|
Kappa-carbides
κ-Carbides are a special class of carbide structures. They are most known for appearing in steels containing manganese and aluminium where they have the chemical formula, molecular formula . Properties Structure κ-Carbides crystallise in the perovskite Strukturbericht designation, structure type with the space group ''Pm3m'' (Nr. 221). This structure was, inter alia, elucidated with X-ray crystallography, XRD-measurements on steel alloys containing κ-carbide precipitates but also on single crystals of manganese-κ-carbides with a molecular formula of Mn3.1Al0.9C and a lattice parameter of ''a=3.87Å''. In steel alloys where diverse arrangements of the atoms are possible, a considerable effect of the short range ordering, e.g. of iron and manganese on the microscopic properties of the alloy, has been observed. This is especially important for the role as hydrogen-traps in steels. Composition A first glance at the composition of a steel alloy is achieved by analysing its ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbide
In chemistry, a carbide usually describes a compound composed of carbon and a metal. In metallurgy, carbiding or carburizing is the process for producing carbide coatings on a metal piece. Interstitial / Metallic carbides The carbides of the group 4, 5 and 6 transition metals (with the exception of chromium) are often described as interstitial compounds. These carbides have metallic properties and are refractory. Some exhibit a range of stoichiometries, being a non-stoichiometric mixture of various carbides arising due to crystal defects. Some of them, including titanium carbide and tungsten carbide, are important industrially and are used to coat metals in cutting tools. The long-held view is that the carbon atoms fit into octahedral interstices in a close-packed metal lattice when the metal atom radius is greater than approximately 135 pm: *When the metal atoms are cubic close-packed, (ccp), then filling all of the octahedral interstices with carbon achieves 1:1 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Austenite
Austenite, also known as gamma-phase iron (γ-Fe), is a metallic, non-magnetic allotrope of iron or a solid solution of iron with an alloying element. In plain-carbon steel, austenite exists above the critical eutectoid temperature of 1000 K (727 °C); other alloys of steel have different eutectoid temperatures. The austenite allotrope is named after Sir William Chandler Roberts-Austen (1843–1902). It exists at room temperature in some stainless steels due to the presence of nickel stabilizing the austenite at lower temperatures. Allotrope of iron From alpha iron undergoes a phase transition from body-centered cubic (BCC) to the face-centered cubic (FCC) configuration of gamma iron, also called austenite. This is similarly soft and ductile but can dissolve considerably more carbon (as much as 2.03% by mass at ). This gamma form of iron is present in the most commonly used type of stainless steel for making hospital and food-service equipment. Material Austenitiz ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Precipitate
In an aqueous solution, precipitation is the "sedimentation of a solid material (a precipitate) from a liquid solution". The solid formed is called the precipitate. In case of an inorganic chemical reaction leading to precipitation, the chemical reagent causing the solid to form is called the precipitant. The clear liquid remaining above the precipitated or the centrifuged solid phase is also called the supernate or supernatant. The notion of precipitation can also be extended to other domains of chemistry (organic chemistry and biochemistry) and even be applied to the solid phases (e.g. metallurgy and alloys) when solid impurities segregate from a solid phase. Supersaturation The precipitation of a compound may occur when its concentration exceeds its solubility. This can be due to temperature changes, solvent evaporation, or by mixing solvents. Precipitation occurs more rapidly from a strongly supersaturated solution. The formation of a precipitate can be caused by a ch ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
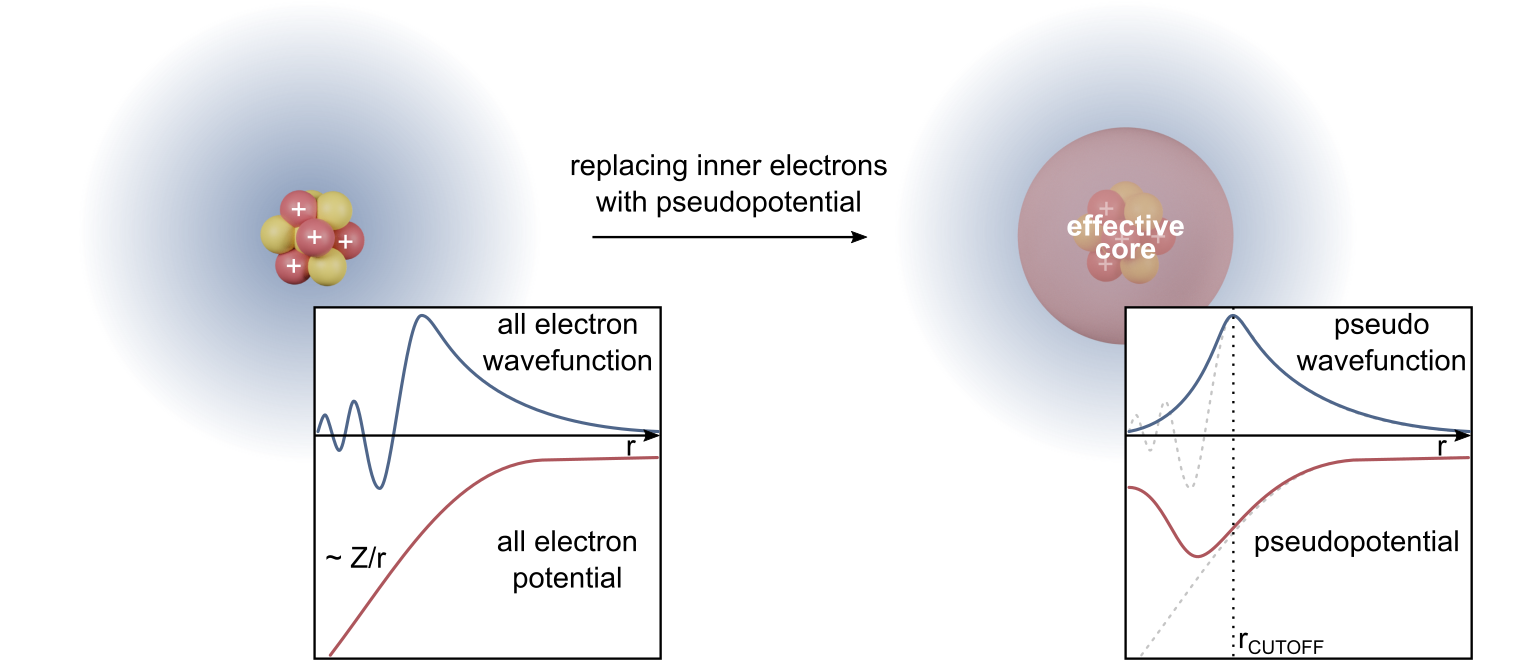
Density Functional Theory
Density functional theory (DFT) is a computational quantum mechanical modelling method used in physics, chemistry and materials science to investigate the electronic structure (or nuclear structure) (principally the ground state) of many-body systems, in particular atoms, molecules, and the condensed phases. Using this theory, the properties of a many-electron system can be determined by using functionals - that is, functions that accept a function as input and output a single real number. In the case of DFT, these are functionals of the spatially dependent electron density. DFT is among the most popular and versatile methods available in condensed-matter physics, computational physics, and computational chemistry. DFT has been very popular for calculations in solid-state physics since the 1970s. However, DFT was not considered accurate enough for calculations in quantum chemistry until the 1990s, when the approximations used in the theory were greatly refined to better m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coercivity
Coercivity, also called the magnetic coercivity, coercive field or coercive force, is a measure of the ability of a ferromagnetic material to withstand an external magnetic field without becoming Magnetization, demagnetized. Coercivity is usually measured in oersted or ampere/meter units and is denoted . An analogous property in electrical engineering and materials science, electric coercivity, is the ability of a ferroelectric material to withstand an external electric field without becoming polarization density, depolarized. Ferromagnetic materials with high coercivity are called magnetically ''hard'', and are used to make permanent magnets. Materials with low coercivity are said to be magnetically ''soft''. The latter are used in transformer and inductor magnetic core, cores, recording heads, microwave devices, and magnetic shielding. Definitions Coercivity in a ferromagnet, ferromagnetic material is the intensity of the applied magnetic field (''H'' field) required ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Magnetic Moment
In electromagnetism, the magnetic moment or magnetic dipole moment is the combination of strength and orientation of a magnet or other object or system that exerts a magnetic field. The magnetic dipole moment of an object determines the magnitude of torque the object experiences in a given magnetic field. When the same magnetic field is applied, objects with larger magnetic moments experience larger torques. The strength (and direction) of this torque depends not only on the magnitude of the magnetic moment but also on its orientation relative to the direction of the magnetic field. Its direction points from the south pole to the north pole of the magnet (i.e., inside the magnet). The magnetic moment also expresses the magnetic force effect of a magnet. The magnetic field of a magnetic dipole is proportional to its magnetic dipole moment. The dipole component of an object's magnetic field is symmetric about the direction of its magnetic dipole moment, and decreases as the inverse ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Remanence
Remanence or remanent magnetization or residual magnetism is the magnetization left behind in a ferromagnetic material (such as iron) after an external magnetic field is removed. Colloquially, when a magnet is "magnetized", it has remanence. The remanence of magnetic materials provides the magnetic memory in magnetic storage devices, and is used as a source of information on the past Earth's magnetic field in paleomagnetism. The word remanence is from remanent + -ence, meaning "that which remains". The equivalent term residual magnetization is generally used in engineering applications. In transformers, electric motors and generators a large residual magnetization is not desirable (see also electrical steel) as it is an unwanted contamination, for example, a magnetization remaining in an electromagnet after the current in the coil is turned off. Where it is unwanted, it can be removed by degaussing. Sometimes the term retentivity is used for remanence measured in units of m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Curie Temperature
In physics and materials science, the Curie temperature (''T''C), or Curie point, is the temperature above which certain materials lose their permanent magnetic properties, which can (in most cases) be replaced by induced magnetism. The Curie temperature is named after Pierre Curie, who showed that magnetism is lost at a critical temperature. The force of magnetism is determined by the magnetic moment, a dipole moment within an atom that originates from the angular momentum and spin of electrons. Materials have different structures of intrinsic magnetic moments that depend on temperature; the Curie temperature is the critical point at which a material's intrinsic magnetic moments change direction. Permanent magnetism is caused by the alignment of magnetic moments, and induced magnetism is created when disordered magnetic moments are forced to align in an applied magnetic field. For example, the ordered magnetic moments (ferromagnetic, Figure 1) change and become disordered ( ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ferromagnet
Ferromagnetism is a property of certain materials (such as iron) that results in a significant, observable magnetic permeability, and in many cases, a significant magnetic coercivity, allowing the material to form a permanent magnet. Ferromagnetic materials are noticeably attracted to a magnet, which is a consequence of their substantial magnetic permeability. Magnetic permeability describes the induced magnetization of a material due to the presence of an external magnetic field. For example, this temporary magnetization inside a steel plate accounts for the plate's attraction to a magnet. Whether or not that steel plate then acquires permanent magnetization depends on both the strength of the applied field and on the coercivity of that particular piece of steel (which varies with the steel's chemical composition and any heat treatment it may have undergone). In physics, multiple types of material magnetism have been distinguished. Ferromagnetism (along with the similar eff ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Crystallite
A crystallite is a small or even microscopic crystal which forms, for example, during the cooling of many materials. Crystallites are also referred to as grains. Bacillite is a type of crystallite. It is rodlike with parallel Wikt:longulite , longulites. Structure The orientation of crystallites can be random with no preferred direction, called random Texture (chemistry), texture, or directed, possibly due to growth and processing conditions. While the structure of a single crystal is highly ordered and its crystal lattice, lattice is continuous and unbroken, amorphous solid, amorphous materials, such as glass and many polymers, are non-crystalline and do not display any structures, as their constituents are not arranged in an ordered manner. Polycrystalline structures and paracrystalline phases are in between these two extremes. Polycrystalline materials, or polycrystals, are solids that are composed of many crystallites of varying size and orientation. Most materials are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
SQUID
A squid (: squid) is a mollusc with an elongated soft body, large eyes, eight cephalopod limb, arms, and two tentacles in the orders Myopsida, Oegopsida, and Bathyteuthida (though many other molluscs within the broader Neocoleoidea are also called ''squid'' despite not strictly fitting these criteria). Like all other cephalopods, squid have a distinct head, Symmetry (biology)#Bilateral symmetry, bilateral symmetry, and a mantle (mollusc), mantle. They are mainly soft-bodied, like octopuses, but have a small internal skeleton in the form of a rod-like gladius (cephalopod), gladius or pen, made of chitin. Squid diverged from other cephalopods during the Jurassic and occupy a similar Ecological niche, role to teleost fish as open-water predators of similar size and behaviour. They play an important role in the open-water food web. The two long tentacles are used to grab prey and the eight arms to hold and control it. The beak then cuts the food into suitable size chunks for swal ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |