|
Noscapine
Noscapine (also known as Narcotine, Nectodon, Nospen, Anarcotine and (archaic) Opiane) is a benzylisoquinoline alkaloid, of the phthalideisoquinoline structural subgroup, which has been isolated from numerous species of the family Papaveraceae (poppy family). It lacks significant hypnotic, euphoric, or analgesic effects affording it with very low addictive potential. This agent is primarily used for its antitussive (cough-suppressing) effects. Medical uses Noscapine is often used as an antitussive medication. A 2012 Dutch guideline, however, does not recommend its use for acute coughing. Side effects *Nausea *Vomiting *Loss of coordination *Hallucinations (auditory and visual) *Loss of sexual drive *Swelling of prostate * Loss of appetite * Dilated pupils * Increased heart rate * Shaking and muscle spasms *Chest pains *Increased alertness *Loss of any sleepiness *Loss of stereoscopic vision Interactions Noscapine can increase the effects of centrally sedating substances such ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Noscapine Biosynthesis
Noscapine (also known as Narcotine, Nectodon, Nospen, Anarcotine and (archaic) Opiane) is a benzylisoquinoline alkaloid, of the phthalideisoquinoline structural subgroup, which has been isolated from numerous species of the family Papaveraceae (poppy family). It lacks significant hypnotic, euphoric, or analgesic effects affording it with very low addictive potential. This agent is primarily used for its antitussive (cough-suppressing) effects. Medical uses Noscapine is often used as an antitussive medication. A 2012 Dutch guideline, however, does not recommend its use for acute coughing. Side effects *Nausea *Vomiting *Loss of coordination *Hallucinations (auditory and visual) *Loss of sexual drive *Swelling of prostate *Loss of appetite * Dilated pupils * Increased heart rate * Shaking and muscle spasms *Chest pains *Increased alertness *Loss of any sleepiness *Loss of stereoscopic vision Interactions Noscapine can increase the effects of centrally sedating substances such as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzylisoquinoline
Substitution of the heterocycle isoquinoline at the C1 position by a benzyl group provides 1‑benzylisoquinoline, the most widely examined of the numerous benzylisoquinoline structural isomers. The 1-benzylisoquinoline moiety can be identified within numerous compounds of pharmaceutical interest, such as moxaverine; but most notably it is found within the structures of a wide variety of plant natural products, collectively referred to as benzylisoquinoline alkaloids. This class is exemplified in part by the following compounds: papaverine, noscapine, codeine, morphine, apomorphine, berberine, tubocurarine. Biosynthesis (''S'')- Norcoclaurine ( higenamine) has been identified as the central 1-benzyl-tetrahydro-isoquinoline precursor from which numerous complex biosynthetic pathways eventually emerge. These pathways collectively lead to the structurally disparate compounds comprising the broad classification of plant natural products referred to as benzylisoquinoline alkaloids (BI ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Antitussive
Cold medicines are a group of medications taken individually or in combination as a treatment for the symptoms of the common cold and similar conditions of the upper respiratory tract. The term encompasses a broad array of drugs, including analgesics, antihistamines and decongestants, among many others. It also includes drugs which are marketed as cough suppressants or antitussives, but their effectiveness in reducing cough symptoms is unclear or minimal. While they have been used by 10% of American children in any given week, they are not recommended in Canada or the United States in children six years or younger because of lack of evidence showing effect and concerns of harm. In 2020, one version containing codeine and guaifenesin was the 377th most commonly prescribed medication in the United States with more than 400thousand prescriptions. Types There are a number of different cough and cold medications, which may be used for various coughing symptoms. The commerci ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
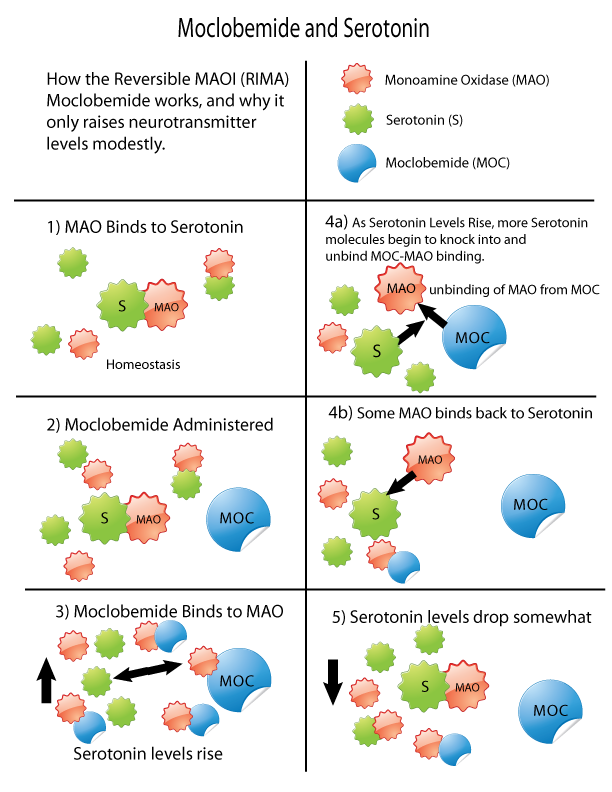
MAOI
Monoamine oxidase inhibitors (MAOIs) are a class of drugs that inhibit the activity of one or both monoamine oxidase enzymes: monoamine oxidase A (MAO-A) and monoamine oxidase B (MAO-B). They are best known as effective antidepressants, especially for treatment-resistant depression and atypical depression. They are also used to treat panic disorder, social anxiety disorder, Parkinson's disease, and several other disorders. Reversible inhibitors of monoamine oxidase A (RIMAs) are a subclass of MAOIs that selectively and reversibly inhibit the MAO-A enzyme. RIMAs are used clinically in the treatment of depression and dysthymia. Due to their reversibility, they are safer in single-drug overdose than the older, irreversible MAOIs, and weaker in increasing the monoamines important in depressive disorder. RIMAs have not gained widespread market share in the United States. Medical uses MAOIs have been found to be effective in the treatment of panic disorder with agoraphobia ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
(S)-norcoclaurine Synthase
The enzyme (''S'')-norcoclaurine synthase () catalyzes the chemical reaction : 4-hydroxyphenylacetaldehyde + 4-(2-aminoethyl)benzene-1,2-diol (Dopamine) \rightleftharpoons (''S'')-norcoclaurine + HO This enzyme belongs to the family of lyases, specifically the hydro-lyases, which cleave carbon-oxygen bonds. The systematic name of this enzyme class is 4-hydroxyphenylacetaldehyde hydro-lyase dding dopamine (''S'')-norcoclaurine-forming''. Other names in common use include (''S'')-norlaudanosoline synthase, and 4-hydroxyphenylacetaldehyde hydro-lyase (adding dopamine). This enzyme participates in benzylisoquinoline alkaloid Alkaloids are a class of basic BASIC (Beginners' All-purpose Symbolic Instruction Code) is a family of general-purpose, high-level programming languages designed for ease of use. The original version was created by John G. Kemeny and Th ... biosynthesis. References * * * EC 4.2.1 Enzymes of unknown structure {{4.2-enzyme-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dopamine
Dopamine (DA, a contraction of 3,4-dihydroxyphenethylamine) is a neuromodulatory molecule that plays several important roles in cells. It is an organic chemical of the catecholamine and phenethylamine families. Dopamine constitutes about 80% of the catecholamine content in the brain. It is an amine synthesized by removing a carboxyl group from a molecule of its precursor chemical, L-DOPA, which is synthesized in the brain and kidneys. Dopamine is also synthesized in plants and most animals. In the brain, dopamine functions as a neurotransmitter—a chemical released by neurons (nerve cells) to send signals to other nerve cells. Neurotransmitters are synthesized in specific regions of the brain, but affect many regions systemically. The brain includes several distinct dopamine pathways, one of which plays a major role in the motivational component of reward-motivated behavior. The anticipation of most types of rewards increases the level of dopamine in the brain, and ma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
3,4-dihydroxyphenylalanine
-DOPA, also known as levodopa and -3,4-dihydroxyphenylalanine, is an amino acid that is made and used as part of the normal biology of some plants and animals, including humans. Humans, as well as a portion of the other animals that utilize -DOPA, make it via biosynthesis from the amino acid -tyrosine. -DOPA is the precursor to the neurotransmitters dopamine, norepinephrine (noradrenaline), and epinephrine (adrenaline), which are collectively known as catecholamines. Furthermore, -DOPA itself mediates neurotrophic factor release by the brain and CNS. -DOPA can be manufactured and in its pure form is sold as a psychoactive drug with the INN levodopa; trade names include Sinemet, Pharmacopa, Atamet, and Stalevo. As a drug, it is used in the clinical treatment of Parkinson's disease and dopamine-responsive dystonia. -DOPA has a counterpart with opposite chirality, -DOPA. As is true for many molecules, the human body produces only one of these isomers (the -DOPA form). The enan ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
4-hydroxyphenylacetaldehyde
4-Hydroxyphenylacetaldehyde, also known as ''p''-hydroxyphenylacetaldehyde, is a natural product with the formula HOC6H4CH2CHO. It is a derivative of phenylacetaldehyde and occurs as a white solid at room temperature. Synthesis 4-Hydroxyphenylacetaldehyde can be synthesized from a parsley tyrosine decarboxylase (L-tyrosine). Occurrence 4-Hydroxyphenylacetaldehyde is produced from the metabolism of tyramine by monoamine oxidase (MAO) enzymes in humans and the tyramine oxidase (tynA) enzyme in ''Escherichia coli''. In both species, it is subsequently metabolized into 4-hydroxyphenylacetate by aldehyde dehydrogenase (ALDH) enzymes in humans and the phenylacetaldehyde dehydrogenase (feaB) enzyme in ''E. coli''. The condensation of 4-hydroxyphenylacetaldehyde and dopamine is a key step in the biosynthesis of benzylisoquinoline alkaloids. These natural products include berberine and morphine Morphine is a strong opiate that is found naturally in opium, a dark brown resin i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Thiamine Pyrophosphate
Thiamine pyrophosphate (TPP or ThPP), or thiamine diphosphate (ThDP), or cocarboxylase is a thiamine (vitamin B1) derivative which is produced by the enzyme thiamine diphosphokinase. Thiamine pyrophosphate is a cofactor that is present in all living systems, in which it catalyzes several biochemical reactions. Thiamine pyrophosphate is synthesized in the cytosol and is required in the cytosol for the activity of transketolase and in the mitochondria for the activity of pyruvate-, oxoglutarate- and branched chain keto acid dehydrogenases. To date, the yeast ThPP carrier (Tpc1p) the human Tpc and the ''Drosophila melanogaster'' have been identified as being responsible for the mitochondrial transport of ThPP and ThMP. It was first discovered as an essential nutrient (vitamin) in humans through its link with the peripheral nervous system disease beriberi, which results from a deficiency of thiamine in the diet. TPP works as a coenzyme in many enzymatic reactions, such as: * Pyruva ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
4-hydroxyphenylpyruvic Acid
4-Hydroxyphenylpyruvic acid (4-HPPA) is an intermediate in the metabolism of the amino acid phenylalanine. The aromatic side chain of phenylalanine is hydroxylated by the enzyme phenylalanine hydroxylase to form tyrosine. The conversion from tyrosine to 4-HPPA is in turn catalyzed by tyrosine aminotransferase. Additionally, 4-HPPA can be converted to homogentisic acid which is one of the precursors to ochronotic pigment. It is an intermediary compound in the biosynthesis of scytonemin. See also * 4-Hydroxyphenylpyruvate dioxygenase 4-Hydroxyphenylpyruvate dioxygenase (HPPD), also known as α-ketoisocaproate dioxygenase (KIC dioxygenase), is an Fe(II)-containing non-heme oxygenase that catalyzes the second reaction in the catabolism of tyrosine - the conversion of 4-hydroxy ... References Natural phenols Phenols Alpha-keto acids Propionic acids Hydroxy acids {{biochem-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pyridoxal Phosphate
Pyridoxal phosphate (PLP, pyridoxal 5'- phosphate, P5P), the active form of vitamin B6, is a coenzyme in a variety of enzymatic reactions. The International Union of Biochemistry and Molecular Biology has catalogued more than 140 PLP-dependent activities, corresponding to ~4% of all classified activities. The versatility of PLP arises from its ability to covalently bind the substrate, and then to act as an electrophilic catalyst, thereby stabilizing different types of carbanionic reaction intermediates. Role as a coenzyme PLP acts as a coenzyme in all transamination reactions, and in certain decarboxylation, deamination, and racemization reactions of amino acids. The aldehyde group of PLP forms a Schiff-base linkage (internal aldimine) with the ε-amino group of a specific lysine group of the aminotransferase enzyme. The α-amino group of the amino acid substrate displaces the ε-amino group of the active-site lysine residue in a process known as transaldimination. The r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tyrosine
-Tyrosine or tyrosine (symbol Tyr or Y) or 4-hydroxyphenylalanine is one of the 20 standard amino acids that are used by cells to synthesize proteins. It is a non-essential amino acid with a polar side group. The word "tyrosine" is from the Greek ''tyrós'', meaning ''cheese'', as it was first discovered in 1846 by German chemist Justus von Liebig in the protein casein from cheese. It is called tyrosyl when referred to as a functional group or side chain. While tyrosine is generally classified as a hydrophobic amino acid, it is more hydrophilic than phenylalanine. It is encoded by the codons UAC and UAU in messenger RNA. Functions Aside from being a proteinogenic amino acid, tyrosine has a special role by virtue of the phenol functionality. It occurs in proteins that are part of signal transduction processes and functions as a receiver of phosphate groups that are transferred by way of protein kinases. Phosphorylation of the hydroxyl group can change the activity of the targ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |