|
Isotopes Of Yttrium
Natural yttrium (39Y) is composed of a single isotope, yttrium-89. The most stable radioisotopes are 88Y, which has a half-life of 106.6 days, and 91Y, with a half-life of 58.51 days. All the other isotopes have half-lives of less than a day, except 87Y, which has a half-life of 79.8 hours, and 90Y, with 64 hours. The dominant decay mode below the stable 89Y is electron capture and the dominant mode after it is beta emission. Thirty-five unstable isotopes have been characterized. 90Y exists in equilibrium with its parent isotope strontium-90, which is a product of nuclear fission. List of isotopes , -id=Yttrium-76 , rowspan=3, 76Y , rowspan=3 style="text-align:right" , 39 , rowspan=3 style="text-align:right" , 37 , rowspan=3, 75.95894(32)# , rowspan=3, 28(9) ms , β+? , 76Sr , rowspan=3, 1−# , rowspan=3, , - , p? , 75Sr , - , β+, p? , 75Rb , -id=Yttrium-77 , rowspan=3, 77Y , rowspan=3 style="text-align:right" , 39 , rowspan=3 style="text-align ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Yttrium
Yttrium is a chemical element; it has Symbol (chemistry), symbol Y and atomic number 39. It is a silvery-metallic transition metal chemically similar to the lanthanides and has often been classified as a "rare-earth element". Yttrium is almost always found in combination with lanthanide elements in rare-earth minerals and is never found in nature as a free element. 89Y is the only stable isotope and the only isotope found in the Crust (geology), Earth's crust. The most important present-day use of yttrium is as a component of phosphors, especially those used in LEDs. Historically, it was once widely used in the red phosphors in television set cathode ray tube displays. Yttrium is also used in the production of electrodes, electrolytes, electronic filters, lasers, superconductors, various medical applications, and Trace element, tracing various materials to enhance their properties. Yttrium has no known Biology, biological role. Exposure to yttrium compounds can cause Respiratory ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
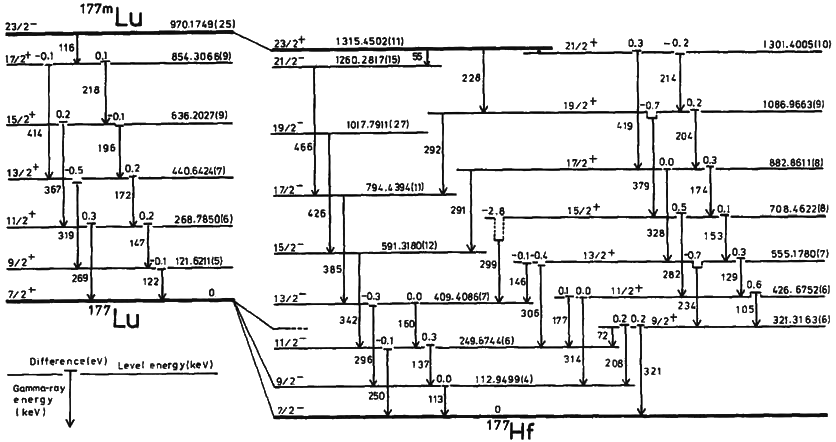
Isomeric Transition
A nuclear isomer is a metastable state of an atomic nucleus, in which one or more nucleons (protons or neutrons) occupy excited state levels (higher energy levels). "Metastable" describes nuclei whose excited states have half-lives of 10−9 seconds or longer, 100 to 1000 times longer than the half-lives of the excited nuclear states that decay with a "prompt" half life (ordinarily on the order of 10−12 seconds). Some references recommend seconds to distinguish the metastable half life from the normal "prompt" gamma-emission half-life. Occasionally the half-lives are far longer than this and can last minutes, hours, or years. For example, the nuclear isomer survives so long (at least years) that it has never been observed to decay spontaneously. The half-life of a nuclear isomer can even exceed that of the ground state of the same nuclide, as shown by as well as , , , , and multiple holmium isomers. Sometimes, the gamma decay from a metastable state is referred ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotopes Of Yttrium
Natural yttrium (39Y) is composed of a single isotope, yttrium-89. The most stable radioisotopes are 88Y, which has a half-life of 106.6 days, and 91Y, with a half-life of 58.51 days. All the other isotopes have half-lives of less than a day, except 87Y, which has a half-life of 79.8 hours, and 90Y, with 64 hours. The dominant decay mode below the stable 89Y is electron capture and the dominant mode after it is beta emission. Thirty-five unstable isotopes have been characterized. 90Y exists in equilibrium with its parent isotope strontium-90, which is a product of nuclear fission. List of isotopes , -id=Yttrium-76 , rowspan=3, 76Y , rowspan=3 style="text-align:right" , 39 , rowspan=3 style="text-align:right" , 37 , rowspan=3, 75.95894(32)# , rowspan=3, 28(9) ms , β+? , 76Sr , rowspan=3, 1−# , rowspan=3, , - , p? , 75Sr , - , β+, p? , 75Rb , -id=Yttrium-77 , rowspan=3, 77Y , rowspan=3 style="text-align:right" , 39 , rowspan=3 style="text-align ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Journal Of Scientific & Industrial Research
The National Institute of Science Communication and Information Resources (NISCAIR), located at New Delhi, India, was an information science institute in India founded in 2002. In 2021, the institute was merged with National Institute of Science, Technology and Development Studies to form National Institute of Science Communication and Policy Research (NIScPR). It operated under the umbrella of the Council of Scientific and Industrial Research (CSIR) that comprise 38 other labs and institutes in India. The institute published several academic journals and magazines. History In 2002, the Indian Scientific Documentation Centre (INSDOC), which came into being in 1952 under CSIR, was merged with NISCOM in 2002, to form National Institute of Science Communication and Information Resources (NISCAIR). General services National Science Library services Open access 18 journals and 3 popular science magazines (''Science Reporter'' and its Hindi and Urdu editions) published by CSIR ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotopes Of Rubidium
Rubidium (37Rb) has 36 isotopes, with naturally occurring rubidium being composed of just two isotopes; 85Rb (72.2%) and the radioactive 87Rb (27.8%). 87Rb has a half-life of . It readily substitutes for potassium in minerals, and is therefore fairly widespread. 87Rb has been used extensively in dating rocks; 87Rb decays to stable strontium-87 by emission of a beta particle (an electron ejected from the nucleus). During fractional crystallization, Sr tends to become concentrated in plagioclase, leaving Rb in the liquid phase. Hence, the Rb/Sr ratio in residual magma may increase over time, resulting in rocks with increasing Rb/Sr ratios with increasing differentiation. The highest ratios (10 or higher) occur in pegmatites. If the initial amount of Sr is known or can be extrapolated, the age can be determined by measurement of the Rb and Sr concentrations and the 87Sr/86Sr ratio. The dates indicate the true age of the minerals only if the rocks have not been subsequently altere ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotopes Of Strontium
The alkaline earth metal strontium (38Sr) has four stable, naturally occurring isotopes: 84Sr (0.56%), 86Sr (9.86%), 87Sr (7.0%) and 88Sr (82.58%). Its standard atomic weight is 87.62(1). Only 87Sr is radiogenic; it is produced by decay from the radioactive alkali metal 87 Rb, which has a half-life of 4.88 × 1010 years (i.e. more than three times longer than the current age of the universe). Thus, there are two sources of 87Sr in any material: primordial, formed during nucleosynthesis along with 84Sr, 86Sr and 88Sr; and that formed by radioactive decay of 87Rb. The ratio 87Sr/86Sr is the parameter typically reported in geologic investigations; ratios in minerals and rocks have values ranging from about 0.7 to greater than 4.0 (see rubidium–strontium dating). Because strontium has an electron configuration similar to that of calcium, it readily substitutes for calcium in minerals. In addition to the four stable isotopes, thirty-two unstable isotopes of strontium are k ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotopes Of Zirconium
Naturally occurring zirconium (40Zr) is composed of four stable isotopes (of which one may in the future be found radioactive), and one very long-lived radioisotope (96Zr), a primordial nuclide that decays via double beta decay with an observed half-life of 2.34 × 1019 years; it can also undergo single beta decay, which is not yet observed, but the theoretically predicted value of t1/2 is 2.4 × 1020 years. The second most stable radioisotope is 93Zr, which has a half-life of 1.61 million years. Thirty other radioisotopes have been observed. All have half-lives less than a day except for 95Zr (64.02 days), 88Zr (83.4 days), and 89Zr (78.41 hours). The primary decay mode is electron capture for isotopes lighter than 92Zr, and the primary mode for heavier isotopes is beta decay. List of isotopes , -id=Zirconium-77 , 77Zr , style="text-align:right" , 40 , style="text-align:right" , 37 , 76.96608(43)# , 100# μs , , , 3/2−# , , , -id=Zirconium-78 , 78Zr , ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neutron Emission
Neutron emission is a mode of radioactive decay in which one or more neutrons are ejected from a Atomic nucleus, nucleus. It occurs in the most neutron-rich/proton-deficient nuclides, and also from excited states of other nuclides as in photodisintegration, photoneutron emission and beta-delayed neutron emission. As only a neutron is lost by this process the number of protons remains unchanged, and an atom does not become an atom of a different element, but a different isotope of the same element. Neutrons are also produced in the spontaneous fission, spontaneous and nuclear fission, induced fission of certain heavy nuclides. Spontaneous neutron emission As a consequence of the Pauli exclusion principle, nuclei with an excess of protons or neutrons have a higher average energy per nucleon. Nuclei with a sufficient excess of neutrons have a greater energy than the combination of a free neutron and a nucleus with one less neutron, and therefore can decay by neutron emission. Nuclei ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Yttrium-90
Yttrium-90 () is a radioactive isotope of yttrium. Yttrium-90 has found a wide range of uses in radiation therapy to treat some forms of cancer Cancer is a group of diseases involving Cell growth#Disorders, abnormal cell growth with the potential to Invasion (cancer), invade or Metastasis, spread to other parts of the body. These contrast with benign tumors, which do not spread. Po .... Along with other isotopes of yttrium, it is sometimes called radioyttrium. Decay undergoes beta particles emissions/decay (beta decay, β− decay) to zirconium-90 with a half-life of 64.1 hours and a decay energy of 2.28 MeV with an average beta energy of 0.9336 MeV. It also produces 0.01% 1.7 MeV photons during its decay process to the 0+ state of 90Zr, followed by pair production. The interaction between emitted electrons and matter can lead to the emission of Bremsstrahlung radiation. Production Yttrium-90 is produced by the nuclear decay of strontium-90 which has a half-l ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fission Product
Nuclear fission products are the atomic fragments left after a large atomic nucleus undergoes nuclear fission. Typically, a large nucleus like that of uranium fissions by splitting into two smaller nuclei, along with a few neutrons, the release of heat energy (kinetic energy of the nuclei), and gamma rays. The two smaller nuclei are the ''fission products''. (See also Fission products (by element)). About 0.2% to 0.4% of fissions are ternary fissions, producing a third light nucleus such as helium-4 (90%) or tritium (7%). The fission products themselves are usually unstable and therefore radioactive. Due to being relatively neutron-rich for their atomic number, many of them quickly undergo beta decay. This releases additional energy in the form of beta particles, antineutrinos, and gamma rays. Thus, fission events normally result in beta and additional gamma radiation that begins immediately after, even though this radiation is not produced directly by the fission even ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Proton Emission
Proton emission (also known as proton radioactivity) is a rare type of radioactive decay in which a proton is ejected from a atomic nucleus, nucleus. Proton emission can occur from high-lying excited states in a nucleus following a beta decay, in which case the process is known as beta-delayed proton emission, or can occur from the ground state (or a low-lying nuclear isomer, isomer) of very proton-rich nuclei, in which case the process is very similar to alpha decay. For a proton to escape a nucleus, the proton separation energy must be negative (Sp < 0)—the proton is therefore unbound, and quantum tunneling, tunnels out of the nucleus in a finite time. The rate of proton emission is governed by the nuclear, Coulomb, and centrifugal potentials of the nucleus, where centrifugal potential affects a large part of the rate of proton emission. The half-life of a nucleus with respect to proton emission is affected by the proton energy and its orbital angular momentum. Proton emiss ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotope
Isotopes are distinct nuclear species (or ''nuclides'') of the same chemical element. They have the same atomic number (number of protons in their Atomic nucleus, nuclei) and position in the periodic table (and hence belong to the same chemical element), but different nucleon numbers (mass numbers) due to different numbers of neutrons in their nuclei. While all isotopes of a given element have similar chemical properties, they have different atomic masses and physical properties. The term isotope is derived from the Greek roots isos (wikt:ἴσος, ἴσος "equal") and topos (wikt:τόπος, τόπος "place"), meaning "the same place"; thus, the meaning behind the name is that different isotopes of a single element occupy the same position on the periodic table. It was coined by Scottish doctor and writer Margaret Todd (doctor), Margaret Todd in a 1913 suggestion to the British chemist Frederick Soddy, who popularized the term. The number of protons within the atomic nuc ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |