|
Photocatalysis
In chemistry, photocatalysis is the acceleration of a photoreaction in the presence of a photocatalyst, the excited state of which "repeatedly interacts with the reaction partners forming reaction intermediates and regenerates itself after each cycle of such interactions." In many cases, the catalyst is a solid that upon irradiation with UV- or visible light generates electron–hole pairs that generate free radicals. Photocatalysts belong to three main groups; heterogeneous, homogeneous, and plasmonic antenna-reactor catalysts. The use of each catalysts depends on the preferred application and required catalysis reaction. History Early mentions (1911–1938) The earliest mention came in 1911, when German chemist Dr. Alexander Eibner integrated the concept in his research of the illumination of zinc oxide (ZnO) on the bleaching of the dark blue pigment, Prussian blue. Around this time, Bruner and Kozak published an article discussing the deterioration of oxalic acid in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Oxide
Zinc oxide is an inorganic compound with the Chemical formula, formula . It is a white powder which is insoluble in water. ZnO is used as an additive in numerous materials and products including cosmetics, Zinc metabolism, food supplements, rubbers, plastics, ceramics, glass, cement, lubricants, paints, sunscreens, ointments, adhesives, sealants, pigments, foods, batteries, ferrites, fire retardants, semi conductors, and first-aid tapes. Although it occurs naturally as the mineral zincite, most zinc oxide is produced synthetically. History Early humans probably used zinc compounds in processed and unprocessed forms, as paint or medicinal ointment; however, their composition is uncertain. The use of ''pushpanjan'', probably zinc oxide, as a salve for eyes and open wounds is mentioned in the Indian medical text the Charaka Samhita, thought to date from 500 BC or before. Zinc oxide ointment is also mentioned by the Greek physician Dioscorides (1st century AD). Galen suggested treatin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alexander Eibner
Alexander Paul Friedrich Eibner (1862–1935), was a German chemist, painter, and educator. He was known for his technical painting techniques. Early life and education Alexander Eibner, was born on September 11, 1862, in Munich. He was the son of Luise Weissenberger, and painter Friedrich Eibner. Eibner studied chemistry at the Technical University of Munich, under Wilhelm von Miller and he obtained a doctorate degree in 1892. Career By 1894, he qualified as a professor, and began researching aromaticity in organic chemistry. He studied the bleaching of pigment of Prussian Blue using zinc oxide. Through that research he introduced the concept of photocatalysis in 1911. In 1903, Eibner became assistant to chemist at the newly founded ''Research Institute and Information Center for Painting Techniques,'' promoting the scientific study of the nature and the properties of painting materials. Starting in 1907, Eibner lead the ''Research Institute and Information Center for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Catalysis
Catalysis () is the increase in rate of a chemical reaction due to an added substance known as a catalyst (). Catalysts are not consumed by the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recycles quickly, very small amounts of catalyst often suffice; mixing, surface area, and temperature are important factors in reaction rate. Catalysts generally react with one or more reactants to form intermediates that subsequently give the final reaction product, in the process of regenerating the catalyst. The rate increase occurs because the catalyst allows the reaction to occur by an alternative mechanism which may be much faster than the noncatalyzed mechanism. However the noncatalyzed mechanism does remain possible, so that the total rate (catalyzed plus noncatalyzed) can only increase in the presence of the catalyst and never decrease. Catalysis may be classified as either homogeneous, whose components are dispersed in the same phase (usual ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Akira Fujishima
is a Japanese chemist and president of Tokyo University of Science. He is known for significant contributions to the discovery and research of photocatalytic and superhydrophilic properties of titanium dioxide (TiO2), which is also known as the '' Honda-Fujishima effect''.(Originally published in Japanese 2 December 2003) Career and research In 1966 he earned his B. A. (Engineering) at the Faculty of Engineering, Yokohama National University, and in 1971 his Ph.D. (Engineering) at the Graduate School of Engineering, the University of Tokyo. In 1967, while working on his Ph.D. under the supervision of professor Kenichi Honda (本多 健一), he discovered the phenomenon of photocatalytic water decomposition (water photolysis) when he exposed a titanium dioxide electrode to strong light, later called the '' Honda-Fujishima effect''. The discovery of self-cleaning properties of titanium dioxide by the group under his supervision initiated a revolution in the ceramic, glass, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
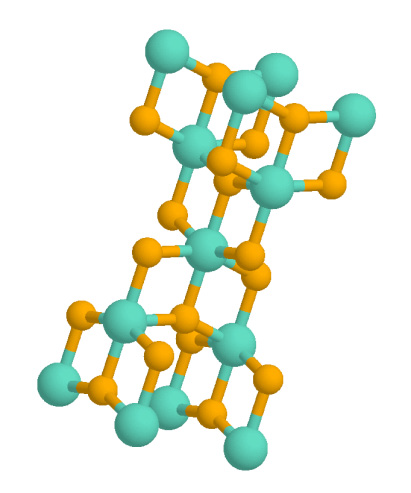
Anatase (titania, TiO2) Photocatalyst Producing Hydrogen
Anatase is a metastable mineral form of titanium dioxide (TiO2) with a tetragonal crystal structure. Although colorless or white when pure, anatase in nature is usually a black solid due to impurities. Three other polymorphs (or mineral forms) of titanium dioxide are known to occur naturally: brookite, akaogiite, and rutile, with rutile being the most common and most stable of the bunch. Anatase is formed at relatively low temperatures and found in minor concentrations in igneous and metamorphic rocks. Glass coated with a thin film of TiO2 shows antifogging and self-cleaning properties under ultraviolet radiation. Anatase is always found as small, isolated, and sharply developed crystals, and like rutile, it crystallizes in a tetragonal system. Anatase is metastable at all temperatures and pressures, with rutile being the equilibrium polymorph. Nevertheless, anatase is often the first titanium dioxide phase to form in many processes due to its lower surface energy, with a tra ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kenichi Honda
Kenichi Honda (本多健一, August 23, 1925 – February 26, 2011) was a Japanese chemist. He made a significant contribution to the discovery and characterization of photocatalytic properties of titanium dioxide (TiO2), for which he shared the 2004 Japan Prize with his former student Akira Fujishima. Biography In 1949 Honda received his bachelor's degree in engineering from the University of Tokyo. He then spent several years in France, where he defended a PhD in 1957 at the University of Paris. After returning to Japan he obtained a second doctorate degree, from the University of Tokyo in 1961. He then worked as a lecturer (1965–1975) and professor (1975–1983) at the University of Tokyo and at Kyoto University (1983–1989). In 1989 he moved to the Tokyo Polytechnic University and served as its president in 1996–2004. Research In the late 1960s Honda and his doctorate student Akira Fujishima discovered the Honda-Fujishima effect - photocatalytic water decomposition (phot ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Organic Solvents
A solvent (from the Latin language, Latin ''wikt:solvo#Latin, solvō'', "loosen, untie, solve") is a substance that dissolves a solute, resulting in a Solution (chemistry), solution. A solvent is usually a liquid but can also be a solid, a gas, or a supercritical fluid. Water is a solvent for Chemical polarity#Polarity of molecules, polar molecules, and the most common solvent used by living things; all the ions and proteins in a Cell (biology), cell are dissolved in water within the cell. Major uses of solvents are in paints, paint removers, inks, and dry cleaning. Specific uses for Organic compound, organic solvents are in dry cleaning (e.g. tetrachloroethylene); as paint thinners (toluene, turpentine); as nail polish removers and solvents of glue (acetone, methyl acetate, ethyl acetate); in spot removers (hexane, petrol ether); in detergents (D-limonene, citrus terpenes); and in perfumes (ethanol). Solvents find various applications in chemical, pharmaceutical, oil, and gas ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkene
In organic chemistry, an alkene, or olefin, is a hydrocarbon containing a carbon–carbon double bond. The double bond may be internal or at the terminal position. Terminal alkenes are also known as Alpha-olefin, α-olefins. The International Union of Pure and Applied Chemistry (IUPAC) Preferred IUPAC name, recommends using the name "alkene" only for Open-chain compound, acyclic hydrocarbons with just one double bond; alkadiene, alkatriene, etc., or polyene for acyclic hydrocarbons with two or more double bonds; cycloalkene, cycloalkadiene, etc. for Cyclic compound, cyclic ones; and "olefin" for the general class – cyclic or acyclic, with one or more double bonds. Acyclic alkenes, with only one double bond and no other functional groups (also known as mono-enes) form a homologous series of hydrocarbons with the general formula with ''n'' being a >1 natural number (which is two hydrogens less than the corresponding alkane). When ''n'' is four or more, isomers are possible, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Photodissociation
Photodissociation, photolysis, photodecomposition, or photofragmentation is a chemical reaction in which molecules of a chemical compound are broken down by absorption of light or photons. It is defined as the interaction of one or more photons with one target molecule that dissociates into two fragments. Here, “light” is broadly defined as radiation spanning the vacuum ultraviolet (VUV), ultraviolet (UV), visible, and infrared (IR) regions of the electromagnetic spectrum. To break covalent bonds, photon energies corresponding to visible, UV, or VUV light are typically required, whereas IR photons may be sufficiently energetic to detach ligands from coordination complexes or to fragment supramolecular complexes. Photolysis in photosynthesis Photolysis is part of the light-dependent reaction, light phase, photochemical phase, or Hill reaction of photosynthesis. The general reaction of photosynthetic photolysis can be given in terms of photons as: :\ce + 2 \text \longrigh ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Platinum
Platinum is a chemical element; it has Symbol (chemistry), symbol Pt and atomic number 78. It is a density, dense, malleable, ductility, ductile, highly unreactive, precious metal, precious, silverish-white transition metal. Its name originates from Spanish language, Spanish , a diminutive of "silver". Platinum is a member of the platinum group of elements and group 10 element, group 10 of the periodic table of elements. It has six naturally occurring isotopes. It is one of the Abundance of elements in Earth's crust, rarer elements in Earth's crust, with an average abundance of approximately 5 microgram, μg/kg, making platinum about 30 times rarer than gold. It occurs in some nickel and copper ores along with some Native element mineral, native deposits, with 90% of current production from deposits across Russia's Ural Mountains, Colombia, the Sudbury Basin, Sudbury basin of Canada, and a large reserve in South Africa. Because of its scarcity in Earth's crust, only a f ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen
Hydrogen is a chemical element; it has chemical symbol, symbol H and atomic number 1. It is the lightest and abundance of the chemical elements, most abundant chemical element in the universe, constituting about 75% of all baryon, normal matter. Under standard conditions, hydrogen is a gas of diatomic molecules with the chemical formula, formula , called dihydrogen, or sometimes hydrogen gas, molecular hydrogen, or simply hydrogen. Dihydrogen is colorless, odorless, non-toxic, and highly combustible. Stars, including the Sun, mainly consist of hydrogen in a plasma state, while on Earth, hydrogen is found as the gas (dihydrogen) and in molecular forms, such as in water and organic compounds. The most common isotope of hydrogen (H) consists of one proton, one electron, and no neutrons. Hydrogen gas was first produced artificially in the 17th century by the reaction of acids with metals. Henry Cavendish, in 1766–1781, identified hydrogen gas as a distinct substance and discovere ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |