|
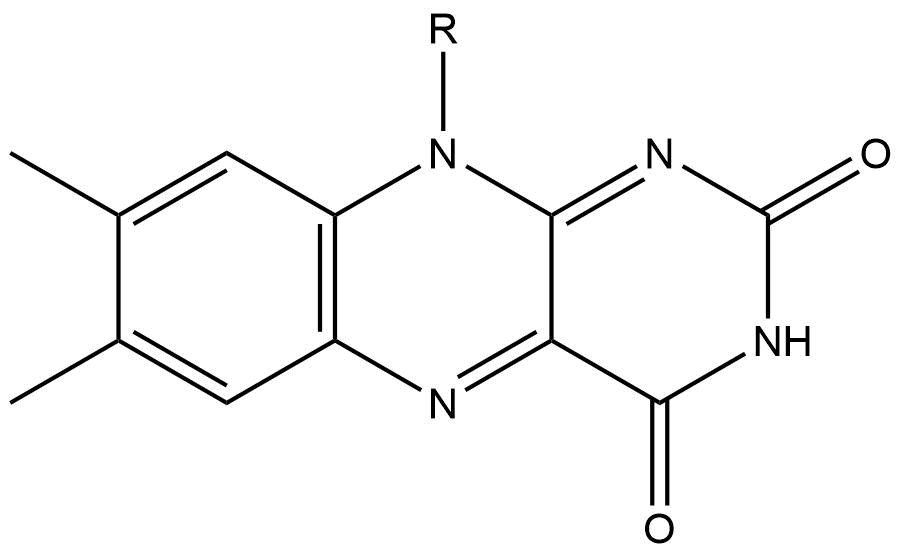
Isoalloxazine Structure
Isoalloxazine is the structural foundation of flavins such as riboflavin (vitamin B2) and is a heterocyclic compound. It has a tricyclic structure which means it has three interconnected rings of atoms and is a tautomer of alloxazine. The structure is formed by primary-secondary aromatic o-diamines and they are a high-melting crystalline substance. The R-group is used to attach various flavin groups It has a similar structure to pteridines which has two interconnected rings. Isoalloxazine was first obtained in 1934 by Richard Kuhn an Austrian-German biochemist and lab mates. Isoalloxazine ring Isoalloxazine rings can exist in different redox and ionization states depending on the chemistry of FMN and FAD associated with it. Using the redox-active isoalloxazine system, FAD and FMN are able to do one and two electron transfer reactions and also be coupled with proton A proton is a stable subatomic particle, symbol , H+, or 1H+ with a positive electric charge of +1 '' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Flavin Group
Flavins (from Latin ''flavus'', "yellow") are organic compounds, like their base, pteridine. They are formed by the tricyclic heterocycle isoalloxazine. The biochemical source is the vitamin riboflavin. The flavin moiety is often attached with an adenosine diphosphate to form flavin adenine dinucleotide (FAD), and, in other circumstances, is found as flavin mononucleotide (or FMN), a phosphorylated form of riboflavin. It is in one or the other of these forms that flavin is present as a prosthetic group in flavoproteins. The flavin group is capable of undergoing oxidation-reduction reactions, and can accept either one electron in a two-step process or two electrons at once. Reduction is made with the addition of hydrogen atoms to specific nitrogen atoms on the isoalloxazine ring system: In aqueous solution, flavins are yellow-coloured when oxidized, taking a red colour in the semi-reduced anionic state or blue in the neutral ( semiquinone) state, and colourless when totally ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Redox
Redox (reduction–oxidation, , ) is a type of chemical reaction in which the oxidation states of substrate (chemistry), substrate change. Oxidation is the loss of Electron, electrons or an increase in the oxidation state, while reduction is the gain of electrons or a decrease in the oxidation state. There are two classes of redox reactions: * ''Electron-transfer'' – Only one (usually) electron flows from the reducing agent to the oxidant. This type of redox reaction is often discussed in terms of redox couples and electrode potentials. * ''Atom transfer'' – An atom transfers from one substrate to another. For example, in the rusting of iron, the oxidation state of iron atoms increases as the iron converts to an oxide, and simultaneously the oxidation state of oxygen decreases as it accepts electrons released by the iron. Although oxidation reactions are commonly associated with the formation of oxides, other chemical species can serve the same function. In hydrogen ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Heterocyclic Compounds With 3 Rings
A heterocyclic compound or ring structure is a cyclic compound that has atoms of at least two different elements as members of its ring(s). Heterocyclic chemistry is the branch of organic chemistry dealing with the synthesis, properties, and applications of these heterocycles. Examples of heterocyclic compounds include all of the nucleic acids, the majority of drugs, most biomass (cellulose and related materials), and many natural and synthetic dyes. More than half of known compounds are heterocycles. 59% of US FDA-approved drugs contain nitrogen heterocycles. Classification The study of heterocyclic chemistry focuses especially on unsaturated derivatives, and the preponderance of work and applications involves unstrained 5- and 6-membered rings. Included are pyridine, thiophene, pyrrole, and furan. Another large class of heterocycles refers to those fused to benzene rings. For example, the fused benzene derivatives of pyridine, thiophene, pyrrole, and furan are quin ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ionization And Redox Of Isoalloxazine Ring
Ionization, or Ionisation is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons, often in conjunction with other chemical changes. The resulting electrically charged atom or molecule is called an ion. Ionization can result from the loss of an electron after collisions with subatomic particles, collisions with other atoms, molecules and ions, or through the interaction with electromagnetic radiation. Heterolytic bond cleavage and heterolytic substitution reactions can result in the formation of ion pairs. Ionization can occur through radioactive decay by the internal conversion process, in which an excited nucleus transfers its energy to one of the inner-shell electrons causing it to be ejected. Uses Everyday examples of gas ionization are such as within a fluorescent lamp or other electrical discharge lamps. It is also used in radiation detectors such as the Geiger-Müller counter or the ionization chamber. The ioni ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Proton
A proton is a stable subatomic particle, symbol , H+, or 1H+ with a positive electric charge of +1 ''e'' elementary charge. Its mass is slightly less than that of a neutron and 1,836 times the mass of an electron (the proton–electron mass ratio). Protons and neutrons, each with masses of approximately one atomic mass unit, are jointly referred to as "nucleons" (particles present in atomic nuclei). One or more protons are present in the Atomic nucleus, nucleus of every atom. They provide the attractive electrostatic central force which binds the atomic electrons. The number of protons in the nucleus is the defining property of an element, and is referred to as the atomic number (represented by the symbol ''Z''). Since each chemical element, element has a unique number of protons, each element has its own unique atomic number, which determines the number of atomic electrons and consequently the chemical characteristics of the element. The word ''proton'' is Greek language, G ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electron
The electron (, or in nuclear reactions) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. The electron's mass is approximately 1/1836 that of the proton. Quantum mechanical properties of the electron include an intrinsic angular momentum ( spin) of a half-integer value, expressed in units of the reduced Planck constant, . Being fermions, no two electrons can occupy the same quantum state, per the Pauli exclusion principle. Like all elementary particles, electrons exhibit properties of both particles and waves: They can collide with other particles and can be diffracted like light. The wave properties of electrons are easier to observe with experiments than those of other particles like neutrons and protons because electrons have a lower mass and hence a longer ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Flavin Adenine Dinucleotide
Flavin may refer to: Placename * Flavin, Aveyron, a commune in southern France Surname * Adrian Flavin (born 1979), a professional rugby player * Christopher Flavin, president of the Worldwatch Institute * Dan Flavin (1933–1996), a minimalist artist famous for using fluorescent light fixtures * Dan Flavin (politician), Louisiana politician * James Flavin (1906–1976), an American character actor * Jennifer Flavin (born 1968), a former model and wife of actor Sylvester Stallone * Martin Flavin (1883–1967), an American playwright and novelist * Martin Flavin (politician) (1841–1917), Irish Nationalist politician, Member of Parliament (MP) for Cork, 1891–1892 * Michael Joseph Flavin (1866-1944), Irish Nationalist politician, Member of Parliament (MP) for North Kerry, 1896-1918 * Mick Flavin Mick Flavin (born 3 August 1950) is an Irish country singer from Ballinamuck in County Longford. Flavin recorded his first album in Athlone in June 1986. His first big hit bei ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Flavin Mononucleotide
Flavin mononucleotide (FMN), or riboflavin-5′-phosphate, is a biomolecule produced from riboflavin (vitamin B2) by the enzyme riboflavin kinase and functions as the prosthetic group of various oxidoreductases, including NADH dehydrogenase, as well as cofactor in biological blue-light photo receptors. During the catalytic cycle, a reversible interconversion of the oxidized (FMN), semiquinone (FMNH•), and reduced (FMNH2) forms occurs in the various oxidoreductases. FMN is a stronger oxidizing agent than NAD and is particularly useful because it can take part in both one- and two-electron transfers. In its role as blue-light photo receptor, (oxidized) FMN stands out from the 'conventional' photo receptors as the signaling state and not an E/Z isomerization. It is the principal form in which riboflavin is found in cells and tissues. It requires more energy to produce, but is more soluble than riboflavin. In cells, FMN occurs freely circulating but also in several covalentl ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ionization
Ionization, or Ionisation is the process by which an atom or a molecule acquires a negative or positive Electric charge, charge by gaining or losing electrons, often in conjunction with other chemical changes. The resulting electrically charged atom or molecule is called an ion. Ionization can result from the loss of an electron after collisions with subatomic particles, collisions with other atoms, molecules and ions, or through the interaction with electromagnetic radiation. Heterolytic bond cleavage and heterolytic substitution reactions can result in the formation of ion pairs. Ionization can occur through radioactive decay by the internal conversion process, in which an excited nucleus transfers its energy to one of the inner-shell electrons causing it to be ejected. Uses Everyday examples of gas ionization are such as within a fluorescent lamp or other electrical discharge lamps. It is also used in radiation detectors such as the Geiger-Müller counter or the ionization cham ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isoalloxazine Structure
Isoalloxazine is the structural foundation of flavins such as riboflavin (vitamin B2) and is a heterocyclic compound. It has a tricyclic structure which means it has three interconnected rings of atoms and is a tautomer of alloxazine. The structure is formed by primary-secondary aromatic o-diamines and they are a high-melting crystalline substance. The R-group is used to attach various flavin groups It has a similar structure to pteridines which has two interconnected rings. Isoalloxazine was first obtained in 1934 by Richard Kuhn an Austrian-German biochemist and lab mates. Isoalloxazine ring Isoalloxazine rings can exist in different redox and ionization states depending on the chemistry of FMN and FAD associated with it. Using the redox-active isoalloxazine system, FAD and FMN are able to do one and two electron transfer reactions and also be coupled with proton A proton is a stable subatomic particle, symbol , H+, or 1H+ with a positive electric charge of +1 '' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Riboflavin
Riboflavin, also known as vitamin B2, is a vitamin found in food and sold as a dietary supplement. It is essential to the formation of two major coenzymes, flavin mononucleotide and flavin adenine dinucleotide. These coenzymes are involved in energy metabolism, cellular respiration, and antibody production, as well as normal growth and development. The coenzymes are also required for the metabolism of niacin, vitamin B6, and folate. Riboflavin is prescribed to treat corneal thinning, and taken orally, may reduce the incidence of migraine headaches in adults. Riboflavin deficiency is rare and is usually accompanied by deficiencies of other vitamins and nutrients. It may be prevented or treated by oral supplements or by injections. As a water-soluble vitamin, any riboflavin consumed in excess of nutritional requirements is not stored; it is either not absorbed or is absorbed and quickly excreted in urine, causing the urine to have a bright yellow tint. Natural sources of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Richard Kuhn
Richard Johann Kuhn (; 3 December 1900 – 1 August 1967) was an Austrian-German biochemist who was awarded the Nobel Prize in Chemistry in 1938 "for his work on carotenoids and vitamins". Biography Early life Kuhn was born in Vienna, Austria, where he attended grammar school and high school. His interest in chemistry surfaced early; however he had many interests and decided late to study chemistry. Between 1910 and 1918 he was a schoolmate of Wolfgang Pauli, who was awarded the Nobel Prize in Physics for 1945. Beginning in 1918, Kuhn attended lectures at the University of Vienna in chemistry. He finished his chemistry studies at University of Munich and received his doctoral degree in 1922 with Richard Willstätter for a scientific work on enzymes. After graduating, Kuhn continued his scientific career, first in Munich, then at the ETH Zurich and from 1929 onwards at the University of Heidelberg, where he was head of the chemistry department beginning in 1937. In 1928 he mar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |