|
Period (periodic Table)
A period on the periodic table is a row of chemical elements. All Chemical element, elements in a row have the same number of electron shells. Each next element in a period has one more proton and is less metallic than its predecessor. Arranged this way, elements in the same group (periodic table), group (column) have similar chemical property, chemical and physical property, physical properties, reflecting the periodic law. For example, the halogens lie in the second-to-last group (Halogen, group 17) and share similar properties, such as high reactivity and the tendency to gain one electron to arrive at a noble-gas electronic configuration. , a total of 118 elements have been discovered and confirmed. Modern quantum mechanics explains these periodic trends in properties in terms of electron shells. As atomic number increases, shells fill with electrons in approximately the order shown in the ordering rule diagram. The filling of each shell corresponds to a row in the table. In t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Simple Periodic Table Chart-blocks
Simple or SIMPLE may refer to: *Simplicity, the state or quality of being simple Arts and entertainment * Simple (album), ''Simple'' (album), by Andy Yorke, 2008, and its title track * Simple (Florida Georgia Line song), "Simple" (Florida Georgia Line song), 2018 * "Simple", a song by Johnny Mathis from the 1984 album ''A Special Part of Me'' * "Simple", a song by Collective Soul from the 1995 album ''Collective Soul (1995 album), Collective Soul'' * "Simple", a song by Katy Perry from the 2005 soundtrack to ''The Sisterhood of the Traveling Pants (film), The Sisterhood of the Traveling Pants'' * "Simple", a song by Khalil from the 2017 album ''Prove It All'' * "Simple", a song by Kreesha Turner from the 2008 album ''Passion (Kreesha Turner album), Passion'' * "Simple", a song by Ty Dolla Sign from the 2017 album ''Beach House 3'' deluxe version * Simple (video game series), ''Simple'' (video game series), budget-priced console games Businesses and organisations * Simple ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
P-block
A block of the periodic table is a set of elements unified by the atomic orbitals their valence electrons or vacancies lie in. The term seems to have been first used by Charles Janet. Each block is named after its characteristic orbital: s-block, p-block, d-block, f-block and g-block. The block names (s, p, d, and f) are derived from the spectroscopic notation for the value of an electron's azimuthal quantum number: sharp (0), principal (1), diffuse (2), and fundamental (3). Succeeding notations proceed in alphabetical order, as g, h, etc., though elements that would belong in such blocks have not yet been found. Characteristics There is an ''approximate'' correspondence between this nomenclature of blocks, based on electronic configuration, and sets of elements based on chemical properties. The s-block and p-block together are usually considered main-group elements, the d-block corresponds to the transition metals, and the f-block corresponds to the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Noble Gas
The noble gases (historically the inert gases, sometimes referred to as aerogens) are the members of Group (periodic table), group 18 of the periodic table: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn) and, in some cases, oganesson (Og). Under Standard temperature and pressure, standard conditions, the first six of these Chemical element, elements are odorless, colorless, monatomic gases with very low chemical reactivity and cryogenics, cryogenic boiling points. The properties of oganesson are uncertain. The intermolecular force between noble gas atoms is the very weak London dispersion force, so their boiling points are all cryogenic, below . The noble gases' Chemically inert, inertness, or tendency not to Chemical reaction, react with other chemical substances, results from their electron configuration: their Electron shell, outer shell of valence electrons is "full", giving them little tendency to participate in chemical reactions. Only a few hun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
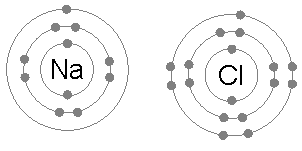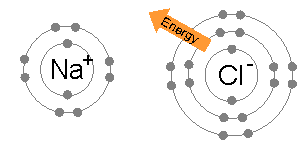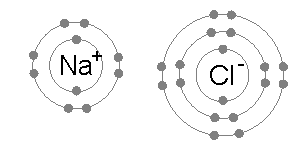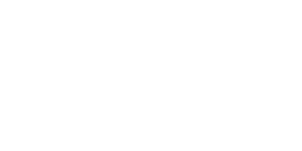
Duplet Rule
The octet rule is a chemical rule of thumb that reflects the theory that main-group elements tend to bond in such a way that each atom has eight electrons in its valence shell, giving it the same electronic configuration as a noble gas. The rule is especially applicable to carbon, nitrogen, oxygen, and the halogens; although more generally the rule is applicable for the s-block and p-block of the periodic table. Other rules exist for other elements, such as the duplet rule for hydrogen and helium, and the 18-electron rule for transition metals. The valence electrons in molecules like carbon dioxide (CO₂) can be visualized using a Lewis electron dot diagram. In covalent bonds, electrons shared between two atoms are counted toward the octet of both atoms. In carbon dioxide each oxygen shares four electrons with the central carbon, two (shown in red) from the oxygen itself and two (shown in black) from the carbon. All four of these electrons are counted in both the carbon oct ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Octet Rule
The octet rule is a chemical rule of thumb that reflects the theory that main-group elements tend to bond in such a way that each atom has eight electrons in its valence shell, giving it the same electronic configuration as a noble gas. The rule is especially applicable to carbon, nitrogen, oxygen, and the halogens; although more generally the rule is applicable for the s-block and p-block of the periodic table. Other rules exist for other elements, such as the duplet rule for hydrogen and helium, and the 18-electron rule for transition metals. The valence electrons in molecules like carbon dioxide (CO₂) can be visualized using a Lewis electron dot diagram. In covalent bonds, electrons shared between two atoms are counted toward the octet of both atoms. In carbon dioxide each oxygen shares four electrons with the central carbon, two (shown in red) from the oxygen itself and two (shown in black) from the carbon. All four of these electrons are counted in both the carbon ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Helium
Helium (from ) is a chemical element; it has chemical symbol, symbol He and atomic number 2. It is a colorless, odorless, non-toxic, inert gas, inert, monatomic gas and the first in the noble gas group in the periodic table. Its boiling point is the lowest among all the Chemical element, elements, and it does not have a melting point at standard pressures. It is the second-lightest and second-most Abundance of the chemical elements, abundant element in the observable universe, after hydrogen. It is present at about 24% of the total elemental mass, which is more than 12 times the mass of all the heavier elements combined. Its abundance is similar to this in both the Sun and Jupiter, because of the very high nuclear binding energy (per nucleon) of helium-4 with respect to the next three elements after helium. This helium-4 binding energy also accounts for why it is a product of both nuclear fusion and radioactive decay. The most common isotope of helium in the universe is helium-4, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen
Hydrogen is a chemical element; it has chemical symbol, symbol H and atomic number 1. It is the lightest and abundance of the chemical elements, most abundant chemical element in the universe, constituting about 75% of all baryon, normal matter. Under standard conditions, hydrogen is a gas of diatomic molecules with the chemical formula, formula , called dihydrogen, or sometimes hydrogen gas, molecular hydrogen, or simply hydrogen. Dihydrogen is colorless, odorless, non-toxic, and highly combustible. Stars, including the Sun, mainly consist of hydrogen in a plasma state, while on Earth, hydrogen is found as the gas (dihydrogen) and in molecular forms, such as in water and organic compounds. The most common isotope of hydrogen (H) consists of one proton, one electron, and no neutrons. Hydrogen gas was first produced artificially in the 17th century by the reaction of acids with metals. Henry Cavendish, in 1766–1781, identified hydrogen gas as a distinct substance and discovere ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Helium
Helium (from ) is a chemical element; it has chemical symbol, symbol He and atomic number 2. It is a colorless, odorless, non-toxic, inert gas, inert, monatomic gas and the first in the noble gas group in the periodic table. Its boiling point is the lowest among all the Chemical element, elements, and it does not have a melting point at standard pressures. It is the second-lightest and second-most Abundance of the chemical elements, abundant element in the observable universe, after hydrogen. It is present at about 24% of the total elemental mass, which is more than 12 times the mass of all the heavier elements combined. Its abundance is similar to this in both the Sun and Jupiter, because of the very high nuclear binding energy (per nucleon) of helium-4 with respect to the next three elements after helium. This helium-4 binding energy also accounts for why it is a product of both nuclear fusion and radioactive decay. The most common isotope of helium in the universe is helium-4, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen
Hydrogen is a chemical element; it has chemical symbol, symbol H and atomic number 1. It is the lightest and abundance of the chemical elements, most abundant chemical element in the universe, constituting about 75% of all baryon, normal matter. Under standard conditions, hydrogen is a gas of diatomic molecules with the chemical formula, formula , called dihydrogen, or sometimes hydrogen gas, molecular hydrogen, or simply hydrogen. Dihydrogen is colorless, odorless, non-toxic, and highly combustible. Stars, including the Sun, mainly consist of hydrogen in a plasma state, while on Earth, hydrogen is found as the gas (dihydrogen) and in molecular forms, such as in water and organic compounds. The most common isotope of hydrogen (H) consists of one proton, one electron, and no neutrons. Hydrogen gas was first produced artificially in the 17th century by the reaction of acids with metals. Henry Cavendish, in 1766–1781, identified hydrogen gas as a distinct substance and discovere ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Noble Gas
The noble gases (historically the inert gases, sometimes referred to as aerogens) are the members of Group (periodic table), group 18 of the periodic table: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn) and, in some cases, oganesson (Og). Under Standard temperature and pressure, standard conditions, the first six of these Chemical element, elements are odorless, colorless, monatomic gases with very low chemical reactivity and cryogenics, cryogenic boiling points. The properties of oganesson are uncertain. The intermolecular force between noble gas atoms is the very weak London dispersion force, so their boiling points are all cryogenic, below . The noble gases' Chemically inert, inertness, or tendency not to Chemical reaction, react with other chemical substances, results from their electron configuration: their Electron shell, outer shell of valence electrons is "full", giving them little tendency to participate in chemical reactions. Only a few hun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alkali Metal
The alkali metals consist of the chemical elements lithium (Li), sodium (Na), potassium (K),The symbols Na and K for sodium and potassium are derived from their Latin names, ''natrium'' and ''kalium''; these are still the origins of the names for the elements in some languages, such as German and Russian. rubidium (Rb), caesium (Cs), and francium (Fr). Together with hydrogen they constitute Group (periodic table)#Group names, group 1, which lies in the s-block of the periodic table. All alkali metals have their outermost electron in an s-orbital: this shared electron configuration results in their having very similar characteristic properties. Indeed, the alkali metals provide the best example of periodic trends, group trends in properties in the periodic table, with elements exhibiting well-characterised Homologous series, homologous behaviour. This family of elements is also known as the lithium family after its leading element. The alkali metals are all shiny, hardness, sof ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Periodic Table Group
In chemistry, a group (also known as a family) is a column of elements in the periodic table of the chemical elements. There are 18 numbered groups in the periodic table; the 14 f-block columns, between groups 2 and 3, are not numbered. The elements in a group have similar physical or chemical characteristics of the outermost electron shells of their atoms (i.e., the same core charge), because most chemical properties are dominated by the orbital location of the outermost electron. The modern numbering system of "group 1" to "group 18" has been recommended by the International Union of Pure and Applied Chemistry (IUPAC) since 1988. The 1-18 system is based on each atom's s, p and d electrons beyond those in atoms of the preceding noble gas. Two older incompatible naming schemes can assign the same number to different groups depending on the system being used. The older schemes were used by the Chemical Abstract Service (CAS, more popular in the United States), and by IUPAC befo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |