|
Nucleic Acid Metabolism
Nucleic acid metabolism refers to the set of chemical reactions involved in the synthesis and degradation of nucleic acids (DNA and RNA). Nucleic acids are polymers (biopolymers) composed of monomers called nucleotides. Nucleotide synthesis is an anabolism, anabolic process that typically involves the chemical reaction of a phosphate group, a pentose sugar, and a nitrogenous base. In contrast, the degradation of nucleic acids is a catabolism, catabolic process in which nucleotides or nucleobases are broken down, and their components can be salvaged to form new nucleotides. Both synthesis and degradation reactions require multiple enzymes to facilitate these processes. Defects or deficiencies in these enzymes can lead to a variety of metabolic disorders. Synthesis of nucleotides Nucleotides are the monomers that polymerize to form nucleic acids. Each nucleotide consists of a sugar, a phosphate group, and a nitrogenous base. The nitrogenous bases found in nucleic acids belong to o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nucleic Acids
Nucleic acids are large biomolecules that are crucial in all cells and viruses. They are composed of nucleotides, which are the monomer components: a 5-carbon sugar, a phosphate group and a nitrogenous base. The two main classes of nucleic acids are deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). If the sugar is ribose, the polymer is RNA; if the sugar is deoxyribose, a variant of ribose, the polymer is DNA. Nucleic acids are chemical compounds that are found in nature. They carry information in cells and make up genetic material. These acids are very common in all living things, where they create, encode, and store information in every living cell of every life-form on Earth. In turn, they send and express that information inside and outside the cell nucleus. From the inner workings of the cell to the young of a living thing, they contain and provide information via the nucleic acid sequence. This gives the RNA and DNA their unmistakable 'ladder-step' order of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Inosine Monophosphate
Inosinic acid or inosine monophosphate (IMP) is a nucleotide (that is, a nucleoside monophosphate). Widely used as a flavor enhancer, it is typically obtained from chicken byproducts or other meat industry waste. Inosinic acid is important in metabolism. It is the ribonucleotide of hypoxanthine and the first nucleotide formed during the synthesis of purine nucleotides. It can also be formed by the deamination of adenosine monophosphate by AMP deaminase. It can be hydrolysed to inosine. The enzyme deoxyribonucleoside triphosphate pyrophosphohydrolase, encoded by YJR069C in ''Saccharomyces cerevisiae'' and containing (d)ITPase and (d)XTPase activities, hydrolyzes inosine triphosphate (ITP) releasing pyrophosphate and IMP. Important derivatives of inosinic acid include the purine nucleotides found in nucleic acids and adenosine triphosphate, which is used to store chemical energy in muscle and other tissues. In the food industry, inosinic acid and its salts such as disodium in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
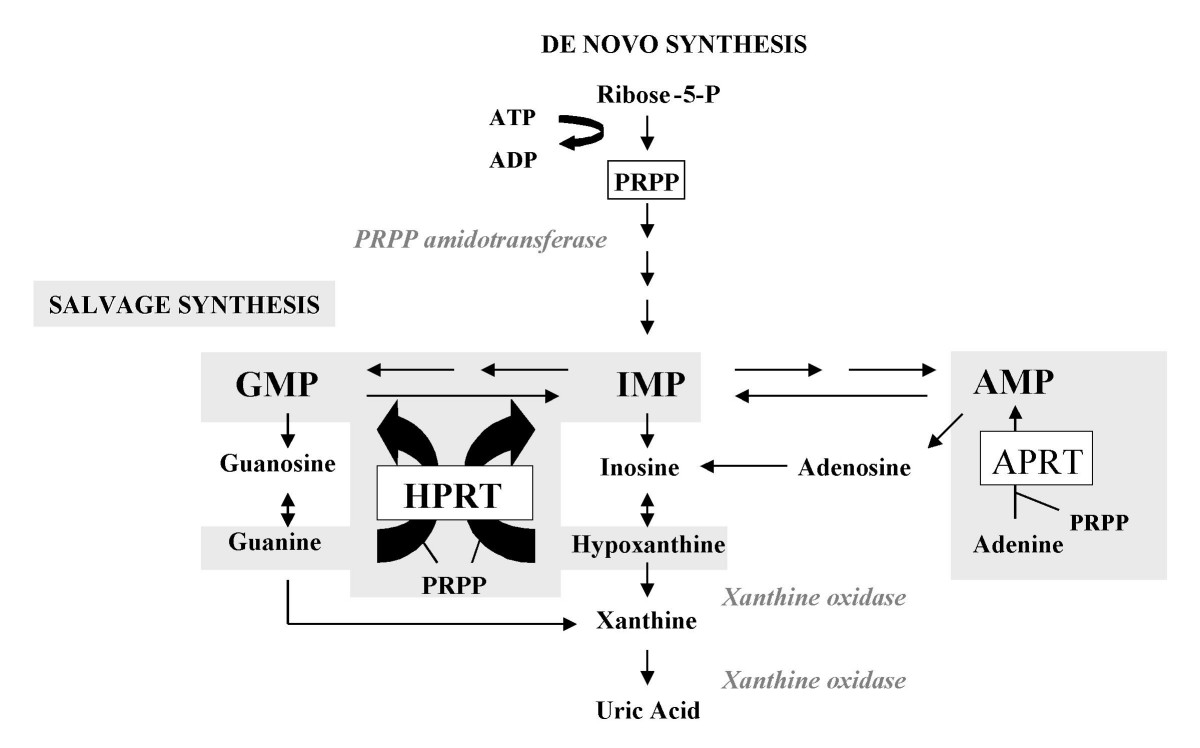
Lesch–Nyhan Syndrome
Lesch–Nyhan syndrome (LNS) is a rare inherited disorder caused by a deficiency of the enzyme hypoxanthine-guanine phosphoribosyltransferase (HGPRT). This deficiency occurs due to mutations in the '' HPRT1'' gene located on the X chromosome. LNS affects about 1 in 380,000 live births. The disorder was first recognized and clinically characterized by American medical student Michael Lesch and his mentor, pediatrician William Nyhan, at Johns Hopkins. The HGPRT deficiency causes a build-up of uric acid in all body fluids. The combination of increased synthesis and decreased utilization of purines leads to high levels of uric acid production. This results in both high levels of uric acid in the blood and urine, associated with severe gout and kidney problems. Neurological signs include poor muscle control and moderate intellectual disability. These complications usually appear in the first year of life. Beginning in the second year of life, a particularly striking feature of L ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glutamate
Glutamic acid (symbol Glu or E; known as glutamate in its anionic form) is an α-amino acid that is used by almost all living beings in the biosynthesis of proteins. It is a Essential amino acid, non-essential nutrient for humans, meaning that the human body can synthesize enough for its use. It is also the most abundant excitatory neurotransmitter in the vertebrate nervous system. It serves as the precursor for the synthesis of the inhibitory gamma-aminobutyric acid (GABA) in GABAergic neurons. Its molecular formula is . Glutamic acid exists in two optically isomeric forms; the optical rotation, dextrorotatory -form is usually obtained by hydrolysis of gluten or from the waste waters of beet-sugar manufacture or by fermentation.Webster's Third New International Dictionary of the English Language Unabridged, Third Edition, 1971. Its molecular structure could be idealized as HOOC−CH()−()2−COOH, with two carboxylic acid, carboxyl groups −COOH and one amine, amino group � ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Xanthosine Monophosphate
Xanthosine monophosphate (xanthylate) is an intermediate in purine metabolism. It is a ribonucleoside monophosphate. It is formed from IMP via the action of IMP dehydrogenase, and it forms GMP via the action of GMP synthase. Also, XMP can be released from XTP by enzyme deoxyribonucleoside triphosphate pyrophosphohydrolase containing (d)XTPase activity. It is abbreviated XMP. See also * Xanthosine Xanthosine is a nucleoside derived from xanthine and ribose. It is the biosynthetic precursor to 7-methylxanthosine by the action of 7-methylxanthosine synthase. 7-Methylxanthosine in turn is the precursor to theobromine (active alkaloid in cho ... References Further reading * * Nucleotides Xanthines {{Biochem-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nicotinamide Adenine Dinucleotide
Nicotinamide adenine dinucleotide (NAD) is a Cofactor (biochemistry), coenzyme central to metabolism. Found in all living cell (biology), cells, NAD is called a dinucleotide because it consists of two nucleotides joined through their phosphate groups. One nucleotide contains an adenine nucleobase and the other, nicotinamide. NAD exists in two forms: an Redox, oxidized and reduced form, abbreviated as NAD and NADH (H for hydrogen), respectively. In cellular metabolism, NAD is involved in redox reactions, carrying electrons from one reaction to another, so it is found in two forms: NAD is an oxidizing agent, accepting electrons from other molecules and becoming reduced; with H+, this reaction forms NADH, which can be used as a reducing agent to donate electrons. These electron transfer reactions are the main function of NAD. It is also used in other cellular processes, most notably as a substrate (biochemistry), substrate of enzymes in adding or removing chemical groups to or fr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fumarate
Fumaric acid or ''trans''-butenedioic acid is an organic compound with the formula HO2CCH=CHCO2H. A white solid, fumaric acid occurs widely in nature. It has a fruit-like taste and has been used as a food additive. Its E number is E297. The salts and esters are known as fumarates. Fumarate can also refer to the ion (in solution). Fumaric acid is the ''trans'' isomer of butenedioic acid, while maleic acid is the ''cis'' isomer. Biosynthesis and occurrence It is produced in eukaryotic organisms from succinate in complex 2 of the electron transport chain via the enzyme succinate dehydrogenase. Fumaric acid is found in fumitory (''Fumaria officinalis''), bolete mushrooms (specifically ''Boletus fomentarius var. pseudo-igniarius''), lichen, and Iceland moss. Fumarate is an intermediate in the citric acid cycle used by cells to produce energy in the form of adenosine triphosphate (ATP) from food. It is formed by the oxidation of succinate by the enzyme succinate dehydr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Guanosine Triphosphate
Guanosine-5'-triphosphate (GTP) is a purine nucleoside triphosphate. It is one of the building blocks needed for the synthesis of RNA during the transcription process. Its structure is similar to that of the guanosine nucleoside, the only difference being that nucleotides like GTP have phosphates on their ribose sugar. GTP has the guanine nucleobase attached to the 1' carbon of the ribose and it has the triphosphate moiety attached to ribose's 5' carbon. It also has the role of a source of energy or an activator of substrates in metabolic reactions, like that of ATP, but more specific. It is used as a source of energy for protein synthesis and gluconeogenesis. GTP is essential to signal transduction, in particular with G-proteins, in second-messenger mechanisms where it is converted to guanosine diphosphate (GDP) through the action of GTPases. Uses Energy transfer GTP is involved in energy transfer within the cell. For instance, a GTP molecule is generated by one of the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Guanosine Monophosphate
Guanosine monophosphate (GMP), also known as 5′-guanidylic acid or guanylic acid (conjugate base guanylate), is a nucleotide that is used as a monomer in RNA. It is an ester of phosphoric acid with the nucleoside guanosine. GMP consists of the phosphate group, the pentose sugar ribose, and the nucleobase guanine; hence it is a ribonucleotide monophosphate. Guanosine monophosphate is commercially produced by microbial fermentation. As an acyl substituent, it takes the form of the prefix guanylyl-. ''De novo'' synthesis GMP synthesis starts with D-ribose 5′-phosphate, a product of the pentose phosphate pathway. The synthesis proceeds by the gradual formation of the purine ring on carbon-1 of ribose, with CO2, glutamine, glycine, aspartate and one-carbon derivatives of tetrahydrofolate donating various elements towards the building of the ring. As inhibitor of guanosine monophosphate synthesis in experimental models, the glutamine analogue DON can be used.''Ahluwalia GS et al ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Adenosine Monophosphate
Adenosine monophosphate (AMP), also known as 5'-adenylic acid, is a nucleotide. AMP consists of a phosphate group, the sugar ribose, and the nucleobase adenine. It is an ester of phosphoric acid and the nucleoside adenosine. As a substituent it takes the form of the prefix adenylyl-. AMP plays an important role in many cellular metabolic processes, being interconverted to adenosine triphosphate (ATP) and adenosine diphosphate (ADP), as well as allosterically activating enzymes such as myophosphorylase-b. AMP is also a component in the synthesis of RNA. AMP is present in all known forms of life. Production and degradation AMP does not have the high energy phosphoanhydride bond associated with ADP and ATP. AMP can be produced from ADP by the myokinase (adenylate kinase) reaction when the ATP reservoir in the cell is low: : 2 ADP → ATP + AMP Or AMP may be produced by the hydrolysis of one high energy phosphate bond of ADP: : ADP + H2O → AMP + Pi AMP can also be forme ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Adenosine Triphosphate
Adenosine triphosphate (ATP) is a nucleoside triphosphate that provides energy to drive and support many processes in living cell (biology), cells, such as muscle contraction, nerve impulse propagation, and chemical synthesis. Found in all known forms of life, it is often referred to as the "molecular unit of currency" for intracellular energy transfer. When consumed in a Metabolism, metabolic process, ATP converts either to adenosine diphosphate (ADP) or to adenosine monophosphate (AMP). Other processes regenerate ATP. It is also a Precursor (chemistry), precursor to DNA and RNA, and is used as a coenzyme. An average adult human processes around 50 kilograms (about 100 mole (unit), moles) daily. From the perspective of biochemistry, ATP is classified as a nucleoside triphosphate, which indicates that it consists of three components: a nitrogenous base (adenine), the sugar ribose, and the Polyphosphate, triphosphate. Structure ATP consists of three parts: a sugar, an amine base ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |