|
Saquinavir Structure
Saquinavir (SQV), sold under the brand names Invirase and Fortovase, is an antiretroviral drug used together with other medications to treat or prevent HIV/AIDS. Typically it is used with ritonavir or lopinavir/ritonavir to increase its effect. It is taken by mouth. Common side effects include nausea, vomiting, diarrhea, and feeling tired. More serious side effects include problems with QT prolongation, heart block, high blood lipids, and liver problems. It appears to be safe in pregnancy. It is in the protease inhibitor class and works by blocking the HIV protease. Saquinavir was patented in 1988 and first sold in 1995. Medical uses Saquinavir is used together with other medications to treat or prevent HIV/AIDS. Typically it is used with ritonavir or lopinavir/ritonavir to increase its effect. Side effects The most frequent adverse events with saquinavir in either formulation are mild gastrointestinal symptoms, including diarrhoea, nausea, loose stools and abdominal dis ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ritonavir
Ritonavir, sold under the brand name Norvir, is an antiretroviral drug used along with other medications to treat HIV/AIDS. This combination treatment is known as highly active antiretroviral therapy (HAART). Ritonavir is a protease inhibitor and is used with other protease inhibitors. It may also be used in combination with other medications to treat hepatitis C and COVID-19. It is taken by mouth. Tablets of ritonavir are not bioequivalent to capsules, as the tablets may result in higher peak plasma concentrations. Common side effects include nausea, vomiting, loss of appetite, diarrhea, and numbness of the hands and feet. Serious side effects include liver problems, pancreatitis, allergic reactions, and arrythmias. Serious interactions may occur with a number of other medications including amiodarone and simvastatin. At low doses it is considered to be acceptable for use during pregnancy. Ritonavir is of the protease inhibitor class. Typically, however, it is used to inhibit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
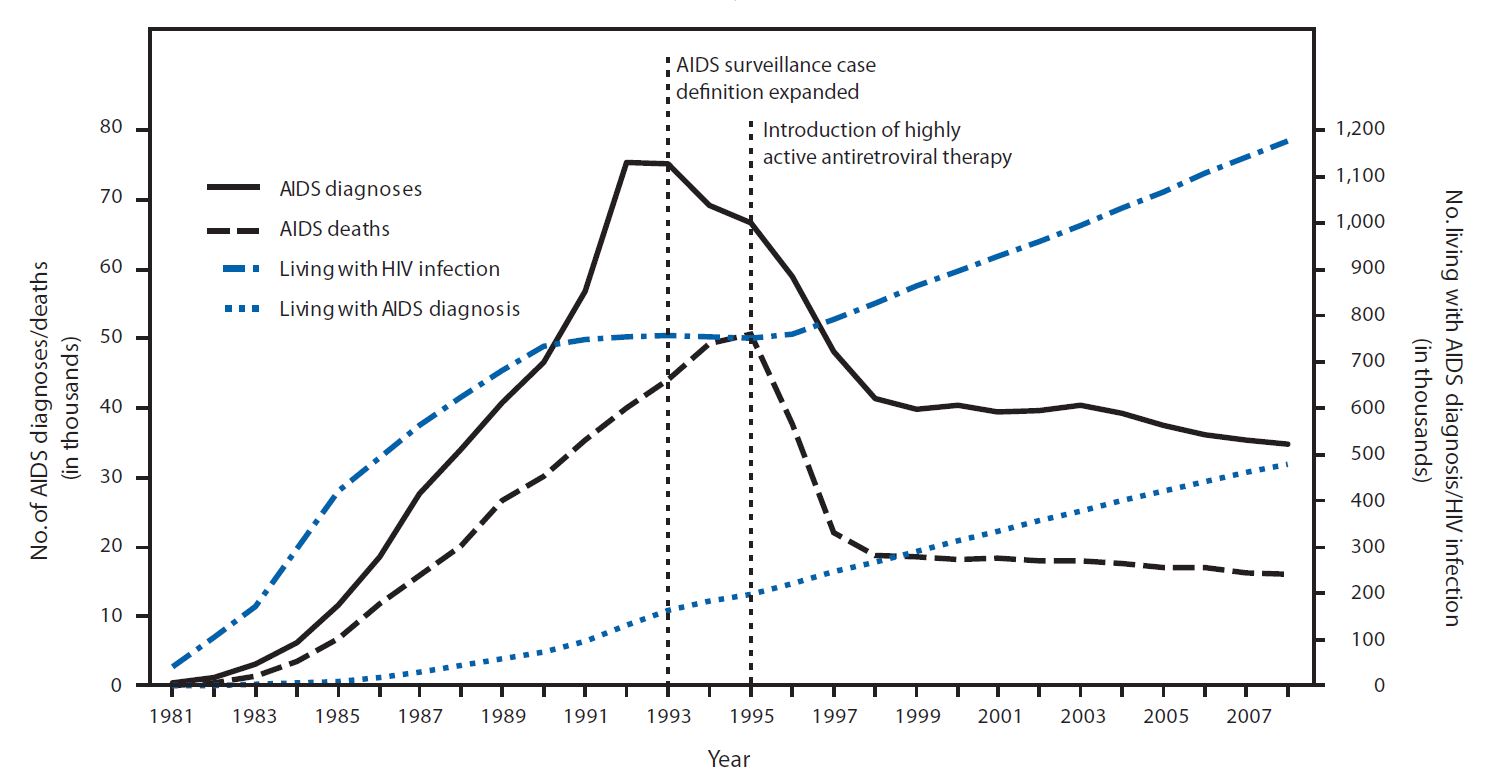
Subtypes Of HIV
The subtypes of HIV include two major types, HIV type 1 (HIV-1) and HIV type 2 (HIV-2). HIV-1 is related to viruses found in chimpanzees and gorillas living in western Africa, while HIV-2 viruses are related to viruses found in the sooty mangabey, a vulnerable West African primate. HIV-1 viruses can be further divided into groups M, N, O and P. The HIV-1 group M viruses predominate and are responsible for the AIDS pandemic. Group M can be further subdivided into subtypes based on genetic sequence data. Some of the subtypes are known to be more virulent or are resistant to different medications. Likewise, HIV-2 viruses are thought to be less virulent and transmissible than HIV-1 M group viruses, although HIV-2 is also known to cause AIDS. One of the obstacles to treatment of the human immunodeficiency virus (HIV) is its high genetic variability. Major types HIV-1 HIV-1 is the most common and pathogenic strain of the virus. Over 2 million such infections occur annually. Scientists ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hepatotoxins
Hepatotoxicity (from ''hepatic toxicity'') implies chemical-driven liver damage. Drug-induced liver injury is a cause of acute and chronic liver disease caused specifically by medications and the most common reason for a drug to be withdrawn from the market after approval. The liver plays a central role in transforming and clearing chemicals and is susceptible to the toxicity from these agents. Certain medicinal agents, when taken in overdoses (e.g. paracetamol) and sometimes even when introduced within therapeutic ranges (e.g. halothane), may injure the organ. Other chemical agents, such as those used in laboratories and industries, natural chemicals (e.g., microcystins), and herbal remedies (two prominent examples being kava, mechanism unknown, and comfrey, through its pyrrolizidine alkaloid content) can also induce hepatotoxicity. Chemicals that cause liver injury are called hepatotoxins. More than 900 drugs have been implicated in causing liver injury (see LiverTox, ext ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Genentech Brands
Genentech, Inc., is an American biotechnology corporation headquartered in South San Francisco, California. It became an independent subsidiary of Roche in 2009. Genentech Research and Early Development operates as an independent center within Roche. Historically, the company is regarded as the world's first biotechnology company. As of July 2021, Genentech employed 13,539 people. History The company was founded in 1976 by venture capitalist Robert A. Swanson and biochemist Herbert Boyer. Boyer is considered to be a pioneer in the field of recombinant DNA technology. In 1973, Boyer and his colleague Stanley Norman Cohen demonstrated that restriction enzymes could be used as "scissors" to cut DNA fragments of interest from one source, to be ligated into a similarly cut plasmid vector. While Cohen returned to the laboratory in academia, Swanson contacted Boyer to found the company. Boyer worked with Arthur Riggs and Keiichi Itakura from the Beckman Research Instit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
CYP3A4 Inhibitors
Cytochrome P450 3A4 (abbreviated CYP3A4) () is an important enzyme in the body, mainly found in the liver and in the intestine. It oxidizes small foreign organic molecules ( xenobiotics), such as toxins or drugs, so that they can be removed from the body. It is highly homologous to CYP3A5, another important CYP3A enzyme. While many drugs are deactivated by CYP3A4, there are also some drugs which are ''activated'' by the enzyme. Some substances, such as some drugs and furanocoumarins present in grapefruit juice, interfere with the action of CYP3A4. These substances will therefore either amplify or weaken the action of those drugs that are modified by CYP3A4. CYP3A4 is a member of the cytochrome P450 family of oxidizing enzymes. Several other members of this family are also involved in drug metabolism, but CYP3A4 is the most common and the most versatile one. Like all members of this family, it is a hemoprotein, i.e. a protein containing a heme group with an iron atom. In humans ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Microemulsion
Microemulsions are clear, thermodynamically stable isotropic liquid mixtures of oil, water and surfactant, frequently in combination with a cosurfactant. The aqueous phase may contain salt(s) and/or other ingredients, and the "oil" may actually be a complex mixture of different hydrocarbons. In contrast to ordinary emulsions, microemulsions form upon simple mixing of the components and do not require the high shear conditions generally used in the formation of ordinary emulsions. The three basic types of microemulsions are direct (oil dispersed in water, o/w), reversed (water dispersed in oil, w/o) and bicontinuous. In ternary systems such as microemulsions, where two immiscible phases (water and ‘oil’) are present with a surfactant, the surfactant molecules may form a monolayer at the interface between the oil and water, with the hydrophobic tails of the surfactant molecules dissolved in the oil phase and the hydrophilic head groups in the aqueous phase. Uses Microemulsion ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mesylate
In organosulfur chemistry, a mesylate is any salt or ester of methanesulfonic acid (). In salts, the mesylate is present as the anion. When modifying the international nonproprietary name of a pharmaceutical substance containing the group or anion, the spelling used is sometimes mesilate (as in ''imatinib mesilate'', the mesylate salt of imatinib). Mesylate esters are a group of organic compounds that share a common functional group with the general structure , abbreviated , where R is an organic substituent. Mesylate is considered a leaving group in nucleophilic substitution reactions. Preparation Mesylates are generally prepared by treating an alcohol and methanesulfonyl chloride in the presence of a base, such as triethylamine. Mesyl Related to mesylate is the mesyl (Ms) or methanesulfonyl (CH3SO2) functional group. Methanesulfonyl chloride is often referred to as mesyl chloride. Whereas mesylates are often hydrolytically labile, mesyl groups, when attached to nit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Generic Medication
A generic drug is a pharmaceutical drug that contains the same chemical substance as a drug that was originally protected by chemical patents. Generic drugs are allowed for sale after the patents on the original drugs expire. Because the active chemical substance is the same, the medical profile of generics is equivalent in performance. A generic drug has the same active pharmaceutical ingredient (API) as the original, but it may differ in some characteristics such as the manufacturing process, formulation, excipients, color, taste, and packaging. Although they may not be associated with a particular company, generic drugs are usually subject to government regulations in the countries in which they are dispensed. They are labeled with the name of the manufacturer and a generic non-proprietary name such as the United States Adopted Name (USAN) or International Nonproprietary Name (INN) of the drug. A generic drug must contain the same active ingredients as the original brand-n ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bioavailability
In pharmacology, bioavailability is a subcategory of absorption and is the fraction (%) of an administered drug that reaches the systemic circulation. By definition, when a medication is administered intravenously, its bioavailability is 100%. However, when a medication is administered via routes other than intravenous, its bioavailability is generally lower than that of intravenous due to intestinal endothelium absorption and first-pass metabolism. Thereby, mathematically, bioavailability equals the ratio of comparing the area under the plasma drug concentration curve versus time (AUC) for the extravascular formulation to the AUC for the intravascular formulation. AUC is used because AUC is proportional to the dose that has entered the systemic circulation. Bioavailability of a drug is an average value; to take population variability into account, deviation range is shown as ±. To ensure that the drug taker who has poor absorption is dosed appropriately, the bottom value ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Indinavir
Indinavir (IDV; trade name Crixivan, made by Merck) is a protease inhibitor used as a component of highly active antiretroviral therapy to treat HIV/AIDS. It is soluble white powder administered orally in combination with other antiviral drugs. The drug prevents protease from functioning normally. Consequently, HIV viruses cannot reproduce, causing a decrease in the viral load. Commercially sold indinavir is indinavir anhydrous, which is indinavir with an additional amine in the hydroxyethylene backbone. This enhances its solubility and oral bioavailability, making it easier for users to intake. It was synthetically produced for the purpose of inhibiting the protease in the HIV virus. Currently, it is not recommended for use in HIV/AIDS treatment due to its side effects. Furthermore, it is controversial for many reasons starting from its development to its usage. It was patented in 1991 and approved for medical use in 1996. Medical uses Indinavir does not cure HIV/AIDS, but ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |