|
Taurides
Taurates (or taurides) are a group of mild anionic surfactants. They are composed of a hydrophilic head group, consisting of N-Methyltaurine, ''N''-methyltaurine (2-methylaminoethanesulfonic acid) and a lipophilic residue, consisting of a long-chain carboxylic acid (fatty acid), both linked via an amide bond. The fatty acids used could be lauric (C12), myristic (C14), palmitic (C16) or stearic acid (C18), but mainly mixtures of oleic acid (C18:1) and coconut fatty acid (C8 – C18) are used. Besides sodium, no other counterions play a relevant role (these could be e. g. ammonium or other alkali or alkaline earth metals). History The surfactant group of the taurates was developed by I.G. Farben in Germany (just like the isethionates) and produced under the trade name Igepon at the Hoechst plant. Taurates rapidly spread due to their lime resistance and their oil-removing effect in textile treatment, as detergent raw material and in cosmetics applications. They had a breakthrough in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
N-Methyltaurine
''N''-Methyltaurine (2-methylaminoethanesulfonic acid) is an aminosulfonic acid which is present as a zwitterion in the crystalline state and in polar solvents (just like amino acids). In contrast to the widespread taurine, ''N''-methyltaurine has been found in nature only in red algae, where it is formed by methylation of taurine. It is suitable for esterification (actually amide formation) with long-chain carboxylic acids to taurides (acylaminoethansulfonaten) because of its high polarity and the relatively good solubility of its alkaline earth metal salts, which are also used as mild anionic surfactants. Preparation The synthesis of ''N''-methyltaurine was reported as early as 1878, with methylamine being reacted with the silver salt of 2-chloroethanesulfonic acid. An obvious modification for this reaction is the replacement of the silver salt of 2-chloroethanesulfonic acid by the sodium salt of 2-chloroethanesulfonic acid. The addition of methylamine to sodium vinylsulfonate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boric Acid
Boric acid, more specifically orthoboric acid, is a compound of boron, oxygen, and hydrogen with formula . It may also be called hydrogen orthoborate, trihydroxidoboron or boracic acid. It is usually encountered as colorless crystals or a white powder, that dissolves in water, and occurs in nature as the mineral sassolite. It is a weak acid that yields various borate anions and salt (chemistry), salts, and can react with Alcohol (chemistry), alcohols to form borate esters. Boric acid is often used as an antiseptic, insecticide, flame retardant, neutron absorber, or precursor to other boron compounds. The term "boric acid" is also used generically for any oxyacid of boron, such as metaboric acid and tetraboric acid . History Orthoboric acid was first prepared by Wilhelm Homberg (1652–1715) from borax, by the action of mineral acids, and was given the name ("sedative salt of Homberg"). However, boric acid and borates have been used since the time of the ancient Greece, anc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crop Protection
Pest control is the regulation or management of a species defined as a pest; such as any animal, plant or fungus that impacts adversely on human activities or environment. The human response depends on the importance of the damage done and will range from tolerance, through deterrence and management, to attempts to completely eradicate the pest. Pest control measures may be performed as part of an integrated pest management strategy. In agriculture, pests are kept at bay by mechanical, cultural, chemical and biological means. Ploughing and cultivation of the soil before sowing mitigate the pest burden, and crop rotation helps to reduce the build-up of a certain pest species. Concern about environment means limiting the use of pesticides in favour of other methods. This can be achieved by monitoring the crop, only applying pesticides when necessary, and by growing varieties and crops which are resistant to pests. Where possible, biological means are used, encouraging the natur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Shampoo
Shampoo () is a hair care product, typically in the form of a viscous liquid, that is formulated to be used for cleaning (scalp) hair. Less commonly, it is available in solid bar format. (" Dry shampoo" is a separate product.) Shampoo is used by applying it to wet hair, massaging the product in the hair, roots and scalp, and then rinsing it out. Some users may follow a shampooing with the use of hair conditioner. Shampoo is typically used to remove the unwanted build-up of sebum (natural oils) in the hair without stripping out so much as to make hair unmanageable. Shampoo is generally made by combining a surfactant, most often sodium lauryl sulfate or sodium laureth sulfate, with a co-surfactant, most often cocamidopropyl betaine in water. The sulfate ingredient acts as a surfactant, trapping oils and other contaminants, similarly to soap. Shampoos are marketed to people with hair. There are also shampoos intended for animals that may contain insecticides or other medica ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
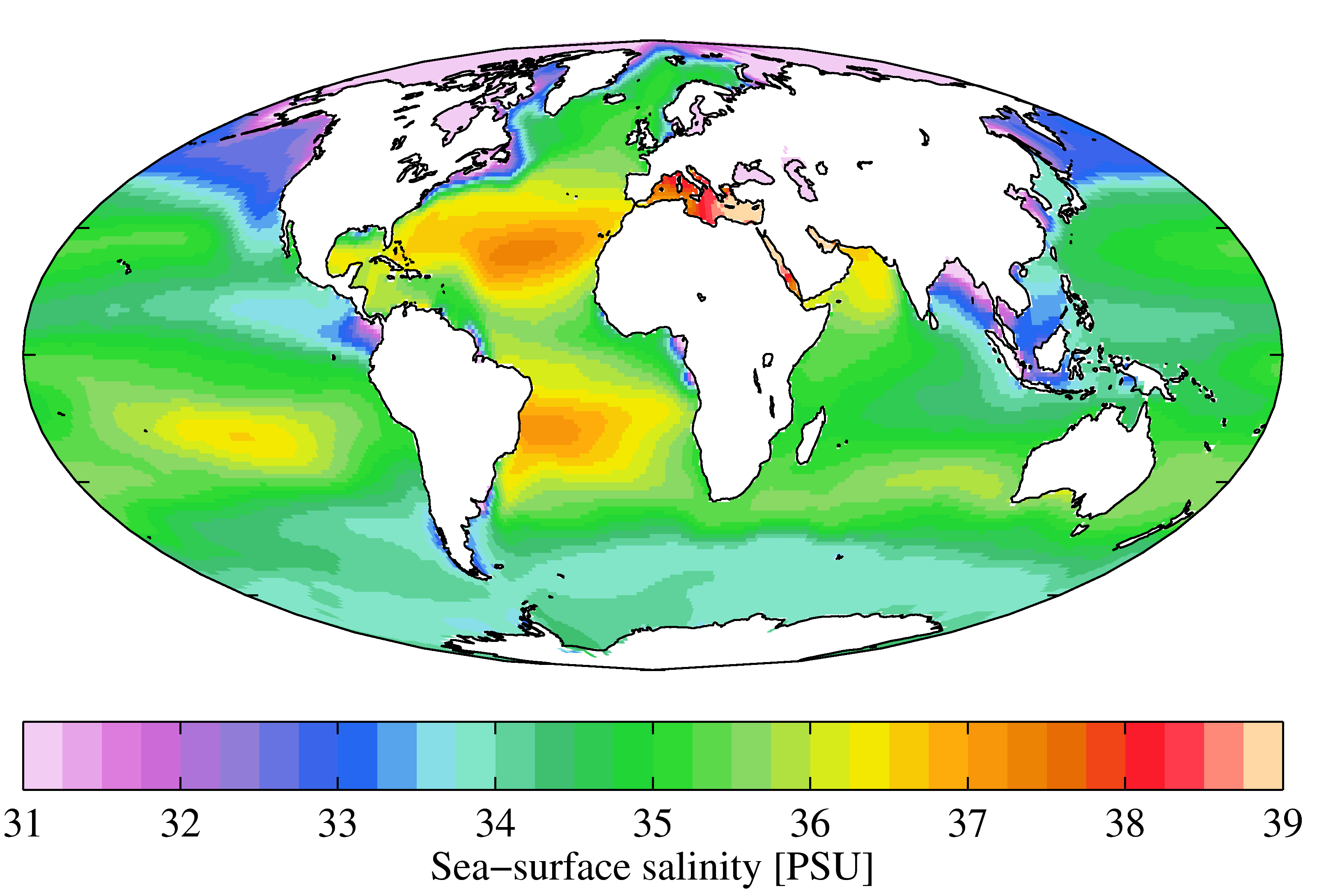
Seawater
Seawater, or sea water, is water from a sea or ocean. On average, seawater in the world's oceans has a salinity of about 3.5% (35 g/L, 35 ppt, 600 mM). This means that every kilogram (roughly one liter by volume) of seawater has approximately of dissolved salts (predominantly sodium () and chloride () ions). The average density at the surface is 1.025 kg/L. Seawater is denser than both fresh water and pure water (density 1.0 kg/L at ) because the dissolved salts increase the mass by a larger proportion than the volume. The freezing point of seawater decreases as salt concentration increases. At typical salinity, it freezes at about . The coldest seawater still in the liquid state ever recorded was found in 2010, in a stream under an Antarctic glacier: the measured temperature was . Seawater pH is typically limited to a range between 7.5 and 8.4. However, there is no universally accepted reference pH-scale for seawater and the difference between measuremen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hard Water
Hard water is water that has a high mineral content (in contrast with "soft water"). Hard water is formed when water percolates through deposits of limestone, chalk or gypsum, which are largely made up of calcium and magnesium carbonates, bicarbonates and sulfates. Drinking hard water may have moderate health benefits. It can pose critical problems in industrial settings, where water hardness is monitored to avoid costly breakdowns in boilers, cooling towers, and other equipment that handles water. In domestic settings, hard water is often indicated by a lack of foam formation when soap is agitated in water, and by the formation of limescale in kettles and water heaters. Wherever water hardness is a concern, water softening is commonly used to reduce hard water's adverse effects. Origins Natural rainwater, snow and other forms of precipitation typically have low concentrations of divalent cations such as calcium and magnesium. They may have small concentrations of ion ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ester
In chemistry, an ester is a compound derived from an acid (either organic or inorganic) in which the hydrogen atom (H) of at least one acidic hydroxyl group () of that acid is replaced by an organyl group (R). These compounds contain a distinctive functional group. Analogues derived from oxygen replaced by other chalcogens belong to the ester category as well. According to some authors, organyl derivatives of acidic hydrogen of other acids are esters as well (e.g. amides), but not according to the IUPAC. Glycerides are fatty acid esters of glycerol; they are important in biology, being one of the main classes of lipids and comprising the bulk of animal fats and vegetable oils. Lactones are cyclic carboxylic esters; naturally occurring lactones are mainly 5- and 6-membered ring lactones. Lactones contribute to the aroma of fruits, butter, cheese, vegetables like celery and other foods. Esters can be formed from oxoacids (e.g. esters of acetic acid, carbonic acid ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amide Bond
In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a compound with the general formula , where R, R', and R″ represent any group, typically organyl groups or hydrogen atoms. The amide group is called a peptide bond when it is part of the main chain of a protein, and an isopeptide bond when it occurs in a side chain, as in asparagine and glutamine. It can be viewed as a derivative of a carboxylic acid () with the hydroxyl group () replaced by an amino group (); or, equivalently, an acyl (alkanoyl) group () joined to an amino group. Common amides are formamide (), acetamide (), benzamide (), and dimethylformamide (). Some uncommon examples of amides are ''N''-chloroacetamide () and chloroformamide (). Amides are qualified as primary, secondary, and tertiary according to the number of acyl groups bounded to the nitrogen atom. Nomenclature The core of amides is called the amide group (specifically, carboxamide group). In the usual n ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Surfactant
Surfactants are chemical compounds that decrease the surface tension or interfacial tension between two liquids, a liquid and a gas, or a liquid and a solid. The word ''surfactant'' is a Blend word, blend of "surface-active agent", coined in 1950. As they consist of a water-repellent and a water-attracting part, they enable water and oil to mix; they can form foam and facilitate the detachment of dirt. Surfactants are among the most widespread and commercially important chemicals. Private households as well as many industries use them in large quantities as detergent, detergents and cleaning agents, but also for example as emulsion#Emulsifiers, emulsifiers, wetting agents, foaming agents, Antistatic agent, antistatic additives, or dispersants. Surfactants occur naturally in traditional plant-based detergents, e.g. Aesculus, horse chestnuts or Sapindus, soap nuts; they can also be found in the secretions of some caterpillars. Today one of the most commonly used anionic surfa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bioaccumulation
Bioaccumulation is the gradual accumulation of substances, such as pesticides or other chemicals, in an organism. Bioaccumulation occurs when an organism absorbs a substance faster than it can be lost or eliminated by catabolism and excretion. Thus, the longer the biological half-life of a toxic substance, the greater the risk of chronic poisoning, even if environmental levels of the toxin are not very high. Bioaccumulation, for example in fish, can be predicted by models. Hypothesis for molecular size cutoff criteria for use as bioaccumulation potential indicators are not supported by data. Biotransformation can strongly modify bioaccumulation of chemicals in an organism. Toxicity induced by metals is associated with bioaccumulation and biomagnification. Storage or uptake of a metal faster than it is metabolized and excreted leads to the accumulation of that metal. The presence of various chemicals and harmful substances in the environment can be analyzed and assessed with a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |