|
Iron Triad
In chemistry and physics, the iron group refers to elements that are in some way related to iron; mostly in period (row) 4 of the periodic table. The term has different meanings in different contexts. In chemistry, the term is largely obsolete, but it often means iron, cobalt, and nickel, also called the iron triad;M. Green, ed. (2002): Organometallic Chemistry', volume 10, page 283. Royal Society of Chemistry; 430 pages, . It may sometimes refer to other elements that resemble iron in some chemical aspects, such as the stable group 8 elements (). In astrophysics and nuclear physics, the term is still quite common, and it typically means those three plus chromium and manganese—five elements that are exceptionally abundant, both on Earth and elsewhere in the universe, compared to their neighbors in the periodic table. Titanium and vanadium are also produced in Type Ia supernovae. General chemistry In chemistry, "iron group" used to refer to iron and the next two elements in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ruthenium
Ruthenium is a chemical element; it has symbol Ru and atomic number 44. It is a rare transition metal belonging to the platinum group of the periodic table. Like the other metals of the platinum group, ruthenium is unreactive to most chemicals. Karl Ernst Claus, a Russian scientist of Baltic-German ancestry, discovered the element in 1844 at Kazan State University and named it in honor of Russia. (He used the Latin name '' Ruthenia'', which can have other meanings, but specifically stated that the element was named in honor of his "motherland".) Ruthenium is usually found as a minor component of platinum ores; the annual production has risen from about 19 tonnes in 2009 to some 35.5 tonnes in 2017. Most ruthenium produced is used in wear-resistant electrical contacts and thick-film resistors. A minor application for ruthenium is in platinum alloys and as a chemical catalyst. A new application of ruthenium is as the capping layer for extreme ultraviolet photoma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Type Ia Supernova
A Type Ia supernova (read: "type one-A") is a type of supernova that occurs in binary systems (two stars orbiting one another) in which one of the stars is a white dwarf. The other star can be anything from a giant star to an even smaller white dwarf. Physically, carbon–oxygen white dwarfs with a low rate of rotation are limited to below 1.44 solar masses (). Beyond this "Chandrasekhar limit, critical mass", they reignite and in some cases trigger a supernova explosion; this critical mass is often referred to as the Chandrasekhar mass, but is marginally different from the absolute Chandrasekhar limit, where electron degeneracy pressure is unable to prevent catastrophic collapse. If a white dwarf gradually accretes mass from a binary companion, or merges with a second white dwarf, the general hypothesis is that a white dwarf's core will reach the ignition temperature for Carbon burning process, carbon fusion as it approaches the Chandrasekhar mass. Within a few seconds of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chloride
The term chloride refers to a compound or molecule that contains either a chlorine anion (), which is a negatively charged chlorine atom, or a non-charged chlorine atom covalently bonded to the rest of the molecule by a single bond (). The pronunciation of the word "chloride" is . Chloride salts such as sodium chloride are often soluble in water.Green, John, and Sadru Damji. "Chapter 3." ''Chemistry''. Camberwell, Vic.: IBID, 2001. Print. It is an essential electrolyte located in all body fluids responsible for maintaining acid/base balance, transmitting nerve impulses and regulating liquid flow in and out of cells. Other examples of ionic chlorides include potassium chloride (), calcium chloride (), and ammonium chloride (). Examples of covalent chlorides include methyl chloride (), carbon tetrachloride (), sulfuryl chloride (), and monochloramine (). Electronic properties A chloride ion (diameter 167 pm) is much larger than a chlorine atom (diameter 99 pm ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Faraday Lecture
The Faraday Lectureship Prize, previously known simply as the Faraday Lectureship, is awarded once every two years (approximately) by the Royal Society of Chemistry for "exceptional contributions to physical or theoretical chemistry".. Named after Michael Faraday, the first Faraday Lecture was given in 1869, two years after Faraday's death, by Jean-Baptiste Dumas.. As of 2009, the prize was worth £5000, with the recipient also receiving a medal and a certificate. As the name suggests, the recipient also gives a public lecture describing his or her work. Winners SourceRSC * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * See also * List of chemistry awards This list of chemistry awards is an index to articles about notable awards for chemistry. It includes awards by the Royal Society of Chemistry, the American Chemical Society, the Society of Chemical Industry and awards by other ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Atomic Weight
Relative atomic mass (symbol: ''A''; sometimes abbreviated RAM or r.a.m.), also known by the deprecated synonym atomic weight, is a dimensionless physical quantity defined as the ratio of the average mass of atoms of a chemical element in a given sample to the atomic mass constant. The atomic mass constant (symbol: ''m'') is defined as being of the mass of a carbon-12 atom. Since both quantities in the ratio are masses, the resulting value is dimensionless. These definitions remain valid even after the 2019 revision of the SI. For a single given sample, the relative atomic mass of a given element is the weighted arithmetic mean of the masses of the individual atoms (including all its isotopes) that are present in the sample. This quantity can vary significantly between samples because the sample's origin (and therefore its radioactive history or diffusion history) may have produced combinations of isotopic abundances in varying ratios. For example, due to a different mixture ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
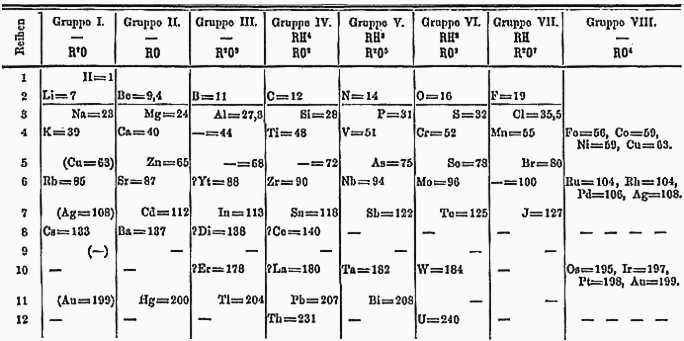
Dmitri Mendeleev
Dmitri Ivanovich Mendeleev ( ; ) was a Russian chemist known for formulating the periodic law and creating a version of the periodic table of elements. He used the periodic law not only to correct the then-accepted properties of some known elements, such as the valence and atomic weight of uranium, but also to predict the properties of three elements that were yet to be discovered (germanium, gallium and scandium). Early life Mendeleev was born in the village of Verkhnie Aremzyani, near Tobolsk in Siberia, to Ivan Pavlovich Mendeleev (1783–1847) and Maria Dmitrievna Mendeleeva (née Kornilieva) (1793–1850).''Maria Mendeleeva (1951)''. D. I. Mendeleev's Archive: Autobiographical Writings. Collection of Documents. Volume 1 /Biographical notes about D. I. Mendeleev (written by me – D. Mendeleev), p. 13 – Leningrad: D. I. Mendeleev's Museum-Archive, 207 pages (in Russian) Ivan worked as a school principal and a teacher of fine arts, politics and philosophy at the Tambov an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
John Alexander Reina Newlands
John Alexander Reina Newlands (26 November 1837 – 29 July 1898) was a British chemist who worked concerning the periodicity of elements. Biography Newlands was born in London in England, at West Square in Southwark, the son of a Scottish Presbyterian minister and his Italian wife. Newlands was home-schooled by his father, and later studied at the Royal College of Chemistry, now part of Imperial College London. He was interested in social reform and during 1860 served as a volunteer with Giuseppe Garibaldi in his military campaign to unify Italy. Returning to London, Newlands established himself as an analytical chemist in 1864. In 1868 he became chief chemist of James Duncan's London sugar refinery, where he introduced a number of improvements in processing. Later he quit the refinery and again became an analyst with his brother, Benjamin. Newlands was the first person to devise a periodic table of chemical elements arranged in order of their relative atomic masses publ ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Adolph Strecker
Adolph Strecker (21 October 1822 – 7 November 1871) was a German chemist who is known primarily for his work with amino acids. Life and work Strecker was born in Darmstadt, the son of Friedrich Ludwig Strecker, an archivist working for the hessian Grand Duke, and Henriette Amalie Johannette Koch. Adolph Strecker attended school in Darmstadt until 1838 when he changed to the higher Gewerbeschule. After receiving his abitur in 1840, Strecker began studying science at the University of Giessen, where Justus Liebig was a professor. In August 1842, Strecker received his PhD and began teaching at a realschule in Darmstadt. He refused one offer to work for Liebig, but in 1846 he accepted another and became Liebig's private assistant at the University of Giessen. Strecker finished his habilitation in 1848 and became a lecturer at the university. Strecker investigated a wide variety of problems in both organic and inorganic chemistry during his time at Giessen. Examples include the m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Döbereiner's Triads
In the history of the periodic table, Döbereiner's triads were an early attempt to sort the elements into some logical order and sets based on their physical properties. They are analogous to the groups (columns) on the modern periodic table. 53 elements were known at his time. In 1817, a letter by reported Johann Wolfgang Döbereiner Johann Wolfgang Döbereiner (13 December 1780 – 24 March 1849) was a German chemist who is known best for work that was suggestive of the periodic law for the chemical elements, and for inventing the first lighter, which was known as the Dö ...'s observations of the alkaline earths; namely, that strontium had properties that were intermediate to those of calcium and barium. By 1829, Döbereiner had found other groups of three elements (hence "triads") whose physical properties were similarly related. For an English translation of this article, see:Johann Wolfgang Döbereiner: "An Attempt to Group Elementary Substances according to The ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ferromagnetism
Ferromagnetism is a property of certain materials (such as iron) that results in a significant, observable magnetic permeability, and in many cases, a significant magnetic coercivity, allowing the material to form a permanent magnet. Ferromagnetic materials are noticeably attracted to a magnet, which is a consequence of their substantial magnetic permeability. Magnetic permeability describes the induced magnetization of a material due to the presence of an external magnetic field. For example, this temporary magnetization inside a steel plate accounts for the plate's attraction to a magnet. Whether or not that steel plate then acquires permanent magnetization depends on both the strength of the applied field and on the coercivity of that particular piece of steel (which varies with the steel's chemical composition and any heat treatment it may have undergone). In physics, multiple types of material magnetism have been distinguished. Ferromagnetism (along with the similar effec ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Alloys
An alloy is a mixture of chemical elements of which in most cases at least one is a metallic element, although it is also sometimes used for mixtures of elements; herein only metallic alloys are described. Metallic alloys often have properties that differ from those of the pure elements from which they are made. The vast majority of metals used for commercial purposes are alloyed to improve their properties or behavior, such as increased strength, hardness or corrosion resistance. Metals may also be alloyed to reduce their overall cost, for instance alloys of gold and copper. A typical example of an alloy is 304 grade stainless steel which is commonly used for kitchen utensils, pans, knives and forks. Sometime also known as 18/8, it as an alloy consisting broadly of 74% iron, 18% chromium and 8% nickel. The chromium and nickel alloying elements add strength and hardness to the majority iron element, but their main function is to make it resistant to rust/corrosion. In an al ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Platinum Group
The platinum-group metals (PGMs) are six noble, precious metallic elements clustered together in the periodic table. These elements are all transition metals in the d-block (groups 8, 9, and 10, periods 5 and 6). The six platinum-group metals are ruthenium, rhodium, palladium, osmium, iridium, and platinum. They have similar physical and chemical properties, and tend to occur together in the same mineral deposits. However, they can be further subdivided into the ''iridium-group platinum-group elements'' (IPGEs: Os, Ir, Ru) and the ''palladium-group platinum-group elements'' (PPGEs: Rh, Pt, Pd) based on their behaviour in geological systems. The three elements above the platinum group in the periodic table (iron, nickel and cobalt) are all ferromagnetic; these, together with the lanthanide element gadolinium (at temperatures below 20 °C), are the only known transition metals that display ferromagnetism near room temperature. History Naturally occurring plati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |