|
Energy Poverty And Cooking
One aspect of energy poverty is lack of access to clean, modern fuels and technologies for cooking. As of 2020, more than 2.6 billion people in developing countries routinely cook with fuels such as wood, animal dung, coal, or kerosene. Burning these types of fuels in open fires or traditional stoves causes harmful household air pollution, resulting in an estimated 3.8 million deaths annually according to the World Health Organization (WHO), and contributes to various health, socio-economic, and environmental problems. A high priority in global sustainable development is making clean cooking facilities universally available and affordable. Stoves and appliances that run on electricity, liquid petroleum gas (LPG), piped natural gas (PNG), biogas, alcohol, and solar heat meet WHO guidelines for clean cooking. Universal access to clean cooking facilities would benefit the environment and gender equality greatly. Stoves that burn wood and other solid fuels more efficiently than trad ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Global Burden Of Disease Study
The Global Burden of Disease Study (GBD) is a comprehensive regional and global research program of disease burden that assesses mortality and disability from major diseases, injuries, and risk factors. GBD is a collaboration of over 12,000 researchers from more than 160 countries. Under principal investigator Christopher J.L. Murray, GBD is based in the Institute for Health Metrics and Evaluation (IHME) at the University of Washington and funded by the Bill and Melinda Gates Foundation. History The Global Burden of Disease Study began in 1990 as a single World Bank–commissioned study that quantified the health effects of more than 100 diseases and injuries for eight regions of the world, giving estimates of morbidity and mortality by age, sex, and region. It also introduced the disability-adjusted life year (DALY) as a new metric to quantify the burden of diseases, injuries, and risk factors, to aid comparisons. GBD 1990 was "institutionalized" at the World Health Organizati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Burn
A burn is an injury to skin, or other tissues, caused by heat, electricity, chemicals, friction, or ionizing radiation (such as sunburn, caused by ultraviolet radiation). Most burns are due to heat from hot fluids (called scalding), solids, or fire. Burns occur mainly in the home or the workplace. In the home, risks are associated with domestic kitchens, including stoves, flames, and hot liquids. In the workplace, risks are associated with fire and chemical and electric burns. Alcoholism and smoking are other risk factors. Burns can also occur as a result of self-harm or violence between people (assault). Burns that affect only the superficial skin layers are known as superficial or first-degree burns. They appear red without blisters, and pain typically lasts around three days. When the injury extends into some of the underlying skin layer, it is a partial-thickness or second-degree burn. Blisters are frequently present and they are often very painful. Healing can re ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
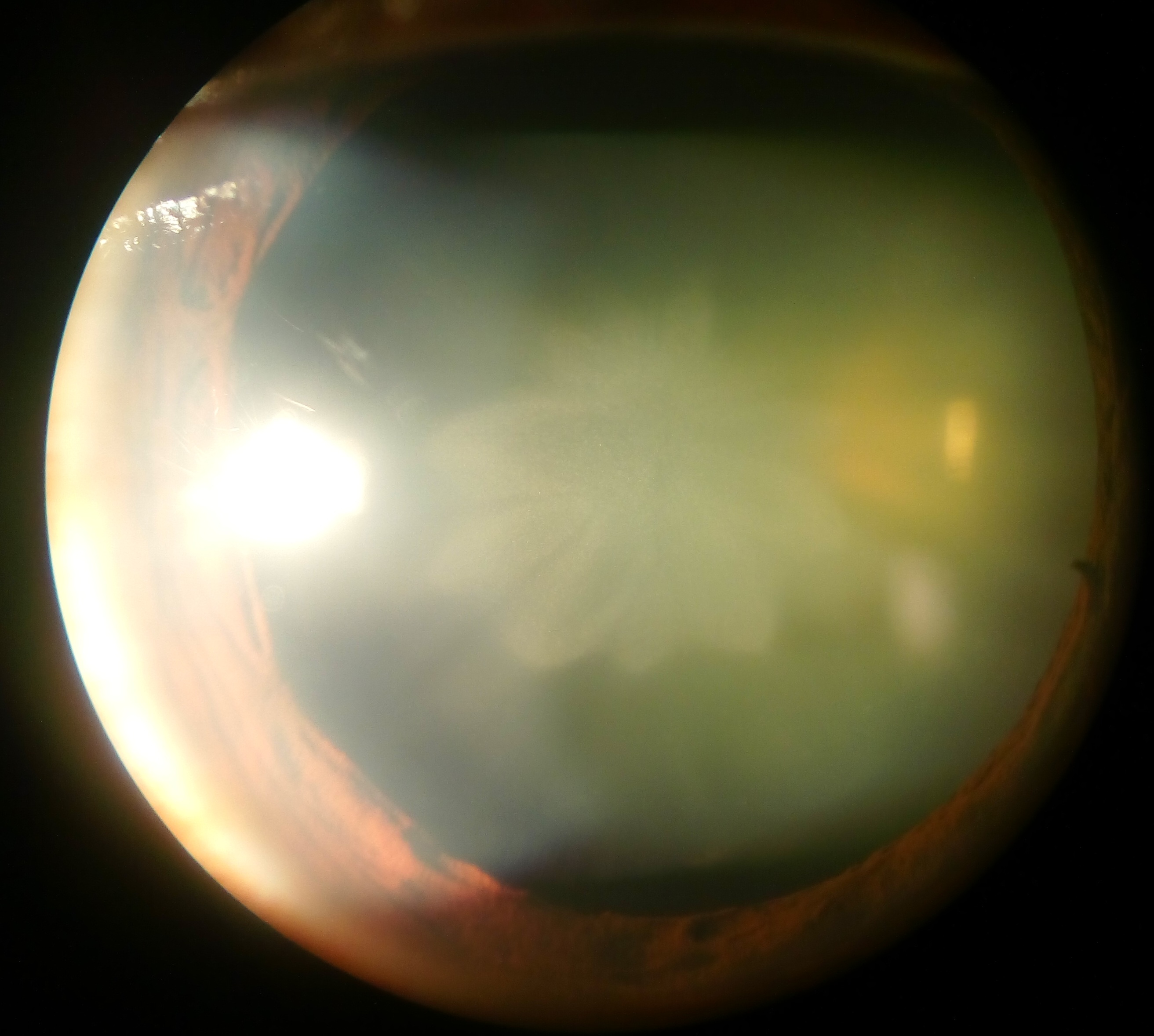
Cataracts
A cataract is a cloudy area in the lens of the eye that leads to a decrease in vision of the eye. Cataracts often develop slowly and can affect one or both eyes. Symptoms may include faded colours, blurry or double vision, halos around light, trouble with bright lights, and difficulty seeing at night. This may result in trouble driving, reading, or recognizing faces. Poor vision caused by cataracts may also result in an increased risk of falling and depression. Cataracts cause 51% of all cases of blindness and 33% of visual impairment worldwide. Cataracts are most commonly due to aging but may also occur due to trauma or radiation exposure, be present from birth, or occur following eye surgery for other problems. Risk factors include diabetes, longstanding use of corticosteroid medication, smoking tobacco, prolonged exposure to sunlight, and alcohol. In addition to these, poor nutrition, obesity, chronic kidney disease, and autoimmune diseases have been recognized in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pneumonia
Pneumonia is an Inflammation, inflammatory condition of the lung primarily affecting the small air sacs known as Pulmonary alveolus, alveoli. Symptoms typically include some combination of Cough#Classification, productive or dry cough, chest pain, fever, and Shortness of breath, difficulty breathing. The severity of the condition is variable. Pneumonia is usually caused by infection with viruses or bacteria, and less commonly by other microorganisms. Identifying the responsible pathogen can be difficult. Diagnosis is often based on symptoms and physical examination. Chest X-rays, blood tests, and Microbiological culture, culture of the sputum may help confirm the diagnosis. The disease may be classified by where it was acquired, such as community- or hospital-acquired or healthcare-associated pneumonia. Risk factors for pneumonia include cystic fibrosis, chronic obstructive pulmonary disease (COPD), sickle cell disease, asthma, diabetes, heart failure, a history of smoking, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzo(a)pyrene
Benzo 'a''yrene (B''a''P or B ) is a polycyclic aromatic hydrocarbon and the result of incomplete combustion of organic matter at temperatures between and . The ubiquitous compound can be found in coal tar, tobacco smoke and many foods, especially grilled meats. The substance with the formula C20H12 is one of the benzopyrenes, formed by a benzene ring fused to pyrene. Its diol epoxide metabolites, more commonly known as BPDE, react with and bind to DNA, resulting in mutations and eventually cancer. It is listed as a Group 1 carcinogen by the IARC. In the 18th century a scrotal cancer of chimney sweepers, the chimney sweeps' carcinoma, was already known to be connected to soot. Description Benzo 'a''yrene (B''a''P) is a polycyclic aromatic hydrocarbon found in coal tar with the formula C20H12. The compound is one of the benzopyrenes, formed by a benzene ring fused to pyrene, and is the result of incomplete combustion at temperatures between and . Sources The ma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polycyclic Aromatic Hydrocarbon
A Polycyclic aromatic hydrocarbon (PAH) is any member of a class of organic compounds that is composed of multiple fused aromatic rings. Most are produced by the incomplete combustion of organic matter— by engine exhaust fumes, tobacco, incinerators, in roasted meats and cereals, or when biomass burns at lower temperatures as in forest fires. The simplest representative is naphthalene, having two aromatic rings, and the three-ring compounds anthracene and phenanthrene. PAHs are uncharged, non-polar and planar. Many are colorless. Many of them are also found in fossil fuel deposits such as coal and in petroleum. Exposure to PAHs can lead to different types of cancer, to fetal development complications, and to cardiovascular issues. Polycyclic aromatic hydrocarbons are discussed as possible starting materials for abiotic syntheses of materials required by the earliest forms of life. Nomenclature and structure The terms polyaromatic hydrocarbon, or polynuclear aromatic hydro ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1,3-Butadiene
1,3-Butadiene () is the organic compound with the formula CH2=CH-CH=CH2. It is a colorless gas that is easily condensed to a liquid. It is important industrially as a precursor to synthetic rubber. The molecule can be viewed as the union of two vinyl groups. It is the simplest conjugated diene. Although butadiene breaks down quickly in the atmosphere, it is nevertheless found in ambient air in urban and suburban areas as a consequence of its constant emission from motor vehicles. The name butadiene can also refer to the isomer, 1,2-butadiene, which is a cumulated diene with structure H2C=C=CH−CH3. This allene has no industrial significance. History In 1863, French chemist E. Caventou isolated butadiene from the pyrolysis of amyl alcohol. This hydrocarbon was identified as butadiene in 1886, after Henry Edward Armstrong isolated it from among the pyrolysis products of petroleum. In 1910, the Russian chemist Sergei Lebedev polymerized butadiene and obtained a material w ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Benzene
Benzene is an Organic compound, organic chemical compound with the Chemical formula#Molecular formula, molecular formula C6H6. The benzene molecule is composed of six carbon atoms joined in a planar hexagonal Ring (chemistry), ring with one hydrogen atom attached to each. Because it contains only carbon and hydrogen atoms, benzene is classed as a hydrocarbon. Benzene is a natural constituent of petroleum and is one of the elementary petrochemicals. Due to the cyclic continuous pi bonds between the carbon atoms, benzene is classed as an aromatic hydrocarbon. Benzene is a colorless and highly Combustibility and flammability, flammable liquid with a sweet smell, and is partially responsible for the aroma of gasoline. It is used primarily as a Precursor (chemistry), precursor to the manufacture of chemicals with more complex structures, such as ethylbenzene and cumene, of which billions of kilograms are produced annually. Although benzene is a major Chemical industry, industrial che ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Formaldehyde
Formaldehyde ( , ) (systematic name methanal) is an organic compound with the chemical formula and structure , more precisely . The compound is a pungent, colourless gas that polymerises spontaneously into paraformaldehyde. It is stored as aqueous solutions (formalin), which consists mainly of the hydrate CH2(OH)2. It is the simplest of the aldehydes (). As a precursor to many other materials and chemical compounds, in 2006 the global production of formaldehyde was estimated at 12 million tons per year. It is mainly used in the production of industrial resins, e.g., for particle board and coatings. Formaldehyde also occurs naturally. It is derived from the degradation of serine, dimethylglycine, and lipids. Demethylases act by converting N-methyl groups to formaldehyde. Formaldehyde is classified as a group 1 carcinogen and can cause respiratory and skin irritation upon exposure. Forms Formaldehyde is more complicated than many simple carbon compounds in that i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Volatile Organic Compound
Volatile organic compounds (VOCs) are organic compounds that have a high vapor pressure at room temperature. They are common and exist in a variety of settings and products, not limited to Indoor mold, house mold, Upholstery, upholstered furniture, Handicraft, arts and crafts supplies, Dry cleaning, dry cleaned clothing, and Cleaning agent, cleaning supplies. VOCs are responsible for the odor of scents and perfumes as well as pollutants. They play an important role in communication between animals and plants, such as attractants for pollinators, protection from predation, and even inter-plant interactions. Some VOCs are dangerous to human health or cause harm to the natural environment, environment, often despite the odor being perceived as pleasant, such as "new car smell". Human impact on the environment, Anthropogenic VOCs are regulated by law, especially indoors, where concentrations are the highest. Most VOCs are not acutely toxic, but may have long-term chronic health effect ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Sulfur Oxide
Sulfur oxide Sulfur oxides (SOx) are a group of chemical compounds formed by the combination of sulfur and oxygen. The most common SOx are sulfur dioxide (SO2) and sulfur trioxide (SO3). SOx are produced naturally through volcanic activity and are also released into the atmosphere from human activities like burning fossil fuels and industrial processes. Sulfur oxide (SO''x'') refers to one or more of the following: * Lower sulfur oxides (S''n''O, S7O2 and S6O2) * Sulfur monoxide (SO) and its dimer, Disulfur dioxide (S2O2) * Sulfur dioxide Sulfur dioxide (IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula . It is a colorless gas with a pungent smell that is responsible for the odor of burnt matches. It is r ... (SO2) * Sulfur trioxide (SO3) * Higher sulfur oxides (SO3 and SO4 and polymeric condensates of them) * Disulfur monoxide (S2O) {{Authority control * ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |