|
Silex Logo 2018
Separation of isotopes by laser excitation (SILEX) is a process for enriching uranium to fuel nuclear reactors that may also present a growing nuclear weapons proliferation risk. It is strongly suspected that SILEX utilizes laser condensation repression to excite a vibrational mode of the uranium-235 isotope in uranium hexaflouride (UF6), allowing this lighter molecule to move more rapidly to the outer rim of a gaseous jet and resist condensing compared to the heavier, unexcited 238UF6. This differs greatly from previous methods of laser enrichment explored for their commercial prospects: one using atomic uranium (Atomic Vapor Laser Isotope Separation (AVLIS)) and another molecular method that uses lasers to dissociate a fluorine atom from 235UF6 (Molecular Laser Isotope Separation (MLIS)), allowing the enriched product to precipitate out as a solid. While the Australian company Silex Systems Limited is the most prominent developer of this technology (as part of the Global Laser En ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molecular Laser Isotope Separation
Molecular laser isotope separation (MLIS) is a method of isotope separation, where specially tunable laser, tuned lasers are used to separate isotopes of uranium using selective ionization of hyperfine transitions of uranium hexafluoride molecules. It is similar to AVLIS. Its main advantage over AVLIS is low energy consumption and use of uranium hexafluoride instead of vaporized uranium. MLIS was conceived in 1971 at the Los Alamos National Laboratory. MLIS operates in cascade (chemical engineering), cascade setup, like the gaseous diffusion process. Instead of vaporized uranium as in AVLIS the working medium of the MLIS is uranium hexafluoride which requires a much lower temperature to vaporize. The UF6 gas is mixed with a suitable carrier gas (a noble gas including some hydrogen) which allows the molecules to remain in the gaseous phase after being cooled by expansion through a supersonic de Laval nozzle. A scavenger gas (e.g. methane) is also included in the mixture to bind with ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Low-enriched Uranium
Enriched uranium is a type of uranium in which the percent composition of uranium-235 (written 235U) has been increased through the process of isotope separation. Naturally occurring uranium is composed of three major isotopes: uranium-238 (238U with 99.2732–99.2752% natural abundance), uranium-235 (235U, 0.7198–0.7210%), and uranium-234 (234U, 0.0049–0.0059%). 235U is the only nuclide existing in nature (in any appreciable amount) that is fissile with thermal neutrons. Enriched uranium is a critical component for both civil nuclear power generation and military nuclear weapons. Low-enriched uranium (20% 235U, typically >85%) is used for the cores of many nuclear weapons, as well as compact reactors for naval propulsion and research, as well as breeder reactors. There are about 2,000 tonnes of highly enriched uranium in the world. Enrichment methods were first developed on a large scale by the Manhattan Project. Its gaseous diffusion method was used in the 1940s a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbon
Carbon () is a chemical element; it has chemical symbol, symbol C and atomic number 6. It is nonmetallic and tetravalence, tetravalent—meaning that its atoms are able to form up to four covalent bonds due to its valence shell exhibiting 4 electrons. It belongs to group 14 of the periodic table. Carbon makes up about 0.025 percent of Earth's crust. Three Isotopes of carbon, isotopes occur naturally, carbon-12, C and carbon-13, C being stable, while carbon-14, C is a radionuclide, decaying with a half-life of 5,700 years. Carbon is one of the timeline of chemical element discoveries#Pre-modern and early modern discoveries, few elements known since antiquity. Carbon is the 15th abundance of elements in Earth's crust, most abundant element in the Earth's crust, and the abundance of the chemical elements, fourth most abundant element in the universe by mass after hydrogen, helium, and oxygen. Carbon's abundance, its unique diversity of organic compounds, and its unusual abi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molybdenum
Molybdenum is a chemical element; it has Symbol (chemistry), symbol Mo (from Neo-Latin ''molybdaenum'') and atomic number 42. The name derived from Ancient Greek ', meaning lead, since its ores were confused with lead ores. Molybdenum minerals have been known throughout history, but the element was discovered (in the sense of differentiating it as a new entity from the mineral salts of other metals) in 1778 by Carl Wilhelm Scheele. The metal was first isolated in 1781 by Peter Jacob Hjelm. Molybdenum does not occur naturally as a Native metal, free metal on Earth; in its minerals, it is found only in oxidation state, oxidized states. The free element, a silvery metal with a grey cast, has the List of elements by melting point, sixth-highest melting point of any element. It readily forms hard, stable carbides in alloys, and for this reason most of the world production of the element (about 80%) is used in steel alloys, including high-strength alloys and superalloys. Most molybdenum ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chlorine
Chlorine is a chemical element; it has Symbol (chemistry), symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. Chlorine is a yellow-green gas at room temperature. It is an extremely reactive element and a strong oxidizing agent, oxidising agent: among the elements, it has the highest electron affinity and the third-highest electronegativity on the revised Electronegativity#Pauling electronegativity, Pauling scale, behind only oxygen and fluorine. Chlorine played an important role in the experiments conducted by medieval Alchemy, alchemists, which commonly involved the heating of chloride Salt (chemistry), salts like ammonium chloride (sal ammoniac) and sodium chloride (common salt), producing various chemical substances containing chlorine such as hydrogen chloride, mercury(II) chloride (corrosive sublimate), and . However, the nature of fre ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrogen
Nitrogen is a chemical element; it has Symbol (chemistry), symbol N and atomic number 7. Nitrogen is a Nonmetal (chemistry), nonmetal and the lightest member of pnictogen, group 15 of the periodic table, often called the Pnictogen, pnictogens. It is a common element in the universe, estimated at Abundance of the chemical elements, seventh in total abundance in the Milky Way and the Solar System. At standard temperature and pressure, two atoms of the element chemical bond, bond to form N2, a colourless and odourless diatomic molecule, diatomic gas. N2 forms about 78% of Atmosphere of Earth, Earth's atmosphere, making it the most abundant chemical species in air. Because of the volatility of nitrogen compounds, nitrogen is relatively rare in the solid parts of the Earth. It was first discovered and isolated by Scottish physician Daniel Rutherford in 1772 and independently by Carl Wilhelm Scheele and Henry Cavendish at about the same time. The name was suggested by French chemist ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Argon
Argon is a chemical element; it has symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third most abundant gas in Earth's atmosphere, at 0.934% (9340 ppmv). It is more than twice as abundant as water vapor (which averages about 4000 ppmv, but varies greatly), 23 times as abundant as carbon dioxide (400 ppmv), and more than 500 times as abundant as neon (18 ppmv). Argon is the most abundant noble gas in Earth's crust, comprising 0.00015% of the crust. Nearly all argon in Earth's atmosphere is radiogenic argon-40, derived from the decay of potassium-40 in Earth's crust. In the universe, argon-36 is by far the most common argon isotope, as it is the most easily produced by stellar nucleosynthesis in supernovas. The name "argon" is derived from the Greek word , neuter singular form of meaning 'lazy' or 'inactive', as a reference to the fact that the element undergoes almost no chemical reactions. The complete oc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
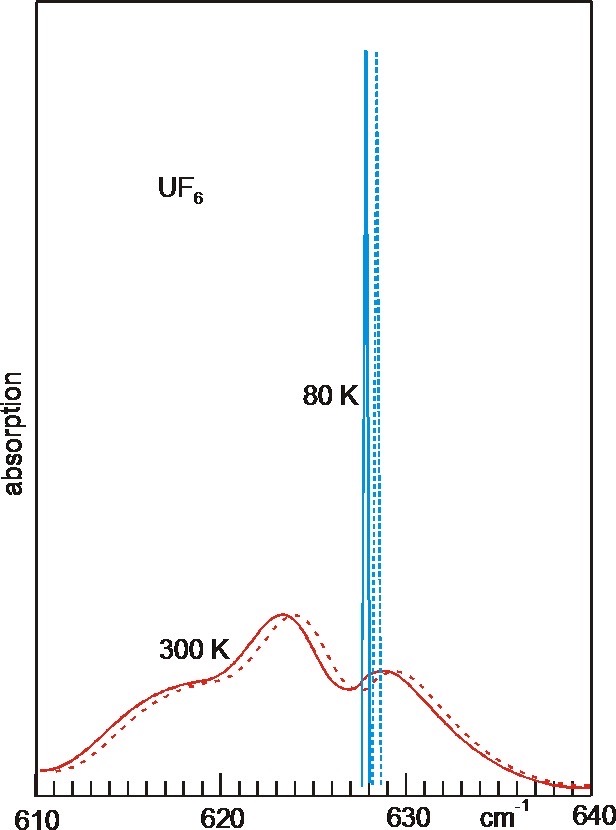
UF6 IR
Uranium hexafluoride, sometimes called hex, is the inorganic compound with the formula . Uranium hexafluoride is a volatile, white solid that is used in enriching uranium for nuclear reactors and nuclear weapons. Preparation Uranium dioxide is converted with hydrofluoric acid (HF) to uranium tetrafluoride: : The resulting is subsequently oxidized with fluorine to give the hexafluoride: : In samples contaminated with uranium trioxide, uranyl fluoride, an oxyfluoride compound is produced in the HF step: : which can be fluorinated to produce the same product, uranium hexafluoride. : The fluorination step in both reactions above are highly exothermic. Properties Physical properties At atmospheric pressure, sublimes at 56.5 °C. The solid-state structure was determined by neutron diffraction at 77 K and 293 K.J. C. Taylor, P. W. Wilson, J. W. Kelly: „The structures of fluorides. I. Deviations from ideal symmetry in the structure of crystalline UF6: a neutron diffraction ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gaseous Diffusion
Gaseous diffusion is a technology that was used to produce enriched uranium by forcing gaseous uranium hexafluoride (UF6) through microporous membranes. This produces a slight separation (enrichment factor 1.0043) between the molecules containing uranium-235 (235U) and uranium-238 (238U). By use of a large cascade of many stages, high separations can be achieved. It was the first process to be developed that was capable of producing enriched uranium in industrially useful quantities, but is nowadays considered obsolete, having been superseded by the more-efficient gas centrifuge process (enrichment factor 1.05 to 1.2). Gaseous diffusion was devised by Francis Simon and Nicholas Kurti at the Clarendon Laboratory in 1940, tasked by the MAUD Committee with finding a method for separating uranium-235 from uranium-238 in order to produce a bomb for the British Tube Alloys project. The prototype gaseous diffusion equipment itself was manufactured by Metropolitan-Vickers (MetroVick ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Paducah Gaseous Diffusion Plant
The Paducah Gaseous Diffusion Plant (PGDP) is a facility located in McCracken County, Kentucky, near Paducah, Kentucky that produced enriched uranium from 1952 to 2013. It is owned by the U.S. Department of Energy (DOE). The PGDP was the only operating uranium enrichment facility in the United States from 2001 to 2010. The Paducah plant produced low-enriched uranium, originally as feedstock for military reactors and weapons, and later for commercial nuclear power fuel. The gaseous diffusion plant covers of a site. The four process buildings cover , and consumed a peak electrical demand of 3,040 megawatts. DOE leased the facility to a publicly held company, USEC, from the mid-1990s. USEC ceased operations in 2013 and returned the facility to the Department of Energy for decontamination and decommissioning. History The former Kentucky Ordnance Works site was chosen from a candidate list of eight sites in 1950. The construction contractor was F.H. McGraw of Hartford, Connecticu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |