|
Hypercalcaemic Crisis
Hypercalcemia, also spelled hypercalcaemia, is a high calcium (Ca2+) level in the blood serum. The normal range for total calcium is 2.1–2.6 mmol/L (8.8–10.7 mg/dL, 4.3–5.2 mEq/L), with levels greater than 2.6 mmol/L defined as hypercalcemia. Those with a mild increase that has developed slowly typically have no symptoms. In those with greater levels or rapid onset, symptoms may include abdominal pain, bone pain, confusion, depression, weakness, kidney stones or an abnormal heart rhythm including cardiac arrest. Most outpatient cases are due to primary hyperparathyroidism and inpatient cases due to cancer. Other causes of hypercalcemia include sarcoidosis, tuberculosis, Paget disease, multiple endocrine neoplasia (MEN), vitamin D toxicity, familial hypocalciuric hypercalcaemia and certain medications such as lithium and hydrochlorothiazide. Diagnosis should generally include either a corrected calcium or ionized calcium level and be confirmed after a we ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
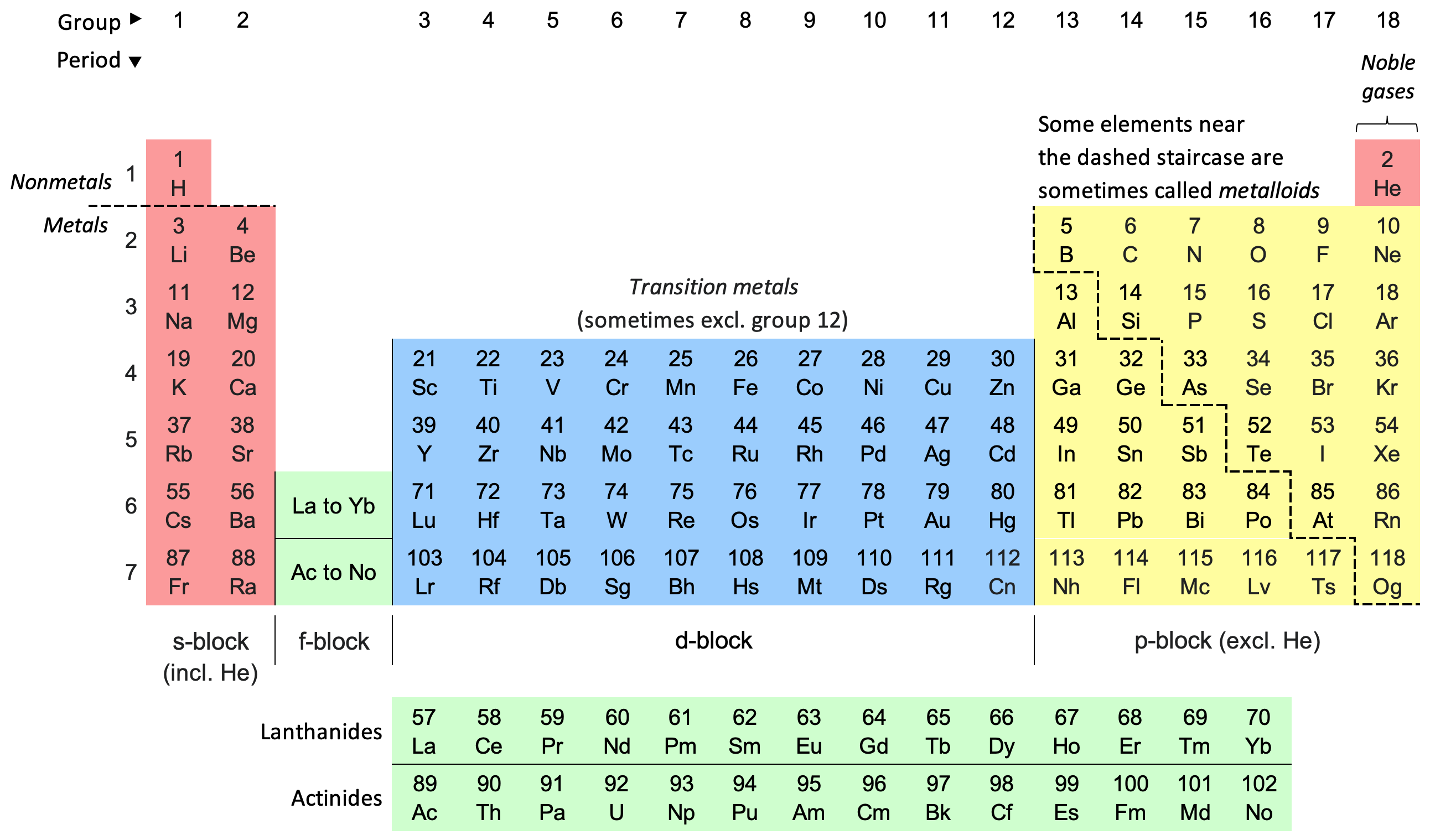
Periodic Table
The periodic table, also known as the periodic table of the elements, is an ordered arrangement of the chemical elements into rows (" periods") and columns (" groups"). It is an icon of chemistry and is widely used in physics and other sciences. It is a depiction of the periodic law, which states that when the elements are arranged in order of their atomic numbers an approximate recurrence of their properties is evident. The table is divided into four roughly rectangular areas called blocks. Elements in the same group tend to show similar chemical characteristics. Vertical, horizontal and diagonal trends characterize the periodic table. Metallic character increases going down a group and from right to left across a period. Nonmetallic character increases going from the bottom left of the periodic table to the top right. The first periodic table to become generally accepted was that of the Russian chemist Dmitri Mendeleev in 1869; he formulated the periodic law as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Corrected Calcium
Calcium ions (Ca2+) contribute to the physiology and biochemistry of organisms' cells. They play an important role in signal transduction pathways, where they act as a second messenger, in neurotransmitter release from neurons, in contraction of all muscle cell types, and in fertilization. Many enzymes require calcium ions as a cofactor, including several of the coagulation factors. Extracellular calcium is also important for maintaining the potential difference across excitable cell membranes, as well as proper bone formation. Plasma calcium levels in mammals are tightly regulated, electronic-book electronic- with bone acting as the major mineral storage site. Calcium ions, Ca2+, are released from bone into the bloodstream under controlled conditions. Calcium is transported through the bloodstream as dissolved ions or bound to proteins such as serum albumin. Parathyroid hormone secreted by the parathyroid gland regulates the resorption of Ca2+ from bone, reabsorption in the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Primary Hyperparathyroidism
Primary hyperparathyroidism (or PHPT) is a medical condition where the parathyroid gland (or a benign tumor within it) produce excess amounts of parathyroid hormone (PTH). The symptoms of the condition relate to the resulting elevated serum calcium (Hypercalcaemia, hypercalcemia), which can cause digestive symptoms, Nephrolithiasis, kidney stones, psychiatric abnormalities, and bone disease. The diagnosis is initially made on blood tests; an elevated level of calcium together with a raised (or inappropriately high) level of parathyroid hormone are typically found. To identify the source of the excessive hormone secretion, medical imaging may be performed. Parathyroidectomy, the surgical removal of one or more parathyroid glands, may be required to control symptoms. Signs and symptoms The signs and symptoms of primary hyperparathyroidism are those of hypercalcaemia, hypercalcemia. They are classically summarized by "stones, bones, abdominal groans, thrones and psychiatric overtone ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Kidney Stones
Kidney stone disease (known as nephrolithiasis, renal calculus disease, or urolithiasis) is a crystallopathy and occurs when there are too many minerals in the urine and not enough liquid or hydration. This imbalance causes tiny pieces of crystal to aggregate and form hard masses, or calculi (stones) in the upper urinary tract. Because renal calculi typically form in the kidney, if small enough, they are able to leave the urinary tract via the urine stream. A small calculus may pass without causing symptoms. However, if a stone grows to more than , it can cause blockage of the ureter, resulting in extremely sharp and severe pain ( renal colic) in the lower back that often radiates downward to the groin. A calculus may also result in blood in the urine, vomiting (due to severe pain), or painful urination. About half of all people who have had a kidney stone are likely to develop another within ten years. ''Renal'' is Latin for "kidney", while "nephro" is the Greek equival ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Abdominal Pain
Abdominal pain, also known as a stomach ache, is a symptom associated with both non-serious and serious medical issues. Since the abdomen contains most of the body's vital organs, it can be an indicator of a wide variety of diseases. Given that, approaching the examination of a person and planning of a differential diagnosis is extremely important. Common causes of pain in the abdomen include gastroenteritis and irritable bowel syndrome. About 15% of people have a more serious underlying condition such as appendicitis, leaking or ruptured abdominal aortic aneurysm, diverticulitis, or ectopic pregnancy. In a third of cases, the exact cause is unclear. Signs and symptoms The onset of abdominal pain can be abrupt, quick, or gradual. Sudden onset pain happens in a split second. Rapidly onset pain starts mild and gets worse over the next few minutes. Pain that gradually intensifies only after several hours or even days has passed is referred to as gradual onset pain. One can ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
MEq/L
An equivalent (symbol: officially equiv; unofficially but often Eq) is the amount of a substance that reacts with (or is ''equivalent'' to) an arbitrary amount (typically one mole) of another substance in a given chemical reaction. It is an archaic quantity that was used in chemistry and the biological sciences (see '). The mass of an equivalent is called its equivalent weight. Formula The formula from milligrams (mg) to milli-equivalent (mEq) and back is as follows: \begin \text \to \text &: \quad \text \times \frac \\ pt\text \to \text &: \quad \text \times \frac \end where is the valence and is the molecular weight. For elemental compounds: \text \to \text : \quad \frac \times \frac Common examples mEq to milligram Milligram to mEq Formal definition In a more formal definition, the ''equivalent'' is the amount of a substance needed to do one of the following: * react with or supply one mole of hydrogen ions () in an acid–base reaction * react with or supply one ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mmol/L
Molar concentration (also called molarity, amount concentration or substance concentration) is the number of moles of solute per liter of solution. Specifically, It is a measure of the concentration of a chemical species, in particular, of a solute in a solution, in terms of amount of substance per unit volume of solution. In chemistry, the most commonly used unit for molarity is the number of moles per liter, having the unit symbol mol/L or mol/ dm3 (1000 mol/ m3) in SI units. A solution with a concentration of 1 mol/L is said to be 1 molar, commonly designated as 1 M or 1 M. Molarity is often depicted with square brackets around the substance of interest; for example, the molarity of the hydrogen ion is depicted as + Definition Molar concentration or molarity is most commonly expressed in units of moles of solute per litre of solution. For use in broader applications, it is defined as amount of substance of solute per unit volume of solution, or p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Blood Serum
Serum () is the fluid and solvent component of blood which does not play a role in clotting. It may be defined as blood plasma without the clotting factors, or as blood with all cells and clotting factors removed. Serum contains all proteins except clotting factors (involved in blood clotting), including all electrolytes, antibodies, antigens, hormones; and any exogenous substances (e.g., drugs, microorganisms). Serum also does not contain all the formed elements of blood, which include blood cells, white blood cells (leukocytes, lymphocytes), red blood cells ( erythrocytes), and platelets. The study of serum is serology. Serum is used in numerous diagnostic tests as well as blood typing. Measuring the concentration of various molecules can be useful for many applications, such as determining the therapeutic index of a drug candidate in a clinical trial. To obtain serum, a blood sample is allowed to clot (coagulation). The sample is then centrifuged to remove the clot and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcium
Calcium is a chemical element; it has symbol Ca and atomic number 20. As an alkaline earth metal, calcium is a reactive metal that forms a dark oxide-nitride layer when exposed to air. Its physical and chemical properties are most similar to its heavier homologues strontium and barium. It is the fifth most abundant element in Earth's crust, and the third most abundant metal, after iron and aluminium. The most common calcium compound on Earth is calcium carbonate, found in limestone and the fossils of early sea life; gypsum, anhydrite, fluorite, and apatite are also sources of calcium. The name comes from Latin ''calx'' " lime", which was obtained from heating limestone. Some calcium compounds were known to the ancients, though their chemistry was unknown until the seventeenth century. Pure calcium was isolated in 1808 via electrolysis of its oxide by Humphry Davy, who named the element. Calcium compounds are widely used in many industries: in foods and pharmaceuticals for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hemodialysis
Hemodialysis, American and British English spelling differences#ae and oe, also spelled haemodialysis, or simply ''"'dialysis'"'', is a process of filtering the blood of a person whose kidneys are not working normally. This type of Kidney dialysis, dialysis achieves the extracorporeal removal of waste products such as creatinine and urea and free water from the blood when the kidneys are in a state of kidney failure. Hemodialysis is one of three renal replacement therapy, renal replacement therapies (the other two being kidney transplant and peritoneal dialysis). An alternative method for extracorporeal separation of blood components such as plasma or cells is apheresis. Hemodialysis can be an outpatient or inpatient therapy. Routine hemodialysis is conducted in a dialysis outpatient facility, either a purpose-built room in a hospital or a dedicated, stand-alone clinic. Less frequently hemodialysis is done at home hemodialysis, home. Dialysis treatments in a clinic are initiated ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pamidronate
Pamidronic acid or pamidronate disodium or APD (marketed as Aredia among others), is a nitrogen-containing bisphosphonate used to prevent osteoporosis. It was patented in 1971 and approved for medical use in 1987. Medical uses It is used to prevent bone loss, and treat osteoporosis. It is also used to strengthen bone in Paget's disease, to prevent bone loss due to steroid use, and in certain cancers with high propensity to bone, such as multiple myeloma. Due to its ability to sequester calcium in bone, it is also used to treat high calcium levels. It is also used as an experimental treatment of the bone disorder osteogenesis imperfecta. It has been studied in the treatment of complex regional pain syndrome. Administration Intravenous, usually 90 mg monthly. 30 mg, 60 mg, 90 mg and for hospitals, 120 mg vials are available, mixed with mannitol. Side effects Common side effects include bone pain, low calcium levels, nausea, and dizziness. Osteone ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Calcitonin
Calcitonin is a 32 amino acid peptide hormone secreted by parafollicular cells (also known as C cells) of the thyroid (or endostyle) in humans and other chordates in the ultimopharyngeal body. It acts to reduce blood calcium (Ca2+), opposing the effects of parathyroid hormone (PTH). Its importance in humans has not been as well established as its importance in other animals, as its function is usually not significant in the regulation of normal Calcium metabolism, calcium homeostasis. It belongs to the calcitonin-like protein family. Historically calcitonin has also been called thyrocalcitonin. Biosynthesis and regulation Calcitonin is formed by the proteolytic cleavage of a larger prepropeptide, which is the product of the CALC1 gene (). It is functionally an antagonist with PTH and Vitamin D3. The CALC1 gene belongs to a superfamily of related protein hormone precursors including islet amyloid precursor protein, calcitonin gene-related peptide, and the precursor of adrenomedul ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |