|
Semi-crystalline
Crystallinity refers to the degree of structural order in a solid. In a crystal, the atoms or molecules are arranged in a regular, periodic manner. The degree of crystallinity has a large influence on hardness, density, transparency and diffusion. In an ideal gas, the relative positions of the atoms or molecules are completely random. Amorphous materials, such as liquids and glasses, represent an intermediate case, having order over short distances (a few atomic or molecular spacings) but not over longer distances. Many materials, such as glass-ceramics and some polymers, can be prepared in such a way as to produce a mixture of crystalline and amorphous regions. In such cases, crystallinity is usually specified as a percentage of the volume of the material that is crystalline. Even within materials that are completely crystalline, however, the degree of structural perfection can vary. For instance, most metallic alloys are crystalline, but they usually comprise many independent ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polymer
A polymer () is a chemical substance, substance or material that consists of very large molecules, or macromolecules, that are constituted by many repeat unit, repeating subunits derived from one or more species of monomers. Due to their broad spectrum of properties, both synthetic and natural polymers play essential and ubiquitous roles in everyday life. Polymers range from familiar synthetic plastics such as polystyrene to natural biopolymers such as DNA and proteins that are fundamental to biological structure and function. Polymers, both natural and synthetic, are created via polymerization of many small molecules, known as monomers. Their consequently large molecular mass, relative to small molecule compound (chemistry), compounds, produces unique physical property, physical properties including toughness, high rubber elasticity, elasticity, viscoelasticity, and a tendency to form Amorphous solid, amorphous and crystallization of polymers, semicrystalline structures rath ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Amorphous Solid
In condensed matter physics and materials science, an amorphous solid (or non-crystalline solid) is a solid that lacks the long-range order that is a characteristic of a crystal. The terms "glass" and "glassy solid" are sometimes used synonymously with amorphous solid; however, these terms refer specifically to amorphous materials that undergo a glass transition. Examples of amorphous solids include glasses, metallic glasses, and certain types of plastics and polymers. Etymology The term "Amorphous" comes from the Greek language, Greek ''a'' ("without"), and ''morphé'' ("shape, form"). Structure Amorphous materials have an internal structure of molecular-scale structural blocks that can be similar to the basic structural units in the crystalline phase of the same compound. Unlike in crystalline materials, however, no long-range regularity exists: amorphous materials cannot be described by the repetition of a finite unit cell. Statistical measures, such as the atomic density ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solid
Solid is a state of matter where molecules are closely packed and can not slide past each other. Solids resist compression, expansion, or external forces that would alter its shape, with the degree to which they are resisted dependent upon the specific material under consideration. Solids also always possess the least amount of kinetic energy per atom/molecule relative to other phases or, equivalently stated, solids are formed when matter in the liquid / gas phase is cooled below a certain temperature. This temperature is called the melting point of that substance and is an intrinsic property, i.e. independent of how much of the matter there is. All matter in solids can be arranged on a microscopic scale under certain conditions. Solids are characterized by structural rigidity and resistance to applied external forces and pressure. Unlike liquids, solids do not flow to take on the shape of their container, nor do they expand to fill the entire available volume like a gas. Much ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Grain Boundary
In materials science, a grain boundary is the interface between two grains, or crystallites, in a polycrystalline material. Grain boundaries are two-dimensional defects in the crystal structure, and tend to decrease the electrical and thermal conductivity of the material. Most grain boundaries are preferred sites for the onset of corrosion and for the precipitation of new phases from the solid. They are also important to many of the mechanisms of creep. On the other hand, grain boundaries disrupt the motion of dislocations through a material, so reducing crystallite size is a common way to improve mechanical strength, as described by the Hall–Petch relationship. High and low angle boundaries It is convenient to categorize grain boundaries according to the extent of misorientation between the two grains. ''Low-angle grain boundaries'' (''LAGB'') or ''subgrain boundaries'' are those with a misorientation less than about 15 degrees. Generally speaking they are composed of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crystals
A crystal or crystalline solid is a solid material whose constituents (such as atoms, molecules, or ions) are arranged in a highly ordered microscopic structure, forming a crystal lattice that extends in all directions. In addition, macroscopic single crystals are usually identifiable by their geometrical shape, consisting of flat faces with specific, characteristic orientations. The scientific study of crystals and crystal formation is known as crystallography. The process of crystal formation via mechanisms of crystal growth is called crystallization or solidification. The word ''crystal'' derives from the Ancient Greek word (), meaning both "ice" and " rock crystal", from (), "icy cold, frost". Examples of large crystals include snowflakes, diamonds, and table salt. Most inorganic solids are not crystals but polycrystals, i.e. many microscopic crystals fused together into a single solid. Polycrystals include most metals, rocks, ceramics, and ice. A third category of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Obsidian
Obsidian ( ) is a naturally occurring volcanic glass formed when lava extrusive rock, extruded from a volcano cools rapidly with minimal crystal growth. It is an igneous rock. Produced from felsic lava, obsidian is rich in the lighter elements such as silicon, oxygen, aluminium, sodium, and potassium. It is commonly found within the margins of rhyolite, rhyolitic lava flows known as obsidian flows. These flows have a high content of silicon dioxide, silica, giving them a high viscosity. The high viscosity inhibits the atomic diffusion, diffusion of atoms through the lava, which inhibits the first step (nucleation) in the formation of mineral crystals. Together with rapid cooling, this results in a natural glass forming from the lava. Obsidian is hard, Brittleness, brittle, and amorphous; it therefore Fracture (mineralogy)#Conchoidal fracture, fractures with sharp edges. In the past, it was used to manufacture cutting and piercing tools, and it has been used experimentally as s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
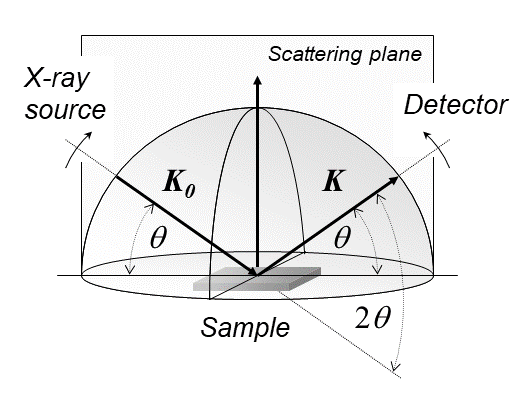
X-ray Diffraction
X-ray diffraction is a generic term for phenomena associated with changes in the direction of X-ray beams due to interactions with the electrons around atoms. It occurs due to elastic scattering, when there is no change in the energy of the waves. The resulting map of the directions of the X-rays far from the sample is called a diffraction pattern. It is different from X-ray crystallography which exploits X-ray diffraction to determine the arrangement of atoms in materials, and also has other components such as ways to map from experimental diffraction measurements to the positions of atoms. This article provides an overview of X-ray diffraction, starting with the early #History, history of x-rays and the discovery that they have the right spacings to be diffracted by crystals. In many cases these diffraction patterns can be #Introduction to x-ray diffraction theory, Interpreted using a single scattering or kinematical theory with conservation of energy (#Ewald's sphere, wave vecto ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Single Crystal
In materials science, a single crystal (or single-crystal solid or monocrystalline solid) is a material in which the crystal lattice of the entire sample is continuous and unbroken to the edges of the sample, with no Grain boundary, grain boundaries. The absence of the crystallographic defect, defects associated with grain boundaries can give monocrystals unique properties, particularly mechanical, optical and electrical, which can also be anisotropic, depending on the type of crystallography, crystallographic structure. These properties, in addition to making some gems precious, are industrially used in technological applications, especially in optics and electronics. Because entropy, entropic effects favor the presence of some imperfections in the microstructure of solids, such as impurity, impurities, inhomogeneous strain and crystallographic defects such as dislocations, perfect single crystals of meaningful size are exceedingly rare in nature. The necessary laboratory condi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boule (crystal)
A boule is a single crystal, single-crystal ingot produced by synthetic means. A boule of silicon is the starting material for most of the integrated circuits used today. In the semiconductor industry synthetic boules can be made by a number of methods, such as the Bridgman technique and the Czochralski process, which result in a cylindrical rod of material. In the Czochralski process a seed crystal is required to create a larger crystal, or ingot. This seed crystal is dipped into the pure molten silicon and slowly extracted. The molten silicon grows on the seed crystal in a crystalline fashion. As the seed is extracted the silicon solidifies and eventually a large, cylindrical boule is produced. A semiconductor crystal boule is normally cut into circular wafer (electronics), wafers using an inside hole diamond saw or diamond wire saw, and each wafer is lapped and polished to provide substrates suitable for the fabrication of semiconductor devices on its surface. The process ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dislocation
In materials science, a dislocation or Taylor's dislocation is a linear crystallographic defect or irregularity within a crystal structure that contains an abrupt change in the arrangement of atoms. The movement of dislocations allow atoms to slide over each other at low stress levels and is known as ''glide'' or Slip (materials science), slip. The crystalline order is restored on either side of a ''glide dislocation'' but the atoms on one side have moved by one position. The crystalline order is not fully restored with a ''partial dislocation''. A dislocation defines the boundary between ''slipped'' and ''unslipped'' regions of material and as a result, must either form a complete loop, intersect other dislocations or defects, or extend to the edges of the crystal. A dislocation can be characterised by the distance and direction of movement it causes to atoms which is defined by the Burgers vector. Plasticity (physics), Plastic deformation of a material occurs by the creation and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crystallographic Defect
A crystallographic defect is an interruption of the regular patterns of arrangement of atoms or molecules in Crystal, crystalline solids. The positions and orientations of particles, which are repeating at fixed distances determined by the Crystal structure#unit cell, unit cell parameters in crystals, exhibit a periodic crystal structure, but this is usually imperfect.Ehrhart, P. (1991Properties and interactions of atomic defects in metals and alloys, volume 25 of Landolt-Börnstein, New Series III, chapter 2, p. 88, Springer, Berlin Several types of defects are often characterized: point defects, line defects, planar defects, bulk defects. Topological homotopy establishes a mathematical method of characterization. Point defects Point defects are defects that occur only at or around a single lattice point. They are not extended in space in any dimension. Strict limits for how small a point defect is are generally not defined explicitly. However, these defects typically involve at ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |