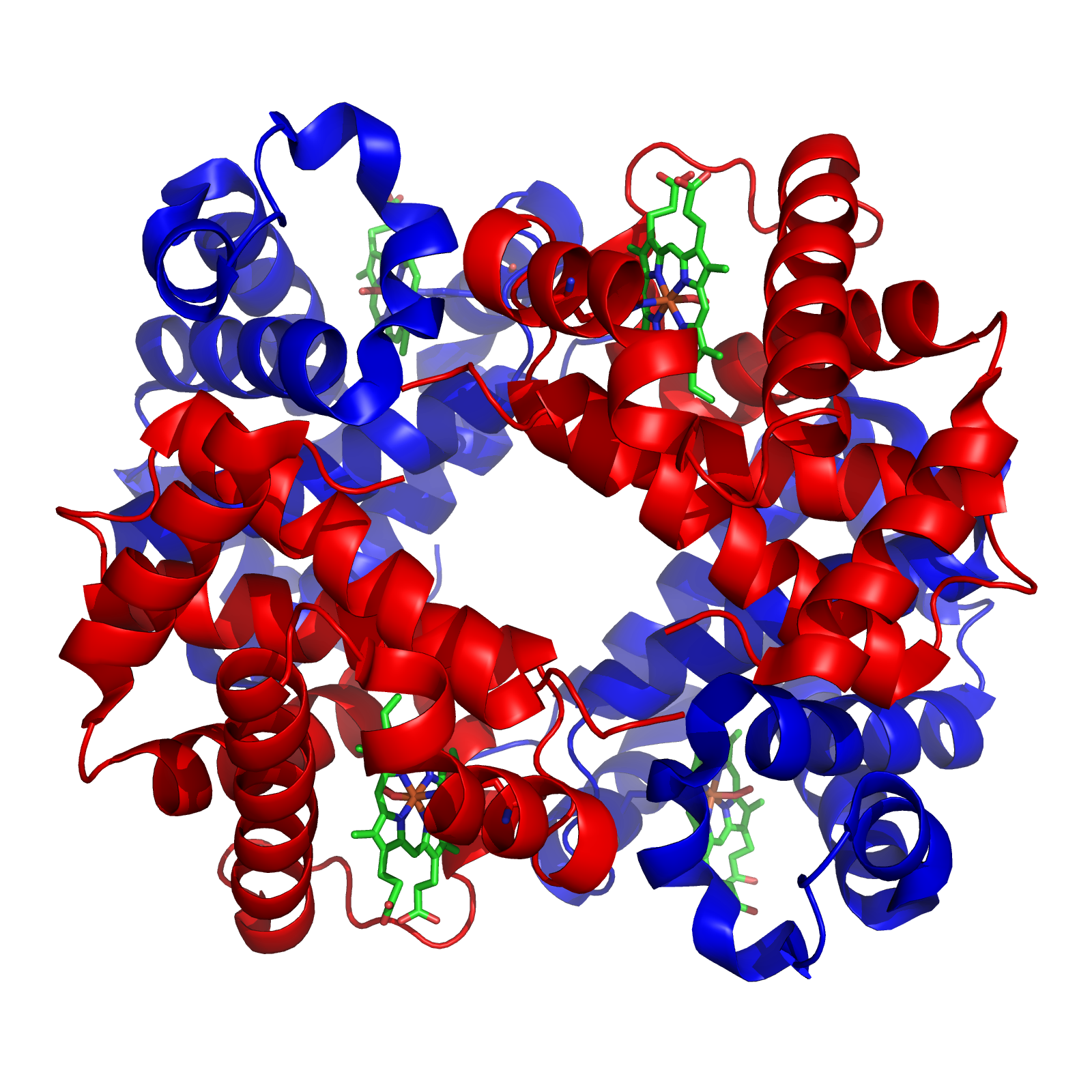
Hemoglobin
Hemoglobin (haemoglobin, Hb or Hgb) is a protein containing iron that facilitates the transportation of oxygen in red blood cells. Almost all vertebrates contain hemoglobin, with the sole exception of the fish family Channichthyidae. Hemoglobin in the blood carries oxygen from the respiratory organs (lungs or gills) to the other tissues of the body, where it releases the oxygen to enable aerobic respiration which powers an animal's metabolism. A healthy human has 12to 20grams of hemoglobin in every 100mL of blood. Hemoglobin is a metalloprotein, a chromoprotein, and a globulin. In mammals, hemoglobin makes up about 96% of a red blood cell's dry matter, dry weight (excluding water), and around 35% of the total weight (including water). Hemoglobin has an oxygen-binding capacity of 1.34mL of O2 per gram, which increases the total blood oxygen capacity seventy-fold compared to dissolved oxygen in blood plasma alone. The mammalian hemoglobin molecule can bind and transport up to four ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Avidin
Avidin is a tetrameric biotin-binding protein produced in the oviducts of birds, reptiles and amphibians and deposited in the whites of their eggs. Dimeric members of the avidin family are also found in some bacteria. In chicken egg white, avidin makes up approximately 0.05% of total protein (approximately 1800 μg per egg). The tetrameric protein contains four identical subunits (homotetramer), each of which can bind to biotin (Vitamin B7, vitamin H) with a high degree of affinity and specificity. The dissociation constant of the avidin-biotin complex is measured to be ''K''D ≈ 10−15 M, making it one of the strongest known non-covalent bonds. In its tetrameric form, avidin is estimated to be 66–69 k Da in size. 10% of the molecular weight is contributed by carbohydrate, composed of four to five mannose and three N-acetylglucosamine residues The carbohydrate moieties of avidin contain at least three unique oligosaccharide structural types that are similar in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Antibody
An antibody (Ab) or immunoglobulin (Ig) is a large, Y-shaped protein belonging to the immunoglobulin superfamily which is used by the immune system to identify and neutralize antigens such as pathogenic bacteria, bacteria and viruses, including those that cause disease. Each individual antibody recognizes one or more specific antigens, and antigens of virtually any size and chemical composition can be recognized. Antigen literally means "antibody generator", as it is the presence of an antigen that drives the formation of an antigen-specific antibody. Each of the branching chains comprising the "Y" of an antibody contains a paratope that specifically binds to one particular epitope on an antigen, allowing the two molecules to bind together with precision. Using this mechanism, antibodies can effectively "tag" the antigen (or a microbe or an infected cell bearing such an antigen) for attack by cells of the immune system, or can neutralize it directly (for example, by blocking a p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Titanium Methoxide
Titanium ethoxide is a chemical compound with the formula Ti4(OCH2CH3)16. It is a commercially available colorless liquid that is soluble in organic solvents but hydrolyzes readily. Its structure is more complex than suggested by its empirical formula. Like other alkoxides of titanium(IV) and zirconium(IV), it finds used in organic synthesis and materials science. Syntheses Titanium ethoxide is prepared by treating titanium tetrachloride with ethanol in the presence of an amine: :TiCl4 + 4 EtOH + 4 Et3N → Ti(OEt)4 + 4 Et3NHCl The purity of titanium ethoxide is commonly assayed by proton NMR spectroscopy. Ti(OEt)4 1H NMR (90 MHz, chloroform-d, ppm): 4.36 (quartet, 8H, CH2), 1.27 (triplet, 12H, CH3). Structure Both Ti(OEt)4 exist mainly as tetramers with an octahedral coordination environment around the metal centers. There are two types of titanium centers, depending on the number of terminal vs bridging alkoxide ligands. Zr(OEt)4 is structurally similar. The virtual symmet ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Merosity
Merosity (from the greek "méros," which means "having parts")) refers to the number of component parts in a distinct whorl of a plant structure. The term is most commonly used in the context of a flower where it refers to the number of sepals in a whorl of the calyx, the number of petals in a whorl of the corolla, the number of stamens in a whorl of the androecium, or the number of carpels in a whorl of the gynoecium. The term may also be used to refer to the number of leaves in a leaf whorl. The adjective ''n''-merous refers to a whorl of ''n'' parts, where ''n'' is any integer greater than one. In nature, five or three parts per whorl have the highest frequency of occurrence, but four or two parts per whorl are not uncommon. Two consecutive whorls of dimerous petals are often mistaken for tetramerous petals. If all of the whorls in a given floral arrangement have the same merosity, the flower is said to be isomerous, otherwise the flower is anisomerous. For example, '' Trilli ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oligomer
In chemistry and biochemistry, an oligomer () is a molecule that consists of a few repeating units which could be derived, actually or conceptually, from smaller molecules, monomers.Quote: ''Oligomer molecule: A molecule of intermediate relative molecular mass, the structure of which essentially comprises a small plurality of units derived, actually or conceptually, from molecules of lower relative molecular mass.'' The name is composed of Greek elements '' oligo-'', "a few" and '' -mer'', "parts". An adjective form is ''oligomeric''. The oligomer concept is contrasted to that of a polymer, which is usually understood to have a large number of units, possibly thousands or millions. However, there is no sharp distinction between these two concepts. One proposed criterion is whether the molecule's properties vary significantly with the removal of one or a few of the units. An oligomer with a specific number of units is referred to by the Greek prefix denoting that number, w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kobophenol A
Kobophenol A is a stilbenoid. It is a tetramer of resveratrol. It can be isolated from '' Caragana chamlagu'',(+)-α-Viniferin, a Stilbene Trimer from Caragana chamlague, Inhibits Acetylcholinesterase. Sang Hyun Sung, So Young Kang, Ki Yong Lee, Mi Jung Park, Jeong Hun Kim, Jong Hee Park, Young Chul Kim, Jinwoong Kim and Young Choong Kim, Biological & Pharmaceutical Bulletin, Vol. 25, 2002 from '''' [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Allosteric Regulation
In the fields of biochemistry and pharmacology an allosteric regulator (or allosteric modulator) is a substance that binds to a site on an enzyme or receptor distinct from the active site, resulting in a conformational change that alters the protein's activity, either enhancing or inhibiting its function. In contrast, substances that bind directly to an enzyme's active site or the binding site of the endogenous ligand of a receptor are called orthosteric regulators or modulators. The site to which the effector binds is termed the ''allosteric site'' or ''regulatory site''. Allosteric sites allow effectors to bind to the protein, often resulting in a conformational change and/or a change in protein dynamics. Effectors that enhance the protein's activity are referred to as ''allosteric activators'', whereas those that decrease the protein's activity are called ''allosteric inhibitors''. Allosteric regulations are a natural example of control loops, such as feedback from do ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
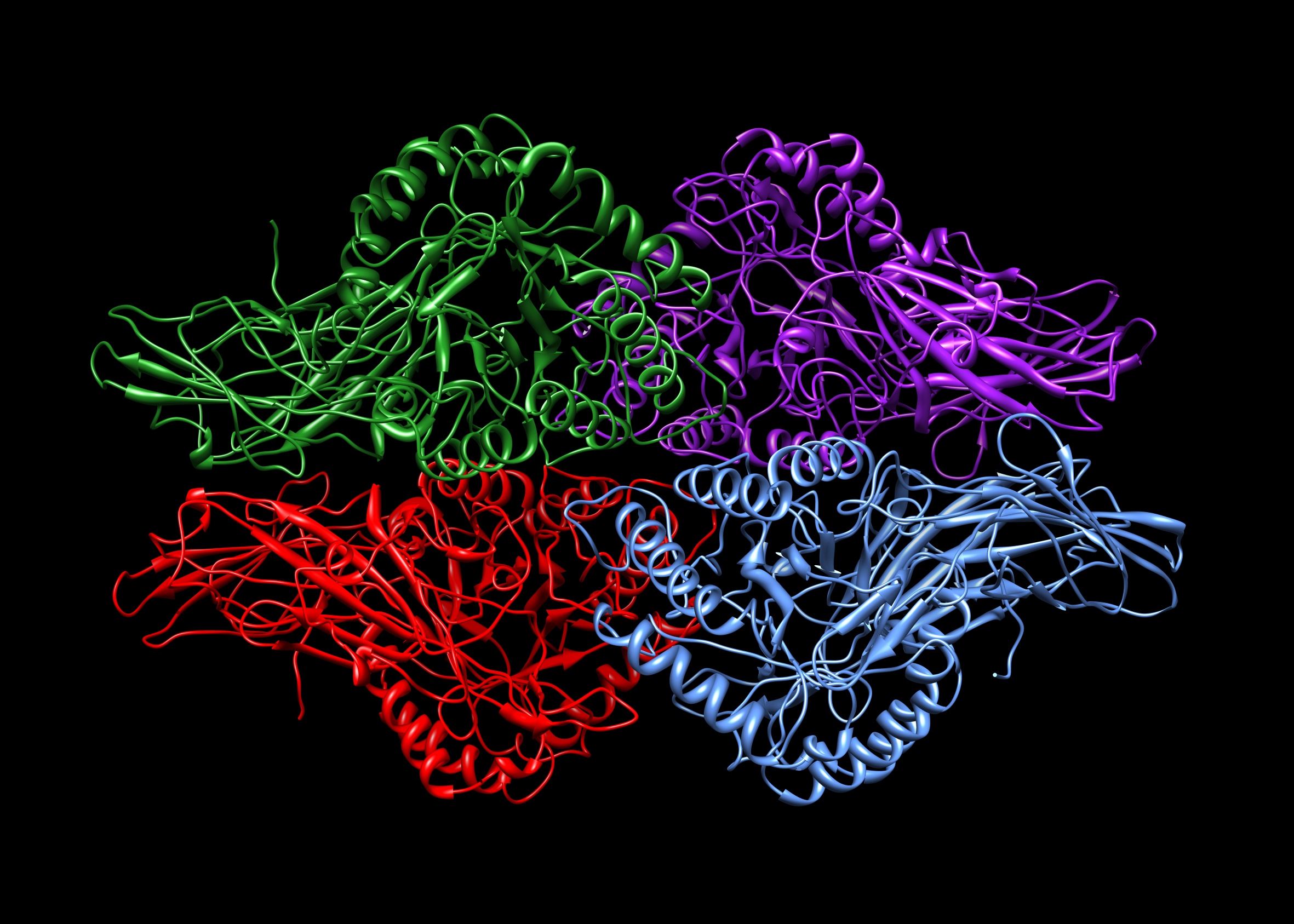
Concanavalin A
Concanavalin A (ConA) is a lectin (carbohydrate-binding protein) originally extracted from the jack-bean (''Canavalia ensiformis''). It is a member of the legume lectin family. It binds specifically to certain structures found in various sugars, glycoproteins, and glycolipids, mainly internal and nonreducing terminal α-D-Mannose, mannosyl and α-D-glucosyl groups. Its physiological function in plants, however, is still unknown. ConA is a plant mitogen, and is known for its ability to stimulate mouse T-cell subsets giving rise to four functionally distinct T cell populations, including precursors to regulatory T cells; a subset of human suppressor T-cells is also sensitive to ConA. ConA was the first lectin to be available on a commercial basis, and is widely used in biology and biochemistry to characterize glycoproteins and other sugar-containing entities on the surface of various cells. It is also used to purify glycosylated macromolecules in lectin affinity chromatography, as w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |