|
Nitryl Chloride
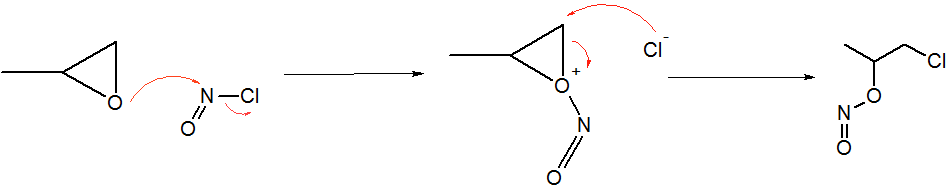
Nitryl chloride is a volatile inorganic compound with formula ClNO2. At standard conditions it is a gas. Formation Nitryl chloride can be formed in the reaction of dinitrogen pentoxide with chlorides or hydrogen chloride: :N2O5 + 2HCl → 2ClNO2 + H2O :N2O5 + NaCl → ClNO2 + NaNO3 Reactions Nitryl chloride adds to olefins in a radical reaction A free-radical reaction is any chemical reaction involving free radicals. This reaction type is abundant in organic reactions. Two pioneering studies into free radical reactions have been the discovery of the triphenylmethyl radical by Moses Gomb .... References {{Cite book , last=Stacey , first=F. W. , title=Organic Reactions , title-link=Organic Reactions , last2=Harris , first2=((J. F., Jr.)) , date=2004-04-30 , publisher=Wiley , isbn=978-0-471-26418-7 , editor-last=Denmark , editor-first=Scott E. , edition=1 , language=en , chapter=Formation of carbon-hetero atom bonds by free-radical chain additions to carbon-carbo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chloro Nitrite
Chlorine is a chemical element; it has Symbol (chemistry), symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between them. Chlorine is a yellow-green gas at room temperature. It is an extremely reactive element and a strong oxidizing agent, oxidising agent: among the elements, it has the highest electron affinity and the third-highest electronegativity on the revised Electronegativity#Pauling electronegativity, Pauling scale, behind only oxygen and fluorine. Chlorine played an important role in the experiments conducted by medieval Alchemy, alchemists, which commonly involved the heating of chloride Salt (chemistry), salts like ammonium chloride (sal ammoniac) and sodium chloride (common salt), producing various chemical substances containing chlorine such as hydrogen chloride, mercury(II) chloride (corrosive sublimate), and . However, the nature of fre ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chlorine Nitrate
Chlorine nitrate, with chemical formula ClONO2 is an important atmospheric gas present in the stratosphere. It is an important sink of reactive chlorine and nitrogen, and thus its formation and destruction play an important role in the depletion of ozone. __TOC__ Chemical properties It explosively reacts with metals, metal chlorides, alcohols, ethers, and most organic materials. When it is heated to decomposition, it emits toxic fumes of Cl2 and NOx. Synthesis and reactions It can be produced by the reaction of dichlorine monoxide and dinitrogen pentoxide at 0 °C: :Cl2O + N2O5 → 2 ClONO2 or by the reaction: :ClF + HNO3 → HF + ClONO2 It can also react with alkenes: :(CH3)2C=CH2 + ClONO2 → O2NOC(CH3)2CH2Cl Chlorine nitrate reacts with metal chloride The term chloride refers to a compound or molecule that contains either a chlorine anion (), which is a negatively charged chlorine atom, or a non-charged chlorine atom covalently bonded to the rest of the molecule ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitryl Fluoride
Nitryl fluoride, NO2F, is a colourless gas and strong oxidizing agent, which is used as a fluorinating agent and has been proposed as an oxidiser in rocket propellants (though never flown). It is a molecular species, not ionic, consistent with its low boiling point. The structure features planar nitrogen with a short N-F bond length of 135 pm. Preparation Henri Moissan and Paul Lebeau recorded the preparation of nitryl fluoride in 1905 by the fluorination of nitrogen dioxide. This reaction is highly exothermic, which leads to contaminated products. The simplest method avoids fluorine gas but uses cobalt(III) fluoride: :NO2 + CoF3 → NO2F + CoF2 The CoF2 can be regenerated to CoF3. Other methods have been described. Thermodynamic properties The thermodynamic properties of this gas were determined by IR and Raman spectroscopy. The standard heat of formation of FNO2 is -19 ± 2 kcal/mol, but the compound becomes increasingly unstable at higher temperature. The homogeneou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitryl Bromide
Nitryl is the nitrogen dioxide (NO2) moiety when it occurs in a larger compound as a univalent fragment. Examples include nitryl fluoride (NO2F) and nitryl chloride (NO2Cl). Like nitrogen dioxide, the nitryl moiety contains a nitrogen atom with two bonds to the two oxygen atoms, and a third bond shared equally between the nitrogen and the two oxygen atoms. The nitrogen-centred radical is then free to form a bond with another univalent fragment (X) to produce an N−X bond, where X can be F, Cl, OH, etc. In organic nomenclature, the nitryl moiety is known as the nitro group. For instance, nitryl benzene is normally called nitrobenzene (PhNO2). See also * Dinitrogen tetroxide * Nitro compound * Nitrosyl (R−N=O) * Isocyanide (R−N≡C) * Nitryl fluoride * Nitrate Nitrate is a polyatomic ion with the chemical formula . salt (chemistry), Salts containing this ion are called nitrates. Nitrates are common components of fertilizers and explosives. Almost all inorganic nitrat ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrosyl Chloride
Nitrosyl chloride is the chemical compound with the formula NOCl. It is a yellow gas that is commonly encountered as a component of aqua regia, a mixture of 3 parts concentrated hydrochloric acid and 1 part of concentrated nitric acid. It is a strong electrophile and oxidizing agent. It is sometimes called Tilden's reagent, after William A. Tilden, who was the first to produce it as a pure compound. Structure and synthesis The molecule is bent. A double bond exists between N and O (distance = 1.16 Å) and a single bond between N and Cl (distance = 1.96 Å). The O=N–Cl angle is 113°. Production Nitrosyl chloride can be produced in many ways. * Combining nitrosylsulfuric acid and HCl affords the compound. This method is used industrially. :HCl + NOHSO4 → H2SO4 + NOCl * A more convenient laboratory method involves the (reversible) dehydration of nitrous acid by HCl : HNO2 + HCl → H2O + NOCl * By the direct combination of chlorine and nitric ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfuryl Chloride
Sulfuryl chloride is an inorganic compound with the formula SO2Cl2. At room temperature, it is a colorless liquid with a pungent odor. Sulfuryl chloride is not found in nature. Sulfuryl chloride is commonly confused with thionyl chloride, SOCl2. The properties of these two sulfur oxychlorides are quite different: sulfuryl chloride is a source of chlorine whereas thionyl chloride is a source of chloride ions. An alternative IUPAC name is sulfuryl dichloride. Sulfur is tetrahedral in SO2Cl2 and the oxidation state of the sulfur atom is +6, as in sulfuric acid. Synthesis SO2Cl2 is prepared by the reaction of sulfur dioxide and chlorine in the presence of a catalyst, such as activated carbon. :SO2 + Cl2 → SO2Cl2 The product can be purified by fractional distillation. Legacy routes Sulfuryl chloride was first prepared in 1838 by the French chemist Henri Victor Regnault. Older routes include oxidation of thionyl chloride: :5 SOCl2 + HgO → ClSSCl + HgCl2 + 3 SO2Cl2 :2 SOCl2 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dinitrogen Pentoxide
Dinitrogen pentoxide (also known as nitrogen pentoxide or nitric anhydride) is the chemical compound with the formula . It is one of the binary nitrogen oxides, a family of compounds that contain only nitrogen and oxygen. It exists as colourless crystals that sublime slightly above room temperature, yielding a colorless gas.Connell, Peter Steele. (1979) The Photochemistry of Dinitrogen Pentoxide'. Ph. D. thesis, Lawrence Berkeley National Laboratory. Dinitrogen pentoxide is an unstable and potentially dangerous oxidizer that once was used as a reagent when dissolved in chloroform for nitrations but has largely been superseded by nitronium tetrafluoroborate (). is a rare example of a compound that adopts two structures depending on the conditions. The solid is a salt, nitronium nitrate, consisting of separate nitronium cations and nitrate anions ; but in the gas phase and under some other conditions it is a covalently-bound molecule. History was first reported by the Fre ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrogen Chloride
The Chemical compound, compound hydrogen chloride has the chemical formula and as such is a hydrogen halide. At room temperature, it is a colorless gas, which forms white fumes of hydrochloric acid upon contact with atmospheric water vapor. Hydrogen chloride gas and hydrochloric acid are important in technology and industry. Hydrochloric acid, the aqueous solution of hydrogen chloride, is also commonly given the formula HCl. Reactions Hydrogen chloride is a diatomic molecule, consisting of a hydrogen atom H and a chlorine atom Cl connected by a Polar-covalent bond, polar covalent bond. The chlorine atom is much more Electronegativity, electronegative than the hydrogen atom, which makes this bond polar. Consequently, the molecule has a large Molecular dipole moment, dipole moment with a negative partial charge (δ−) at the chlorine atom and a positive partial charge (δ+) at the hydrogen atom. In part because of its high polarity, HCl is very soluble in water (and in other ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Olefin
In organic chemistry, an alkene, or olefin, is a hydrocarbon containing a carbon–carbon double bond. The double bond may be internal or at the terminal position. Terminal alkenes are also known as α-olefins. The International Union of Pure and Applied Chemistry (IUPAC) recommends using the name "alkene" only for acyclic hydrocarbons with just one double bond; alkadiene, alkatriene, etc., or polyene for acyclic hydrocarbons with two or more double bonds; cycloalkene, cycloalkadiene, etc. for cyclic ones; and "olefin" for the general class – cyclic or acyclic, with one or more double bonds. Acyclic alkenes, with only one double bond and no other functional groups (also known as mono-enes) form a homologous series of hydrocarbons with the general formula with ''n'' being a >1 natural number (which is two hydrogens less than the corresponding alkane). When ''n'' is four or more, isomers are possible, distinguished by the position and conformation of the double bond. A ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radical Addition
In organic chemistry, free-radical addition is an addition reaction which involves free radicals. These reactions can happen due to the free radicals having an unpaired electron in their valence shell, making them highly reactive. Radical additions are known for a variety of unsaturated substrates, both olefinic or aromatic and with or without heteroatoms. Free-radical reactions depend on one or more relatively weak bonds in a reagent. Under reaction conditions (typically heat or light), some weak bonds homolyse into radicals, which then induce further decomposition in their compatriots before recombination. Different mechanisms typically apply to reagents without such a weak bond. Mechanism and regiochemistry The basic steps in any free-radical process (the radical chain mechanism) divide into: * Radical initiation: A radical is created from a non-radical precursor. * Chain propagation: A radical reacts with a non-radical to produce a new radical species * Chain termi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitro Compounds
In organic chemistry, nitro compounds are organic compounds that contain one or more nitro functional groups (). The nitro group is one of the most common explosophores (functional group that makes a compound explosive) used globally. The nitro group is also strongly electron-withdrawing group, electron-withdrawing. Because of this property, bonds alpha (adjacent) to the nitro group can be acidic. For similar reasons, the presence of nitro groups in aromatic compounds retards electrophilic aromatic substitution but facilitates nucleophilic aromatic substitution. Nitro groups are rarely found in nature. They are almost invariably produced by nitration reactions starting with nitric acid. Synthesis Preparation of aromatic nitro compounds Aromatic nitro compounds are typically synthesized by nitration. Nitration is achieved using a mixture of nitric acid and sulfuric acid, which produce the nitronium ion (), which is the electrophile: + The nitration product produced on ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nitrogen Oxohalides
Nitrogen is a chemical element; it has symbol N and atomic number 7. Nitrogen is a nonmetal and the lightest member of group 15 of the periodic table, often called the pnictogens. It is a common element in the universe, estimated at seventh in total abundance in the Milky Way and the Solar System. At standard temperature and pressure, two atoms of the element bond to form N2, a colourless and odourless diatomic gas. N2 forms about 78% of Earth's atmosphere, making it the most abundant chemical species in air. Because of the volatility of nitrogen compounds, nitrogen is relatively rare in the solid parts of the Earth. It was first discovered and isolated by Scottish physician Daniel Rutherford in 1772 and independently by Carl Wilhelm Scheele and Henry Cavendish at about the same time. The name was suggested by French chemist Jean-Antoine-Claude Chaptal in 1790 when it was found that nitrogen was present in nitric acid and nitrates. Antoine Lavoisier suggested instead the nam ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |