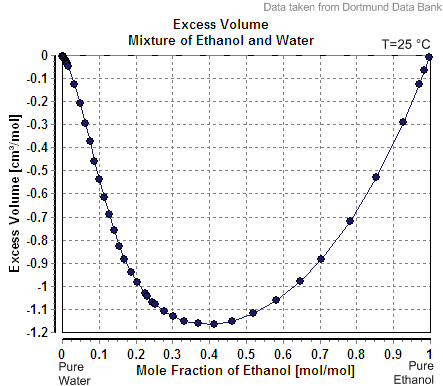
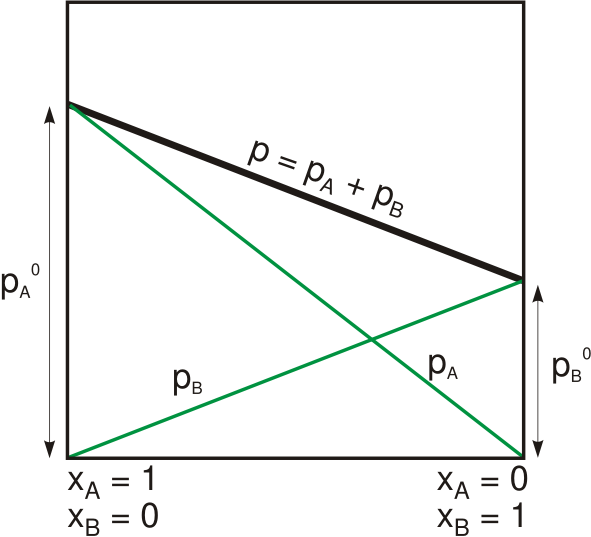
|
Regular Solution
In chemistry, a regular solution is a solution whose entropy of mixing is equal to that of an ideal solution with the same composition, but is non-ideal due to a nonzero enthalpy of mixing.P. Atkins and J. de Paula, ''Atkins' Physical Chemistry'' (8th ed. W.H. Freeman 2006) p.149P.A. Rock, ''Chemical Thermodynamics. Principles and Applications'' (Macmillan 1969) p.263 Such a solution is formed by random mixing of components of similar molar volume and without strong specific interactions, and its behavior diverges from that of an ideal solution by showing phase separation at intermediate compositions and temperatures (a miscibility gap). Its entropy of mixing is equal to that of an ideal solution with the same composition, due to random mixing without strong specific interactions. For two components :\Delta S_ = -nR(x_1\ln x_1 + x_2\ln x_2)\, where R\, is the gas constant, n\, the total number of mole (unit), moles, and x_i\, the mole fraction of each component. Only the enthalpy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemistry
Chemistry is the scientific study of the properties and behavior of matter. It is a physical science within the natural sciences that studies the chemical elements that make up matter and chemical compound, compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during chemical reaction, reactions with other chemical substance, substances. Chemistry also addresses the nature of chemical bonds in chemical compounds. In the scope of its subject, chemistry occupies an intermediate position between physics and biology. It is sometimes called the central science because it provides a foundation for understanding both Basic research, basic and Applied science, applied scientific disciplines at a fundamental level. For example, chemistry explains aspects of plant growth (botany), the formation of igneous rocks (geology), how atmospheric ozone is formed and how environmental pollutants are degraded (ecology), the prop ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Margules Function
The Margules activity model is a simple thermodynamic model for the excess Gibbs free energy of a liquid mixture introduced in 1895 by Max Margules. After Lewis had introduced the concept of the activity coefficient, the model could be used to derive an expression for the activity coefficients \gamma_i of a compound i in a liquid, a measure for the deviation from ideal solubility, also known as Raoult's law Raoult's law ( law) is a relation of physical chemistry, with implications in thermodynamics. Proposed by French chemist François-Marie Raoult in 1887, it states that the partial pressure of each component of an ideal mixture of ''liquids'' is .... In 1900, Jan Zawidzki proved the model via determining the composition of binary mixtures condensed at different temperatures by their refractive indices. In chemical engineering the Margules Gibbs free energy model for liquid mixtures is better known as the Margules activity or activity coefficient model. Although the model ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solid Solution
A solid solution, a term popularly used for metals, is a homogeneous mixture of two compounds in solid state and having a single crystal structure. Many examples can be found in metallurgy, geology, and solid-state chemistry. The word "solution" is used to describe the intimate mixing of components at the atomic level and distinguishes these homogeneous materials from physical mixtures of components. Two terms are mainly associated with solid solutions – ''solvents'' and ''solutes,'' depending on the relative abundance of the atomic species. In general if two compounds are isostructural then a solid solution will exist between the end members (also known as parents). For example sodium chloride and potassium chloride have the same cubic crystal structure so it is possible to make a pure compound with any ratio of sodium to potassium (Na1-xKx)Cl by dissolving that ratio of NaCl and KCl in water and then evaporating the solution. A member of this family is sold under the bra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Joel Henry Hildebrand
Joel Henry Hildebrand (November 16, 1881 – April 30, 1983) was an American educator and a pioneer chemist. He was a major figure in physical chemistry research specializing in liquids and nonelectrolyte solutions. Education and professorship He was born in Camden, New Jersey on November 16, 1881. Hildebrand graduated from the University of Pennsylvania in 1903. He served briefly in the faculty before going to the University of California, Berkeley as a chemistry instructor in 1913. Within five years he became an assistant professor. In 1918 he was elevated to associate professor before finally being granted full professorship in 1919. On August 4, 1919, he was shot and wounded by a chemistry assistant angry at not being recommended for further advancement. He was the dean of the College of Chemistry from 1949 through 1951 and retired from full-time teaching in 1952. Hildebrand Hall on the Berkeley campus is named for him. Accomplishments, discoveries, honors His 1924 mo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Partial Molar Volume
In thermodynamics, a partial molar property is a quantity which describes the variation of an extensive property of a solution or mixture with changes in the molar composition of the mixture at constant temperature and pressure. It is the partial derivative of the extensive property with respect to the amount (number of moles) of the component of interest. Every extensive property of a mixture has a corresponding partial molar property. Definition The partial molar volume is broadly understood as the contribution that a component of a mixture makes to the overall volume of the solution. However, there is more to it than this: When one mole of water is added to a large volume of water at 25 °C, the volume increases by 18 cm3. The molar volume of pure water would thus be reported as 18 cm3 mol−1. However, addition of one mole of water to a large volume of pure ethanol results in an increase in volume of only 14 cm3. The reason that the increase is diff ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Upper Critical Solution Temperature
The upper critical solution temperature (UCST) or upper consolute temperature is the critical temperature above which the components of a mixture are miscible in all proportions. The word ''upper'' indicates that the UCST is an upper bound to a temperature range of partial miscibility, or miscibility for certain compositions only. For example, hexane-nitrobenzene mixtures have a UCST of , so that these two substances are miscible in all proportions above but not at lower temperatures. Examples at higher temperatures are the aniline-water system at (at pressures high enough for liquid water to exist at that temperature), and the lead-zinc system at (a temperature where both metals are liquid). A solid state example is the palladium-hydrogen system which has a solid solution phase (H2 in Pd) in equilibrium with a hydride phase (PdHn) below the UCST at 300 °C. Above this temperature there is a single solid solution phase. In the phase diagram of the mixture components, the U ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Gibbs Free Energy
In thermodynamics, the Gibbs free energy (or Gibbs energy as the recommended name; symbol is a thermodynamic potential that can be used to calculate the maximum amount of Work (thermodynamics), work, other than Work (thermodynamics)#Pressure–volume work, pressure–volume work, that may be performed by a closed system, thermodynamically closed system at constant temperature and pressure. It also provides a necessary condition for processes such as chemical reactions that may occur under these conditions. The Gibbs free energy is expressed as G(p,T) = U + pV - TS = H - TS where: * U is the internal energy of the system * H is the enthalpy of the system * S is the entropy of the system * T is the temperature of the system * V is the volume of the system * p is the pressure of the system (which must be equal to that of the surroundings for mechanical equilibrium). The Gibbs free energy change (, measured in joules in International System of Units, SI) is the ''maximum'' amount of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Raoult's Law
Raoult's law ( law) is a relation of physical chemistry, with implications in thermodynamics. Proposed by French chemist François-Marie Raoult in 1887, it states that the partial pressure of each component of an ideal mixture of ''liquids'' is equal to the vapor pressure of the pure component (liquid or solid) multiplied by its mole fraction in the mixture. In consequence, the relative lowering of vapor pressure of a dilute solution of nonvolatile solute is equal to the mole fraction of solute in the solution. Mathematically, Raoult's law for a single component in an ideal solution is stated as : p_i = p_i^\star x_i where p_i is the partial pressure of the component i in the gaseous mixture above the solution, p_i^\star is the equilibrium vapor pressure of the pure component i, and x_i is the mole fraction of the component i in the liquid or solid solution. Where two volatile liquids A and B are mixed with each other to form a solution, the vapor phase consists of both compo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Entropy Of Mixing
In thermodynamics, the entropy of mixing is the increase in the total entropy when several initially separate systems of different composition, each in a thermodynamic state of internal equilibrium, are mixed without chemical reaction by the thermodynamic operation of removal of impermeable partition(s) between them, followed by a time for establishment of a new thermodynamic state of internal equilibrium in the new unpartitioned closed system. In general, the mixing may be constrained to occur under various prescribed conditions. In the customarily prescribed conditions, the materials are each initially at a common temperature and pressure, and the new system may change its volume, while being maintained at that same constant temperature, pressure, and chemical component masses. The volume available for each material to explore is increased, from that of its initially separate compartment, to the total common final volume. The final volume need not be the sum of the initially se ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mole Fraction
In chemistry, the mole fraction or molar fraction, also called mole proportion or molar proportion, is a quantity defined as the ratio between the amount of a constituent substance, ''ni'' (expressed in unit of moles, symbol mol), and the total amount of all constituents in a mixture, ''n''tot (also expressed in moles): :x_i = \frac It is denoted ''xi'' (lowercase Roman letter '' x''), sometimes (lowercase Greek letter chi). (For mixtures of gases, the letter ''y'' is recommended.) It is a dimensionless quantity with dimension of \mathsf/\mathsf and dimensionless unit of moles per mole (mol/mol or molmol−1) or simply 1; metric prefixes may also be used (e.g., nmol/mol for 10−9). When expressed in percent, it is known as the mole percent or molar percentage (unit symbol %, sometimes "mol%", equivalent to cmol/mol for 10−2). The mole fraction is called amount fraction by the International Union of Pure and Applied Chemistry (IUPAC) and amount-of-substance fractio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mole (unit)
The mole (symbol mol) is a unit of measurement, the base unit in the International System of Units (SI) for ''amount of substance'', an SI base quantity proportional to the number of elementary entities of a substance. One mole is an aggregate of exactly elementary entities (approximately 602 sextillion or 602 billion times a trillion), which can be atoms, molecules, ions, ion pairs, or other particles. The number of particles in a mole is the Avogadro number (symbol ) and the numerical value of the '' Avogadro constant'' (symbol ) expressed in mol−1. The relationship between the mole, Avogadro number, and Avogadro constant can be expressed in the following equation:1\text = \frac = \frac The current SI value of the mole is based on the historical definition of the mole as the amount of substance that corresponds to the number of atoms in 12 grams of 12C, which made the molar mass of a compound in grams per mole, numerically equal to the average molecular mass or ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |