|
Edward Slater
Edward Charles Slater (16 January 1917 – 26 March 2016), also known as Bill Slater, was an Australian biochemist who spent most of his career at the University of Amsterdam. Early life and education Slater was raised in Australia. He received a training in biochemistry at the Ormond College of the University of Melbourne. In 1946, he moved to Cambridge, where he earned his PhD under the supervision of David Keilin. Career In 1955, Slater joined the medical faculty of the University of Amsterdam, where he remained until retiring in 1985. He is recognised for his contributions to the development of Dutch biochemistry. Slater managed the journal ''Biochimica et Biophysica Acta'', turning it into one of the most influential publications in the field. He wrote a history of the journal, ''Biochimica et biophysica acta: the story of a biochemical journal'', which was published in 1986. He served as the president of the International Union of Biochemistry and Molecular Biology ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Melbourne
Melbourne ( ; Boonwurrung/ Woiwurrung: ''Narrm'' or ''Naarm'') is the capital and most populous city of the Australian state of Victoria, and the second-most populous city in both Australia and Oceania. Its name generally refers to a metropolitan area known as Greater Melbourne, comprising an urban agglomeration of 31 local municipalities, although the name is also used specifically for the local municipality of City of Melbourne based around its central business area. The metropolis occupies much of the northern and eastern coastlines of Port Phillip Bay and spreads into the Mornington Peninsula, part of West Gippsland, as well as the hinterlands towards the Yarra Valley, the Dandenong and Macedon Ranges. It has a population over 5 million (19% of the population of Australia, as per 2021 census), mostly residing to the east side of the city centre, and its inhabitants are commonly referred to as "Melburnians". The area of Melbourne has been home to Abori ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iron–sulfur Protein
Iron–sulfur proteins (or iron–sulphur proteins in British spelling) are proteins characterized by the presence of iron–sulfur clusters containing sulfide-linked di-, tri-, and tetrairon centers in variable oxidation states. Iron–sulfur clusters are found in a variety of metalloproteins, such as the ferredoxins, as well as NADH dehydrogenase, hydrogenases, coenzyme Q – cytochrome c reductase, succinate – coenzyme Q reductase and nitrogenase. Iron–sulfur clusters are best known for their role in the oxidation-reduction reactions of electron transport in mitochondria and chloroplasts. Both Complex I and Complex II of oxidative phosphorylation have multiple Fe–S clusters. They have many other functions including catalysis as illustrated by aconitase, generation of radicals as illustrated by SAM-dependent enzymes, and as sulfur donors in the biosynthesis of lipoic acid and biotin. Additionally, some Fe–S proteins regulate gene expression. Fe–S proteins are vulnera ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Royal Netherlands Academy Of Arts And Sciences
The Royal Netherlands Academy of Arts and Sciences ( nl, Koninklijke Nederlandse Akademie van Wetenschappen, abbreviated: KNAW) is an organization dedicated to the advancement of science and literature in the Netherlands. The academy is housed in the Trippenhuis in Amsterdam. In addition to various advisory and administrative functions it operates a number of research institutes and awards many prizes, including the Lorentz Medal in theoretical physics, the Dr Hendrik Muller Prize for Behavioural and Social Science and the Heineken Prizes. Main functions The academy advises the Dutch government on scientific matters. While its advice often pertains to genuine scientific concerns, it also counsels the government on such topics as policy on careers for researchers or the Netherlands' contribution to major international projects. The academy offers solicited and unsolicited advice to parliament, ministries, universities and research institutes, funding agencies and internatio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glyceraldehyde Phosphate Dehydrogenase
Glyceraldehyde 3-phosphate dehydrogenase (abbreviated GAPDH) () is an enzyme of about 37kDa that catalyzes the sixth step of glycolysis and thus serves to break down glucose for energy and carbon molecules. In addition to this long established metabolic function, GAPDH has recently been implicated in several non-metabolic processes, including transcription activation, initiation of apoptosis, ER to Golgi vesicle shuttling, and fast axonal, or axoplasmic transport. In sperm, a testis-specific isoenzyme GAPDHS is expressed. Structure Under normal cellular conditions, cytoplasmic GAPDH exists primarily as a tetramer. This form is composed of four identical 37-kDa subunits containing a single catalytic thiol group each and critical to the enzyme's catalytic function. Nuclear GAPDH has increased isoelectric point (pI) of pH 8.3–8.7. Of note, the cysteine residue C152 in the enzyme's active site is required for the induction of apoptosis by oxidative stress. Notably, pos ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nicotinamide Adenine Dinucleotide
Nicotinamide adenine dinucleotide (NAD) is a coenzyme central to metabolism. Found in all living cells, NAD is called a dinucleotide because it consists of two nucleotides joined through their phosphate groups. One nucleotide contains an adenine nucleobase and the other nicotinamide. NAD exists in two forms: an oxidized and reduced form, abbreviated as NAD and NADH (H for hydrogen), respectively. In metabolism, nicotinamide adenine dinucleotide is involved in redox reactions, carrying electrons from one reaction to another. The cofactor is, therefore, found in two forms in cells: NAD is an oxidizing agent – it accepts electrons from other molecules and becomes reduced. This reaction, also with H+, forms NADH, which can then be used as a reducing agent to donate electrons. These electron transfer reactions are the main function of NAD. However, it is also used in other cellular processes, most notably as a substrate of enzymes in adding or removing chemical ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzyme
Enzymes () are proteins that act as biological catalysts by accelerating chemical reactions. The molecules upon which enzymes may act are called substrate (chemistry), substrates, and the enzyme converts the substrates into different molecules known as product (chemistry), products. Almost all metabolism, metabolic processes in the cell (biology), cell need enzyme catalysis in order to occur at rates fast enough to sustain life. Metabolic pathways depend upon enzymes to catalyze individual steps. The study of enzymes is called ''enzymology'' and the field of pseudoenzyme, pseudoenzyme analysis recognizes that during evolution, some enzymes have lost the ability to carry out biological catalysis, which is often reflected in their amino acid sequences and unusual 'pseudocatalytic' properties. Enzymes are known to catalyze more than 5,000 biochemical reaction types. Other biocatalysts are Ribozyme, catalytic RNA molecules, called ribozymes. Enzymes' Chemical specificity, specific ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ligand
In coordination chemistry, a ligand is an ion or molecule ( functional group) that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's electron pairs, often through Lewis bases. The nature of metal–ligand bonding can range from covalent to ionic. Furthermore, the metal–ligand bond order can range from one to three. Ligands are viewed as Lewis bases, although rare cases are known to involve Lewis acidic "ligands". Metals and metalloids are bound to ligands in almost all circumstances, although gaseous "naked" metal ions can be generated in a high vacuum. Ligands in a complex dictate the reactivity of the central atom, including ligand substitution rates, the reactivity of the ligands themselves, and redox. Ligand selection requires critical consideration in many practical areas, including bioinorganic and medicinal chemistry, homogeneous catalysis, and environm ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
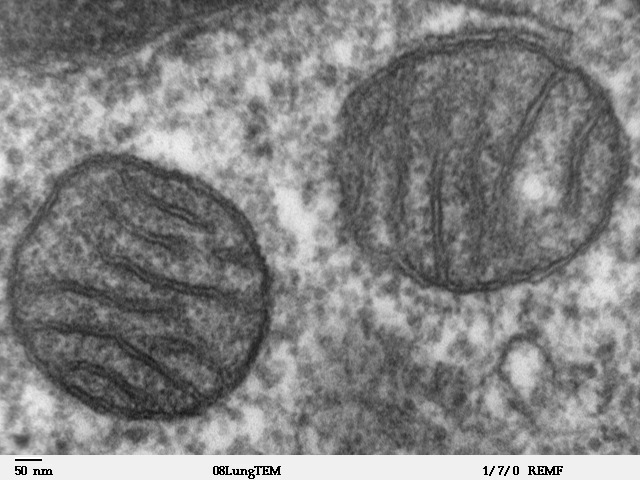
Mitochondrial Membrane
A mitochondrion (; ) is an organelle found in the cells of most Eukaryotes, such as animals, plants and fungi. Mitochondria have a double membrane structure and use aerobic respiration to generate adenosine triphosphate (ATP), which is used throughout the cell as a source of chemical energy. They were discovered by Albert von Kölliker in 1857 in the voluntary muscles of insects. The term ''mitochondrion'' was coined by Carl Benda in 1898. The mitochondrion is popularly nicknamed the "powerhouse of the cell", a phrase coined by Philip Siekevitz in a 1957 article of the same name. Some cells in some multicellular organisms lack mitochondria (for example, mature mammalian red blood cells). A large number of unicellular organisms, such as microsporidia, parabasalids and diplomonads, have reduced or transformed their mitochondria into other structures. One eukaryote, ''Monocercomonoides'', is known to have completely lost its mitochondria, and one multicellular organism, ''Henn ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cooperativity
Cooperativity is a phenomenon displayed by systems involving identical or near-identical elements, which act dependently of each other, relative to a hypothetical standard non-interacting system in which the individual elements are acting independently. One manifestation of this is enzymes or receptors that have multiple binding sites where the affinity of the binding sites for a ligand is ''apparently'' increased, positive cooperativity, or decreased, negative cooperativity, upon the binding of a ligand to a binding site. For example, when an oxygen atom binds to one of hemoglobin's four binding sites, the affinity to oxygen of the three remaining available binding sites increases; i.e. oxygen is more likely to bind to a hemoglobin bound to one oxygen than to an unbound hemoglobin. This is referred to as cooperative binding. We also see cooperativity in large chain molecules made of many identical (or nearly identical) subunits (such as DNA, proteins, and phospholipids), when ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
ATP Synthase
ATP synthase is a protein that catalyzes the formation of the energy storage molecule adenosine triphosphate (ATP) using adenosine diphosphate (ADP) and inorganic phosphate (Pi). It is classified under ligases as it changes ADP by the formation of P-O bond (phosphodiester bond). ATP synthase is a molecular machine. The overall reaction catalyzed by ATP synthase is: * ADP + Pi + 2H+out ATP + H2O + 2H+in The formation of ATP from ADP and Pi is energetically unfavorable and would normally proceed in the reverse direction. In order to drive this reaction forward, ATP synthase couples ATP synthesis during cellular respiration to an electrochemical gradient created by the difference in proton (H+) concentration across the inner mitochondrial membrane in eukaryotes or the plasma membrane in bacteria. During photosynthesis in plants, ATP is synthesized by ATP synthase using a proton gradient created in the thylakoid lumen through the thylakoid membrane and into the chloroplast ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |


4-3D-balls.png)