|
Lithopone
Lithopone, C.I. Pigment White 5, is a mixture of inorganic compounds, widely used as a white pigment powder. It is composed of a mixture of barium sulfate and zinc sulfide. These insoluble compounds blend well with organic compounds and confer opacity. It was made popular by the cheap production costs, greater coverage. Related white pigments include titanium dioxide, zinc oxide ("zinc white"), zinc sulfide, and white lead. History Lithopone was discovered in the 1870s by DuPont. It was manufactured by Krebs Pigments and Chemical Company and other companies. The material came in different "seals", which varied in the content of zinc sulfide. Gold seal and Bronze seals contain 40-50% zinc sulfide, offering more hiding power and strength. Although its popularity peaked around 1920, approximately 223,352 tons were produced in 1990. It is mainly used in paints, putty, and in plastics. Stability and darkening Although barium sulfate is almost completely inert, zinc sulfide deg ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Krebs Pigments And Chemical Company
Krebs Pigments and Chemical Company was founded in 1902 by Henrik J. Krebs and was a manufacturer of lithopone and titanium dioxide. Following Krebs death it would be purchased by DuPont and eventually liquidated simply becoming a part of DuPont. History The company was founded in 1902 by Henrik J. Krebs in Newport, Delaware. After the death of Krebs in 1929 the company was purchased by DuPont. In 1931, DuPont formed a joint venture with Commercial Pigments Corporation to scale up titanium dioxide production for use as a white pigment, to compete against National Lead. The joint venture was named the Krebs Pigment and Color Corporation. By 1933, National Lead and DuPont ended their patent litigation and cross-licensed their patents and synthetic routes. In 1934, DuPont purchased the Commercial Pigments Corporation's 30 percent stake in the company for $7,420,000. In 1935 Phelps was named the assistant general manager. It then became a wholly-owned subsidiary of DuPont. DuP ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Sulfate
Zinc sulfate is an inorganic compound with the formula ZnSO4. It forms hydrates ZnSO4·''n''H2O, where ''n'' can range from 0 to 7. All are colorless solids. The most common form includes water of crystallization as the heptahydrate, with the chemical formula, formula . As early as the 16th century it was prepared on a large scale, and was historically known as "white vitriol" (the name was used, for example, in 1620s by the collective writing under the pseudonym of Basil Valentine). Zinc sulfate and its hydrates are colourless solids. Uses Manufacturing The main application of the heptahydrate is as a coagulant in the production of rayon. It is also a precursor to the pigment lithopone. It is also used as an electrolyte for zinc electroplating, as a mordant in dyeing, and as a preservative for skins and leather. Nutrition Zinc sulfate is used to supply zinc in animal feeds, fertilizers, toothpaste, and agricultural sprays. Zinc sulfate, like many zinc compounds, can be used to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Barium Sulfate
Barium sulfate (or sulphate) is the inorganic compound with the chemical formula Ba SO4. It is a white crystalline solid that is odorless and insoluble in water. It occurs in nature as the mineral barite, which is the main commercial source of barium and materials prepared from it. Its opaque white appearance and its high density are exploited in its main applications.Holleman, A. F. and Wiberg, E. (2001) ''Inorganic Chemistry'', San Diego, CA. Academic Press, . Uses Drilling fluids About 80% of the world's barium sulfate production, mostly purified mineral, is consumed as a component of oil well drilling fluid. It increases the density of the fluid, increasing the hydrostatic pressure in the well and reducing the chance of a blowout. Radiocontrast agent Barium sulfate in suspension is often used medically as a radiocontrast agent for X-ray imaging and other diagnostic procedures. It is most often used in imaging of the GI tract during what is colloquially known as a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
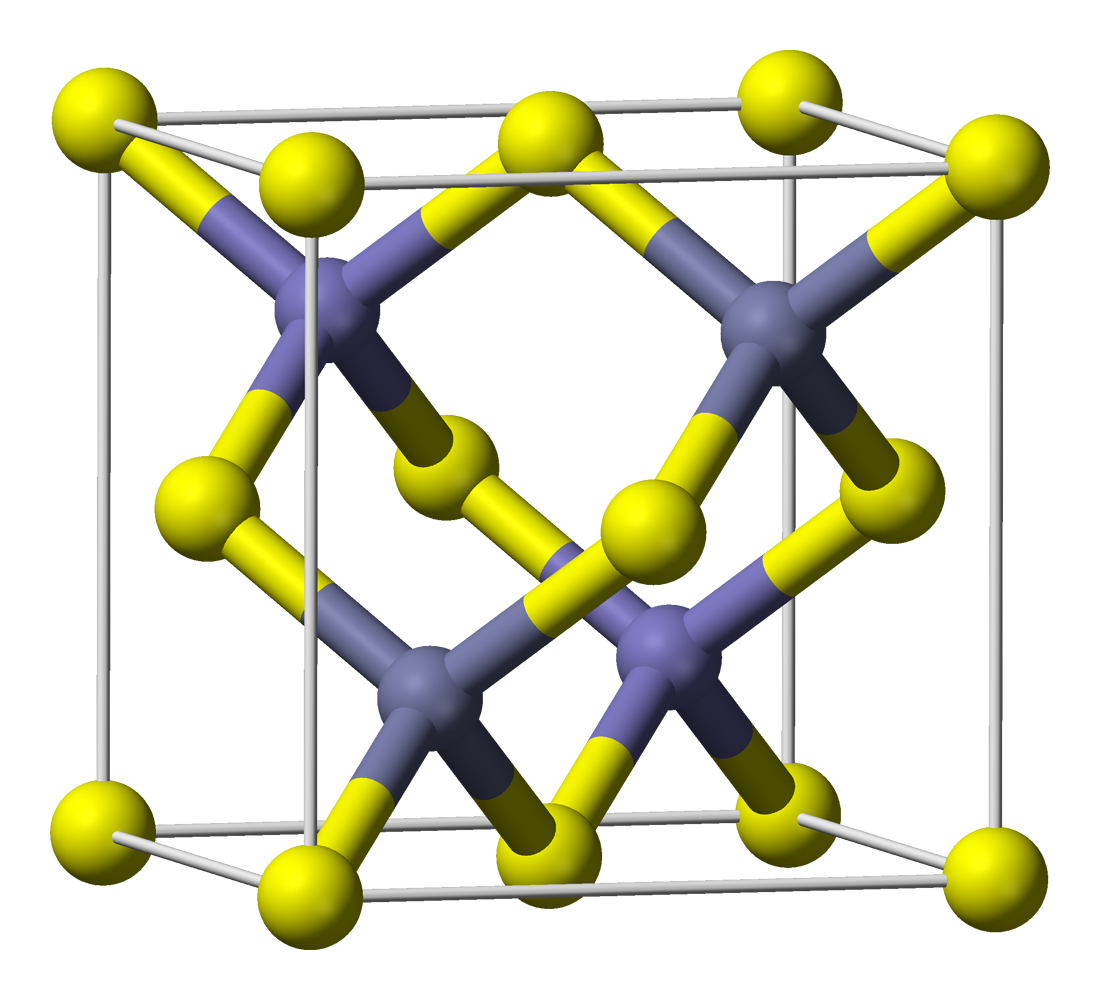
Zinc Sulfide
Zinc sulfide (or zinc sulphide) is an inorganic compound with the chemical formula of ZnS. This is the main form of zinc found in nature, where it mainly occurs as the mineral sphalerite. Although this mineral is usually black because of various impurities, the pure material is white, and it is widely used as a pigment. In its dense synthetic form, zinc sulfide can be transparency and translucency, transparent, and it is used as a window for visible light, visible optics and infrared optics. Structure ZnS exists in two main crystalline forms. This dualism is an example of polymorphism (materials science), polymorphism. In each form, the coordination geometry at Zn and S is tetrahedral. The more stable cubic form is known also as zinc blende or sphalerite. The hexagonal form is known as the mineral wurtzite, although it also can be produced synthetically.. The transition from the sphalerite form to the wurtzite form occurs at around 1020 °C. Applications Luminescent mate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Oxide
Zinc oxide is an inorganic compound with the Chemical formula, formula . It is a white powder which is insoluble in water. ZnO is used as an additive in numerous materials and products including cosmetics, Zinc metabolism, food supplements, rubbers, plastics, ceramics, glass, cement, lubricants, paints, sunscreens, ointments, adhesives, sealants, pigments, foods, batteries, ferrites, fire retardants, semi conductors, and first-aid tapes. Although it occurs naturally as the mineral zincite, most zinc oxide is produced synthetically. History Early humans probably used zinc compounds in processed and unprocessed forms, as paint or medicinal ointment; however, their composition is uncertain. The use of ''pushpanjan'', probably zinc oxide, as a salve for eyes and open wounds is mentioned in the Indian medical text the Charaka Samhita, thought to date from 500 BC or before. Zinc oxide ointment is also mentioned by the Greek physician Dioscorides (1st century AD). Galen suggested treatin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Barium Sulfide
Barium sulfide is the inorganic compound with the formula Ba S. BaS is the barium compound produced on the largest scale. It is an important precursor to other barium compounds including barium carbonate and the pigment lithopone, ZnS/BaSO4. Like other chalcogenides of the alkaline earth metals, BaS is a short wavelength emitter for electronic displays. It is colorless, although like many sulfides, it is commonly obtained in impure colored forms. Discovery BaS was prepared by the Italian alchemist Vincenzo Cascariolo (also known as Vincentius or Vincentinus Casciarolus or Casciorolus, 1571–1624) via the thermo-chemical reduction of BaSO4 (available as the mineral barite). It is currently manufactured by an improved version of Cascariolo's process using coke in place of flour and charcoal. This kind of conversion is called a carbothermic reaction: : and also: : The basic method remains in use today. BaS dissolves in water. These aqueous solutions, when treated with so ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pigment
A pigment is a powder used to add or alter color or change visual appearance. Pigments are completely or nearly solubility, insoluble and reactivity (chemistry), chemically unreactive in water or another medium; in contrast, dyes are colored substances which are soluble or go into solution at some stage in their use. Dyes are often organic compounds whereas pigments are often inorganic compound, inorganic. Pigments of prehistoric and historic value include ochre, charcoal, and lapis lazuli. Economic impact In 2006, around 7.4 million tons of inorganic chemistry, inorganic, organic chemistry, organic, and special pigments were marketed worldwide. According to an April 2018 report by ''Bloomberg Businessweek'', the estimated value of the pigment industry globally is $30 billion. The value of titanium dioxide – used to enhance the white brightness of many products – was placed at $13.2 billion per year, while the color Ferrari red is valued at $300 million each yea ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Sulfate
Zinc sulfate is an inorganic compound with the formula ZnSO4. It forms hydrates ZnSO4·''n''H2O, where ''n'' can range from 0 to 7. All are colorless solids. The most common form includes water of crystallization as the heptahydrate, with the chemical formula, formula . As early as the 16th century it was prepared on a large scale, and was historically known as "white vitriol" (the name was used, for example, in 1620s by the collective writing under the pseudonym of Basil Valentine). Zinc sulfate and its hydrates are colourless solids. Uses Manufacturing The main application of the heptahydrate is as a coagulant in the production of rayon. It is also a precursor to the pigment lithopone. It is also used as an electrolyte for zinc electroplating, as a mordant in dyeing, and as a preservative for skins and leather. Nutrition Zinc sulfate is used to supply zinc in animal feeds, fertilizers, toothpaste, and agricultural sprays. Zinc sulfate, like many zinc compounds, can be used to ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Barium Sulfide
Barium sulfide is the inorganic compound with the formula Ba S. BaS is the barium compound produced on the largest scale. It is an important precursor to other barium compounds including barium carbonate and the pigment lithopone, ZnS/BaSO4. Like other chalcogenides of the alkaline earth metals, BaS is a short wavelength emitter for electronic displays. It is colorless, although like many sulfides, it is commonly obtained in impure colored forms. Discovery BaS was prepared by the Italian alchemist Vincenzo Cascariolo (also known as Vincentius or Vincentinus Casciarolus or Casciorolus, 1571–1624) via the thermo-chemical reduction of BaSO4 (available as the mineral barite). It is currently manufactured by an improved version of Cascariolo's process using coke in place of flour and charcoal. This kind of conversion is called a carbothermic reaction: : and also: : The basic method remains in use today. BaS dissolves in water. These aqueous solutions, when treated with so ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Inorganic Compound
An inorganic compound is typically a chemical compound that lacks carbon–hydrogen bondsthat is, a compound that is not an organic compound. The study of inorganic compounds is a subfield of chemistry known as ''inorganic chemistry''. Inorganic compounds comprise most of the Earth's crust, although the compositions of the deep Mantle (geology), mantle remain active areas of investigation. All allotropes (structurally different pure forms of an element) and some simple carbon compounds are often considered inorganic. Examples include the allotropes of carbon (graphite, diamond, buckminsterfullerene, graphene, etc.), carbon monoxide , carbon dioxide , carbides, and salt (chemistry), salts of inorganic anions such as carbonates, cyanides, cyanates, thiocyanates, isothiocyanates, etc. Many of these are normal parts of mostly organic systems, including organisms; describing a chemical as inorganic does not necessarily mean that it cannot occur within life, living things. History ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfates
The sulfate or sulphate ion is a Polyatomic ion, polyatomic anion with the empirical formula . Salts, acid derivatives, and peroxides of sulfate are widely used in industry. Sulfates occur widely in everyday life. Sulfates are salt (chemistry), salts of sulfuric acid and many are prepared from that acid. Spelling "Sulfate" is the spelling recommended by International Union of Pure and Applied Chemistry, IUPAC, but "sulphate" was traditionally used in British English. Structure The sulfate anion consists of a central sulfur atom surrounded by four equivalent oxygen atoms in a tetrahedron, tetrahedral arrangement. The symmetry of the isolated anion is the same as that of methane. The sulfur atom is in the +6 oxidation state while the four oxygen atoms are each in the −2 state. The sulfate ion carries an overall charge (physics), charge of −2 and it is the conjugate acid, conjugate base of the bisulfate (or hydrogensulfate) ion, , which is in turn the conjugate base of , sulfu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfides
Sulfide (also sulphide in British English) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive. ''Sulfide'' also refers to large families of inorganic and organic compounds, e.g. lead sulfide and dimethyl sulfide. Hydrogen sulfide (H2S) and bisulfide (HS−) are the conjugate acids of sulfide. Chemical properties The sulfide ion does not exist in aqueous alkaline solutions of Na2S. Instead sulfide converts to hydrosulfide: :S2− + H2O → SH− + OH− Upon treatment with an acid, sulfide salts convert to hydrogen sulfide: :S2− + H+ → SH− :SH− + H+ → H2S Oxidation of sulfide is a complicated process. Depending on the conditions, the oxidation can produce elemental sulfur, polysulfides, polythionates, sulfite, or sulfate. Metal sulfides react with halogens, forming sulfur and metal salts. :8 MgS + 8 I2 → S8 + 8 MgI2 Metal derivati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |