|
Dubnium
Dubnium is a synthetic element, synthetic chemical element; it has Chemical symbol, symbol Db and atomic number 105. It is highly radioactive: the most stable known isotopes of dubnium, isotope, dubnium-268, has a half-life of about 16 hours. This greatly limits extended research on the element. Dubnium does not occur naturally on Earth and is produced artificially. The Soviet Joint Institute for Nuclear Research (JINR) claimed the first discovery of the element in 1968, followed by the American Lawrence Berkeley Laboratory in 1970. Both teams proposed their names for the new element and used them without formal approval. The long-standing dispute was resolved in 1993 by an official investigation of the discovery claims by the Transfermium Working Group, formed by the International Union of Pure and Applied Chemistry and the International Union of Pure and Applied Physics, resulting in credit for the discovery being officially shared between both teams. The element was formally ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
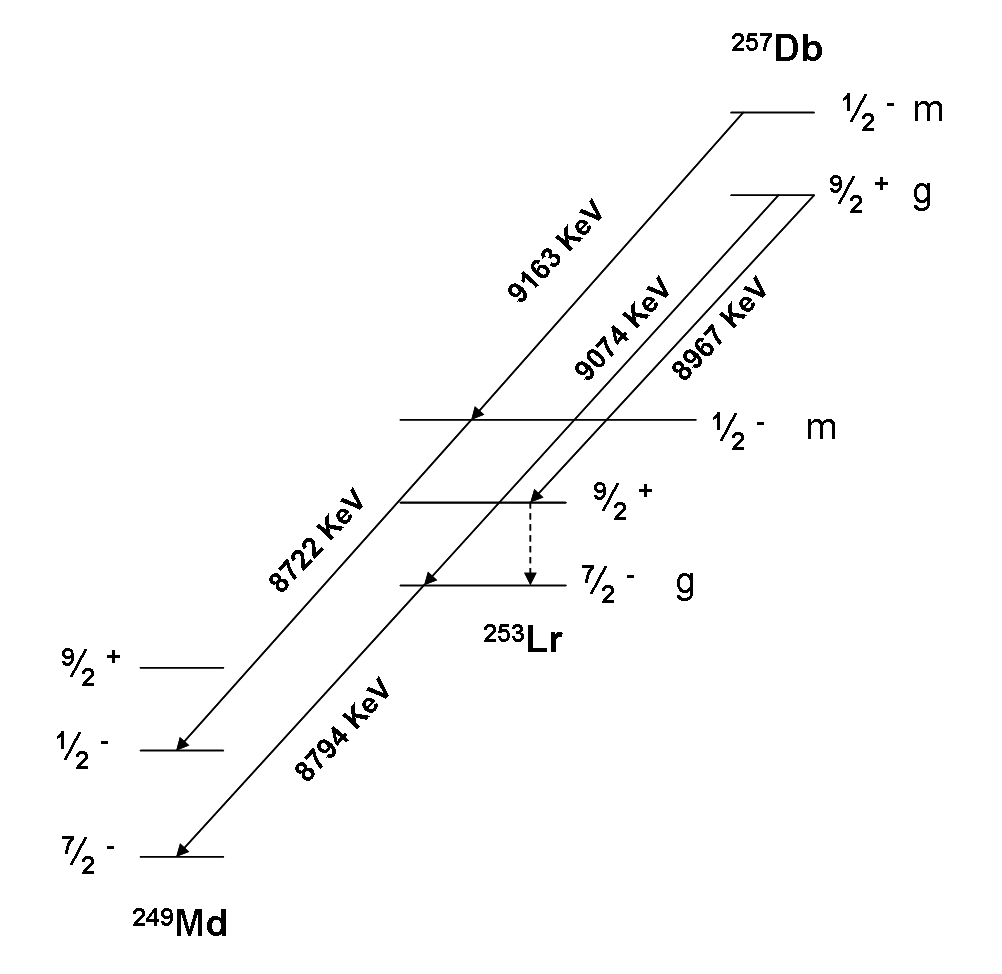
Isotopes Of Dubnium
Dubnium (105Db) is a synthetic element, thus a standard atomic weight cannot be given. Like all synthetic elements, it has no stable isotopes. The first isotope to be synthesized was 261Db in 1968. Thirteen radioisotopes are known, ranging from 255Db to 270Db (except 264Db, 265Db, and 269Db), along with one nuclear isomer, isomer (257mDb); two more isomers have been reported but are unconfirmed. The longest-lived known isotope is 268Db with a half-life of 16 hours. List of isotopes , -id=Dubnium-255 , rowspan=2, 255Db , rowspan=2 style="text-align:right" , 105 , rowspan=2 style="text-align:right" , 150 , rowspan=2, 255.10692(30)# , rowspan=2, 54 ms , Spontaneous fission, SF (67%) , (various) , rowspan=2, 9/2+# , - , alpha decay, α (33%?) , 251Lr , -id=Dubnium-255m , rowspan=2, 255mDbOrder of ground state and isomer is uncertain. , rowspan=2 colspan=3 style="text-indent:2em", 100(100)# keV , rowspan=2, , SF (92%) , (various) , rowspan=2, 1/2−# , - , � ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Group 5 Element
Group 5 is a group of elements in the periodic table. Group 5 contains vanadium (V), niobium (Nb), tantalum (Ta) and dubnium (Db). This group lies in the d-block of the periodic table. This group is sometimes called the vanadium group or vanadium family after its lightest member; however, the group itself has not acquired a trivial name because it belongs to the broader grouping of the transition metals. As is typical for early transition metals, niobium and tantalum have only the group oxidation state of +5 as a major one, and are quite electropositive (it is easy to donate electrons) and have a less rich coordination chemistry (the chemistry of metallic ions bound with molecules). Due to the effects of the lanthanide contraction, the decrease in ionic radii in the lanthanides, they are very similar in properties. Vanadium is somewhat distinct due to its smaller size: it has well-defined +2, +3 and +4 states as well (although +5 is more stable). The lighter three Group 5 el ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transfermium Wars
The names for the chemical elements 104 to 106 were the subject of a major controversy starting in the 1960s, described by some nuclear chemists as the Transfermium Wars because it concerned the elements following fermium (element 100) on the periodic table. This controversy arose from disputes between American scientists and Soviet scientists as to which had first isolated these elements. The final resolution of this controversy in 1997 also decided the names of elements 107 to 109. Controversy By convention, naming rights for newly discovered chemical elements go to their discoverers. For elements 104, 105, and 106, there was a controversy between Soviet researchers at the Joint Institute for Nuclear Research and American researchers at Lawrence Berkeley National Laboratory regarding which group had discovered them first. Both parties suggested their own names for elements 104 and 105, not recognizing the other's name. The American name of seaborgium for element 106 was a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Discovery Of The Chemical Elements
The discoveries of the 118 chemical elements known to exist as of 2025 are presented here in chronological order. The elements are listed generally in the order in which each was first defined as the pure element, as the exact date of discovery of most elements cannot be accurately determined. There are plans to synthesize more elements, and it is not known how many elements are possible. Each element's list of elements by name, name, atomic number, year of first report, name of the discoverer, and notes related to the discovery are listed. Periodic table of elements Graphical timeline ImageSize = width:1600 height:120 # barincrement:0 PlotArea = top:70 bottom:30 right:10 left:10 AlignBars = justify Colors = id:gray1 value:gray(0.85) legend:Independent id:gray2 value:gray(0.95) DateFormat = yyyy Period = from:1665 till:2025 TimeAxis = orientation:horizontal ScaleMajor = unit:year increment:10 start:1670 ScaleMinor = unit:year increment:1 start:1665 TextData = tex ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Symbol
Chemical symbols are the abbreviations used in chemistry, mainly for chemical elements; but also for functional groups, chemical compounds, and other entities. Element symbols for chemical elements, also known as atomic symbols, normally consist of one or two letters from the Latin alphabet and are written with the first letter capitalised. History Earlier symbols for chemical elements stem from classical Latin and Greek language, Greek words. For some elements, this is because the material was known in ancient times, while for others, the name is a more recent invention. For example, Pb is the symbol for lead (''plumbum'' in Latin); Hg is the symbol for mercury (element), mercury (''hydrargyrum'' in Greek); and He is the symbol for helium (a Neo-Latin name) because helium was not known in ancient Roman times. Some symbols come from other sources, like W for tungsten (''Wolfram'' in German) which was not known in Roman times. A three-letter Systematic element name, temporary sym ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electron Configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon atom is , meaning that the 1s, 2s, and 2p subshells are occupied by two, two, and six electrons, respectively. Electronic configurations describe each electron as moving independently in an orbital, in an average field created by the nuclei and all the other electrons. Mathematically, configurations are described by Slater determinants or configuration state functions. According to the laws of quantum mechanics, a level of energy is associated with each electron configuration. In certain conditions, electrons are able to move from one configuration to another by the emission or absorption of a quantum of energy, in the form of a photon. Knowledge of the electron configuration of different atoms is useful in understanding the structu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transactinide Chemistry Apparatus Dubna
Superheavy elements, also known as transactinide elements, transactinides, or super-heavy elements, or superheavies for short, are the chemical elements with atomic number greater than 104. The superheavy elements are those beyond the actinides in the periodic table; the last actinide is lawrencium (atomic number 103). By definition, superheavy elements are also transuranium elements, i.e., having atomic numbers greater than that of uranium (92). Depending on the definition of group 3 adopted by authors, lawrencium may also be included to complete the 6d series. Glenn T. Seaborg first proposed the actinide concept, which led to the acceptance of the actinide series. He also proposed a transactinide series ranging from element 104 to 121 and a superactinide series approximately spanning elements 122 to 153 (though more recent work suggests the end of the superactinide series to occur at element 157 instead). The transactinide seaborgium was named in his honor. Superheavies are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transition Metal
In chemistry, a transition metal (or transition element) is a chemical element in the d-block of the periodic table (groups 3 to 12), though the elements of group 12 (and less often group 3) are sometimes excluded. The lanthanide and actinide elements (the f-block) are called inner transition metals and are sometimes considered to be transition metals as well. They are lustrous metals with good electrical and thermal conductivity. Most (with the exception of group 11 and group 12) are hard and strong, and have high melting and boiling temperatures. They form compounds in any of two or more different oxidation states and bind to a variety of ligands to form coordination complexes that are often coloured. They form many useful alloys and are often employed as catalysts in elemental form or in compounds such as coordination complexes and oxides. Most are strongly paramagnetic because of their unpaired d electrons, as are many of their compounds. All of the elements that are ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Synthetic Element
A synthetic element is a known chemical element that does not occur naturally on Earth: it has been created by human manipulation of fundamental particles in a nuclear reactor, a particle accelerator, or the explosion of an atomic bomb; thus, it is called "synthetic", "artificial", or "man-made". The synthetic elements are those with atomic numbers 95–118, as shown in purple on the accompanying periodic table: these 24 elements were first created between 1944 and 2010. The mechanism for the creation of a synthetic element is to force additional protons into the Atomic nucleus, nucleus of an element with an atomic number lower than 95. All known (see: Island of stability) synthetic elements are unstable, but they radioactive decay, decay at widely varying rates; the half-lives of their longest-lived isotopes range from microseconds to millions of years. Five more elements that were first created artificially are strictly speaking not ''synthetic'' because they were later found in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Elsevier
Elsevier ( ) is a Dutch academic publishing company specializing in scientific, technical, and medical content. Its products include journals such as ''The Lancet'', ''Cell (journal), Cell'', the ScienceDirect collection of electronic journals, ''Trends (journals), Trends'', the ''Current Opinion (Elsevier), Current Opinion'' series, the online citation database Scopus, the SciVal tool for measuring research performance, the ClinicalKey search engine for clinicians, and the ClinicalPath evidence-based cancer care service. Elsevier's products and services include digital tools for Data management platform, data management, instruction, research analytics, and assessment. Elsevier is part of the RELX Group, known until 2015 as Reed Elsevier, a publicly traded company. According to RELX reports, in 2022 Elsevier published more than 600,000 articles annually in over 2,800 journals. As of 2018, its archives contained over 17 million documents and 40,000 Ebook, e-books, with over one b ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Moscow Oblast
Moscow Oblast (, , informally known as , ) is a federal subjects of Russia, federal subject of Russia (an oblast). With a population of 8,524,665 (Russian Census (2021), 2021 Census) living in an area of , it is one of the most densely populated regions in the country and is the list of federal subjects of Russia by population, second most populous federal subject. The oblast has no official administrative center; its public authorities are located in Moscow and Krasnogorsk, Moscow Oblast, Krasnogorsk (the Moscow Oblast Duma and the local government), and also across other locations in the oblast.According to Article 24 of the Charter of Moscow Oblast, the government bodies of the oblast are located in the city of Moscow and throughout the territory of Moscow Oblast. However, Moscow is not named the official administrative center of the oblast. Located in European Russia between latitudes 54th parallel north, 54° and 57th parallel north, 57° N and longitudes 35th meridian ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Soviet Union
The Union of Soviet Socialist Republics. (USSR), commonly known as the Soviet Union, was a List of former transcontinental countries#Since 1700, transcontinental country that spanned much of Eurasia from 1922 until Dissolution of the Soviet Union, it dissolved in 1991. During its existence, it was the list of countries and dependencies by area, largest country by area, extending across Time in Russia, eleven time zones and sharing Geography of the Soviet Union#Borders and neighbors, borders with twelve countries, and the List of countries and dependencies by population, third-most populous country. An overall successor to the Russian Empire, it was nominally organized as a federal union of Republics of the Soviet Union, national republics, the largest and most populous of which was the Russian SFSR. In practice, Government of the Soviet Union, its government and Economy of the Soviet Union, economy were Soviet-type economic planning, highly centralized. As a one-party state go ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |