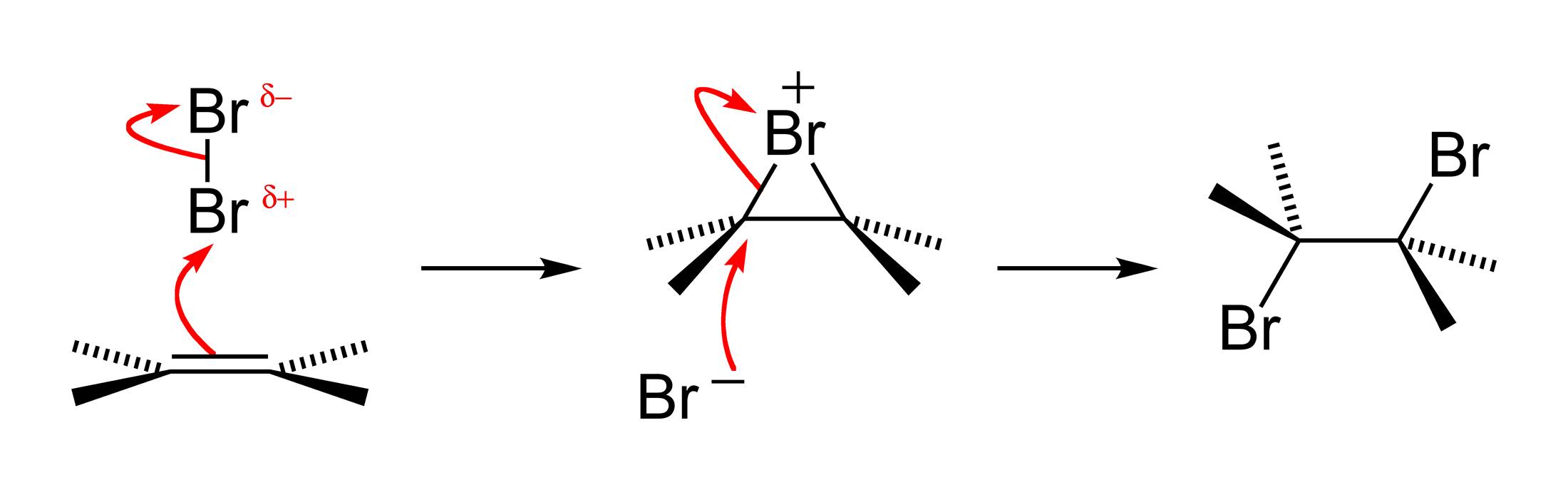|
Electrophilic Synthesis
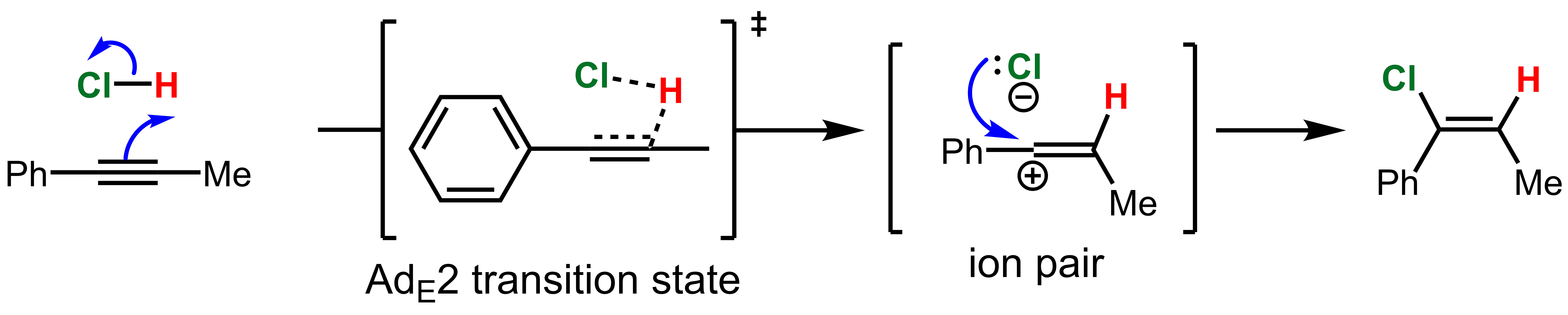
In chemistry, an electrophile is a chemical species that forms bonds with nucleophiles by accepting an electron pair. Because electrophiles accept electrons, they are Lewis acids. Most electrophiles are positively charged, have an atom that carries a partial positive charge, or have an atom that does not have an octet of electrons. Electrophiles mainly interact with nucleophiles through addition and substitution reactions. Frequently seen electrophiles in organic syntheses include cations such as H+ and NO+, polarized neutral molecules such as HCl, alkyl halides, acyl halides, and carbonyl compounds, polarizable neutral molecules such as Cl2 and Br2, oxidizing agents such as organic peracids, chemical species that do not satisfy the octet rule such as carbenes and radicals, and some Lewis acids such as BH3 and DIBAL. Organic chemistry Addition of halogens These occur between alkenes and electrophiles, often halogens as in halogen addition reactions. Common reactions ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemistry
Chemistry is the scientific study of the properties and behavior of matter. It is a physical science within the natural sciences that studies the chemical elements that make up matter and chemical compound, compounds made of atoms, molecules and ions: their composition, structure, properties, behavior and the changes they undergo during chemical reaction, reactions with other chemical substance, substances. Chemistry also addresses the nature of chemical bonds in chemical compounds. In the scope of its subject, chemistry occupies an intermediate position between physics and biology. It is sometimes called the central science because it provides a foundation for understanding both Basic research, basic and Applied science, applied scientific disciplines at a fundamental level. For example, chemistry explains aspects of plant growth (botany), the formation of igneous rocks (geology), how atmospheric ozone is formed and how environmental pollutants are degraded (ecology), the prop ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bromine
Bromine is a chemical element; it has chemical symbol, symbol Br and atomic number 35. It is a volatile red-brown liquid at room temperature that evaporates readily to form a similarly coloured vapour. Its properties are intermediate between those of chlorine and iodine. Isolated independently by two chemists, Carl Jacob Löwig (in 1825) and Antoine Jérôme Balard (in 1826), its name was derived , referring to its sharp and pungent smell. Elemental bromine is very reactive and thus does not occur as a free element in nature. Instead, it can be isolated from colourless soluble crystalline mineral halide Ionic salt, salts analogous to table salt, a property it shares with the other halogens. While it is rather rare in the Earth's crust, the high solubility of the bromide ion (Br) has caused its Bromine cycle, accumulation in the oceans. Commercially the element is easily extracted from brine evaporation ponds, mostly in the United States and Israel. The mass of bromine in the oce ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrophilic Addition Of Br2
In chemistry, an electrophile is a chemical species that forms bonds with nucleophiles by accepting an electron pair. Because electrophiles accept electrons, they are Lewis acids. Most electrophiles are positively Electric charge, charged, have an atom that carries a partial positive charge, or have an atom that does not have an octet of electrons. Electrophiles mainly interact with nucleophiles through Addition reaction, addition and Substitution reaction, substitution reactions. Frequently seen electrophiles in Organic synthesis, organic syntheses include cations such as Hydrogen ion, H+ and nitrosonium, NO+, polarized neutral molecules such as hydrogen chloride, HCl, alkyl halides, acyl halides, and carbonyl compounds, polarizable neutral molecules such as chlorine, Cl2 and bromine, Br2, oxidizing agents such as organic peracids, chemical species that do not satisfy the octet rule such as carbenes and Radical (chemistry), radicals, and some Lewis acids such as Borane, BH3 and Di ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
A European Journal
A, or a, is the first letter and the first vowel letter of the Latin alphabet, used in the modern English alphabet, and others worldwide. Its name in English is '' a'' (pronounced ), plural ''aes''. It is similar in shape to the Ancient Greek letter alpha, from which it derives. The uppercase version consists of the two slanting sides of a triangle, crossed in the middle by a horizontal bar. The lowercase version is often written in one of two forms: the double-storey and single-storey . The latter is commonly used in handwriting and fonts based on it, especially fonts intended to be read by children, and is also found in italic type. In English, '' a'' is the indefinite article, with the alternative form ''an''. Name In English, the name of the letter is the ''long A'' sound, pronounced . Its name in most other languages matches the letter's pronunciation in open syllables. History The earliest known ancestor of A is ''aleph''—the first letter of the Phoenician ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
1,2-Dibromoethane
1,2-Dibromoethane, also known as ethylene dibromide (EDB), is an organobromine compound with the chemical formula . Although trace amounts occur naturally in the ocean, where it is probably formed by algae and kelp, substantial amounts are produced industrially. It is a dense colorless liquid with a faint, sweet odor, detectable at 10 ppm. It is a widely used and sometimes-controversial fumigant. The combustion of 1,2-dibromoethane produces hydrogen bromide gas that is significantly corrosive. Preparation and use It is produced by the reaction of ethylene gas with bromine, in a classic halogen addition reaction: :CH=CH + Br → BrCH–CHBr Historically, 1,2-dibromoethane was used as a component in anti-knock additives in leaded fuels. It reacts with lead residues to generate volatile lead bromides, thereby preventing fouling of the engine with lead deposits. Pesticide It has been used as a pesticide in soil and on various crops. The applications were initiated after the fo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ethene
Ethylene (IUPAC name: ethene) is a hydrocarbon which has the formula or . It is a colourless, flammable gas with a faint "sweet and musky" odour when pure. It is the simplest alkene (a hydrocarbon with carbon–carbon double bonds). Ethylene is widely used in the chemical industry, and its worldwide production (over 150 million tonnes in 2016) exceeds that of any other organic compound. Much of this production goes toward creating polyethylene, which is a widely used plastic containing polymer chains of ethylene units in various chain lengths. Production greenhouse gas emissions, emits greenhouse gases, including methane from feedstock production and carbon dioxide from any non-sustainable energy used. Ethylene is also an important natural plant hormone and is used in agriculture to induce ripening of fruits. The hydrate of ethylene is ethanol. Structure and properties This hydrocarbon has four hydrogen atoms bound to a pair of carbon atoms that are connected by a doub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Titration
Titration (also known as titrimetry and volumetric analysis) is a common laboratory method of Quantitative research, quantitative Analytical chemistry, chemical analysis to determine the concentration of an identified analyte (a substance to be analyzed). A reagent, termed the ''titrant'' or ''titrator'', is prepared as a standard solution of known concentration and volume. The titrant reacts with a Solution (chemistry), solution of ''analyte'' (which may also be termed the ''titrand'') to determine the analyte's concentration. The volume of titrant that reacted with the analyte is termed the ''titration volume''. History and etymology The word "titration" descends from the French word ''titrer'' (1543), meaning the proportion of gold or silver in coins or in works of gold or silver; i.e., a measure of fineness or purity. ''Tiltre'' became ''titre'', which thus came to mean the "fineness of alloyed gold", and then the "concentration of a substance in a given sample". In 1828, t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Halogen Addition Reaction
A halogen addition reaction is a simple organic reaction where a halogen molecule is added to the carbon–carbon double bond of an alkene functional group. The general chemical formula of the halogen addition reaction is: :C=C + X2 → X−C−C−X (X represents the halogens bromine or chlorine, and in this case, a solvent could be CH2Cl2 or CCl4). The product is a vicinal dihalide. This type of reaction is a halogenation and an electrophilic addition. Reaction mechanism The reaction mechanism for an alkene bromination can be described as follows. In the first step of the reaction, a bromine molecule approaches the electron-rich alkene carbon–carbon double bond. The bromine atom closer to the bond takes on a partial positive charge as its electrons are repelled by the electrons of the double bond. The atom is electrophilic at this time and is attacked by the pi electrons of the alkene arbon–carbon double bond It forms for an instant a single sigma bond to ''both' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Diisobutylaluminium Hydride
Diisobutylaluminium hydride (DIBALH, DIBAL, DIBAL-H or DIBAH) is a reducing agent with the chemical formula, formula (''i''-Bu2AlH)2, where ''i''-Bu represents isobutyl (-CH2CH(CH3)2). This organoaluminium chemistry, organoaluminium compound is a reagent in organic synthesis. Properties Like most organoaluminum compounds, the compound's structure is most probably more than that suggested by its empirical formula. A variety of techniques, not including X-ray crystallography, suggest that the compound exists as a dimer (chemistry), dimer and a trimer, consisting of tetrahedral aluminium centers sharing bridging hydride ligands. Hydrides are small and, for aluminium derivatives, are highly base (chemistry), basic, thus they bridge in preference to the alkyl groups. DIBAL can be prepared by heating triisobutylaluminium (itself a dimer) to induce Beta-Hydride elimination, β-hydride elimination: :(''i''-Bu3Al)2 → (''i''-Bu2AlH)2 + 2 isobutene, (CH3)2C=CH2 Although DIBAL can be purcha ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Borane
Borane is an inorganic compound with the chemical formula . Because it tends to dimerize or form adducts, borane is very rarely observed. It normally dimerizes to diborane in the absence of other chemicals. It can be observed directly as a continuously produced, transitory, product in a flow system or from the reaction of laser ablated atomic boron with hydrogen. Structure and properties BH3 is a trigonal planar molecule with D3h symmetry. The experimentally determined B–H bond length is 119 pm. In the absence of other bases, it dimerizes to form diborane. Thus, it is an intermediate in the preparation of diborane according to the reaction: :BX3 +BH4− → HBX3− + (BH3) (X=F, Cl, Br, I) :2 BH3 → B2H6 The standard enthalpy of dimerization of BH3 is estimated to be −170 kJ mol−1. The boron atom in BH3 has 6 valence electrons. Consequently, it is a strong Lewis acid and reacts with any Lewis base ('L' in equation below) to form an adduct: :BH3 + L � ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Radical (chemistry)
In chemistry, a radical, also known as a free radical, is an atom, molecule, or ion that has at least one unpaired valence electron. With some exceptions, these unpaired electrons make radicals highly chemically reactive. Many radicals spontaneously dimerize. Most organic radicals have short lifetimes. A notable example of a radical is the hydroxyl radical (HO·), a molecule that has one unpaired electron on the oxygen atom. Two other examples are triplet oxygen and triplet carbene (꞉) which have two unpaired electrons. Radicals may be generated in a number of ways, but typical methods involve redox reactions. Ionizing radiation, heat, electrical discharges, and electrolysis are known to produce radicals. Radicals are intermediates in many chemical reactions, more so than is apparent from the balanced equations. Radicals are important in combustion, atmospheric chemistry, polymerization, plasma chemistry, biochemistry, and many other chemical processes. A majority ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Carbene
In organic chemistry, a carbene is a molecule containing a neutral carbon atom with a Valence (chemistry), valence of two and two unshared valence electrons. The general formula is or where the R represents substituents or hydrogen atoms. The term "carbene" may also refer to the specific compound , also called methylene radical, methylene, the parent hydride from which all other carbene compounds are formally derived. There are two types of carbenes: singlet state, singlets or triplet state, triplets, depending upon their electronic structure. The different classes undergo different reactions. Most carbenes are extremely reactive and short-lived. A small number (the diHalogen, halocarbenes, carbon monoxide, and carbon monosulfide) can be isolated, and can stabilize as Coordination complex, metal ligands, but otherwise cannot be stored in bulk. A rare exception are the persistent carbenes, which have extensive application in modern organometallic chemistry. Generatio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |