|
Uranium
Uranium is a chemical element; it has chemical symbol, symbol U and atomic number 92. It is a silvery-grey metal in the actinide series of the periodic table. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons. Uranium radioactive decay, radioactively decays, usually by emitting an alpha particle. The half-life of this decay varies between 159,200 and 4.5 billion years for different isotopes of uranium, isotopes, making them useful for dating the age of the Earth. The most common isotopes in natural uranium are uranium-238 (which has 146 neutrons and accounts for over 99% of uranium on Earth) and uranium-235 (which has 143 neutrons). Uranium has the highest atomic weight of the primordial nuclide, primordially occurring elements. Its density is about 70% higher than that of lead and slightly lower than that of gold or tungsten. It occurs naturally in low concentrations of a few Parts-per notation#Parts-per expressions, parts per million in soil, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
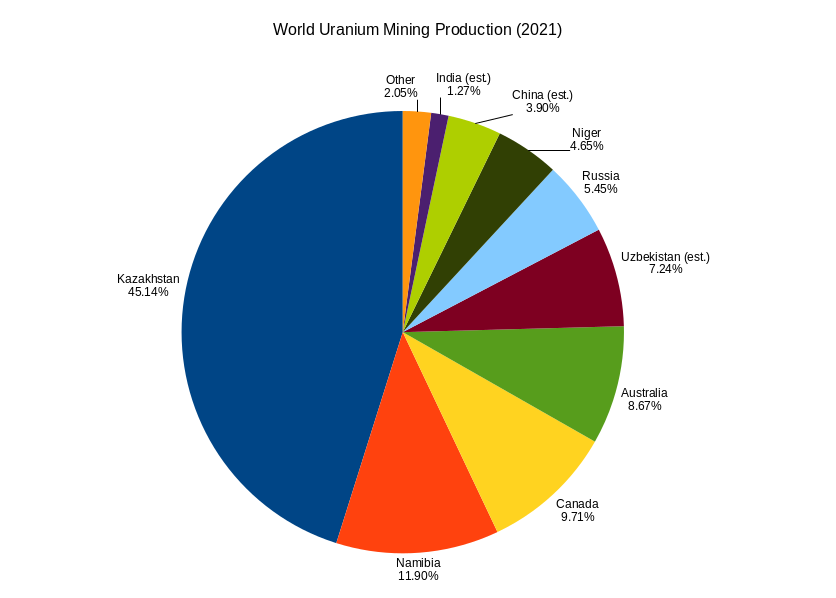
Uranium Mining
Uranium mining is the process of extraction of uranium ore from the earth. Over 50,000 tons of uranium were produced in 2019. Kazakhstan, Canada, and Australia were the top three uranium producers, respectively, and together account for 68% of world production. Other countries producing more than 1,000 tons per year included Namibia, Niger, Russia, Uzbekistan and China. Nearly all of the world's mined uranium is used to power nuclear power plants. Historically uranium was also used in applications such as uranium glass or ferrouranium but those applications have declined due to the radioactivity and toxicity of uranium and are nowadays mostly supplied with a plentiful cheap supply of depleted uranium which is also used in Armour-piercing ammunition, uranium ammunition. In addition to being cheaper, depleted uranium is also less radioactive due to a lower content of short-lived and than natural uranium. Uranium is mined by in-situ leaching (57% of world production) or by convent ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isotopes Of Uranium
Uranium (U) is a naturally occurring radioactive element (radioelement) with no stable isotopes. It has two primordial isotopes, uranium-238 and uranium-235, that have long half-lives and are found in appreciable quantity in Earth's crust. The decay product uranium-234 is also found. Other isotopes such as uranium-233 have been produced in breeder reactors. In addition to isotopes found in nature or nuclear reactors, many isotopes with far shorter half-lives have been produced, ranging from U to U (except for U). The standard atomic weight of natural uranium is . Natural uranium consists of three main isotopes, U (99.2739–99.2752% natural abundance), U (0.7198–0.7202%), and U (0.0050–0.0059%). All three isotopes are radioactive (i.e., they are radioisotopes), and the most abundant and stable is uranium-238, with a half-life of (about the age of the Earth). Uranium-238 is an alpha emitter, decaying through the 18-member uranium series into lead-206. The decay serie ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Actinide
The actinide () or actinoid () series encompasses at least the 14 metallic chemical elements in the 5f series, with atomic numbers from 89 to 102, actinium through nobelium. Number 103, lawrencium, is also generally included despite being part of the 6d transition series. The actinide series derives its name from the first element in the series, actinium. The informal chemical symbol An is used in general discussions of actinide chemistry to refer to any actinide. The 1985 IUPAC nomenclature of inorganic chemistry, IUPAC ''Red Book'' recommends that ''actinoid'' be used rather than ''actinide'', since the suffix ''-ide'' normally indicates a negative ion. However, owing to widespread current use, ''actinide'' is still allowed. Actinium through nobelium are f-block elements, while lawrencium is a d-block element and a transition metal. The series mostly corresponds to the filling of the 5f electron shell, although as isolated atoms in the ground state many have anomalous configu ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Uranium-238
Uranium-238 ( or U-238) is the most common isotope of uranium found in nature, with a relative abundance of 99%. Unlike uranium-235, it is non-fissile, which means it cannot sustain a chain reaction in a thermal-neutron reactor. However, it is fissionable by fast neutrons, and is ''fertile'', meaning it can be transmuted to fissile plutonium-239. 238U cannot support a chain reaction because inelastic scattering reduces neutron energy below the range where fast fission of one or more next-generation nuclei is probable. Doppler broadening of 238U's neutron absorption resonances, increasing absorption as fuel temperature increases, is also an essential negative feedback mechanism for reactor control. Around 99.284% of natural uranium's mass is uranium-238, which has a half-life of 1.41 seconds (4.468 years, or 4.468 billion years). Due to its natural abundance and half-life relative to other radioactive elements, 238U produces ~40% of the radioactive heat produced wit ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Uranium-235
Uranium-235 ( or U-235) is an isotope of uranium making up about 0.72% of natural uranium. Unlike the predominant isotope uranium-238, it is fissile, i.e., it can sustain a nuclear chain reaction. It is the only fissile isotope that exists in nature as a primordial nuclide. Uranium-235 has a half-life of 703.8 million years. It was discovered in 1935 by Arthur Jeffrey Dempster. Its fission cross section for slow thermal neutrons is about Barn (unit), barns. For fast neutrons it is on the order of 1 barn. Most neutron absorptions induce fission, though a minority (about 15%) result in the formation of uranium-236. Fission properties The fission of one atom of uranium-235 releases () inside the reactor. That corresponds to 19.54 TJ/mole (unit), mol, or 83.14 TJ/kg. [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chemical Symbol
Chemical symbols are the abbreviations used in chemistry, mainly for chemical elements; but also for functional groups, chemical compounds, and other entities. Element symbols for chemical elements, also known as atomic symbols, normally consist of one or two letters from the Latin alphabet and are written with the first letter capitalised. History Earlier symbols for chemical elements stem from classical Latin and Greek language, Greek words. For some elements, this is because the material was known in ancient times, while for others, the name is a more recent invention. For example, Pb is the symbol for lead (''plumbum'' in Latin); Hg is the symbol for mercury (element), mercury (''hydrargyrum'' in Greek); and He is the symbol for helium (a Neo-Latin name) because helium was not known in ancient Roman times. Some symbols come from other sources, like W for tungsten (''Wolfram'' in German) which was not known in Roman times. A three-letter Systematic element name, temporary sym ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Uraninite
Uraninite, also known as pitchblende, is a radioactive, uranium-rich mineral and ore with a chemical composition that is largely UO2 but because of oxidation typically contains variable proportions of U3O8. Radioactive decay of the uranium causes the mineral to contain oxides of lead and trace amounts of helium. It may also contain thorium and rare-earth elements. Overview Uraninite used to be known as pitchblende (from '' pitch'', because of its black color, and ''blende'', from ''blenden'' meaning "to deceive", a term used by German miners to denote minerals whose density suggested metal content, but whose exploitation, at the time they were named, was either unknown or not economically feasible). The mineral has been known since at least the 15th century, from silver mines in the Ore Mountains, on the German/Czech border. The type locality is the historic mining and spa town known as Joachimsthal, the modern-day Jáchymov, on the Czech side of the mountains, where F. E. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fissile
In nuclear engineering, fissile material is material that can undergo nuclear fission when struck by a neutron of low energy. A self-sustaining thermal Nuclear chain reaction#Fission chain reaction, chain reaction can only be achieved with fissile material. The predominant Neutron temperature, neutron energy in a system may be typified by either slow neutrons (i.e., a thermal system) or fast neutrons. Fissile material can be used to fuel thermal-neutron reactors, fast-neutron reactors and nuclear explosives. Fissile vs fissionable The term ''fissile'' is distinct from ''fissionable''. A nuclide that can undergo nuclear fission (even with a low probability) after capturing a neutron of high or low energy is referred to as ''fissionable''. A fissionable nuclide that can undergo fission with a high probability after capturing a low-energy thermal neutron is referred to as ''fissile''. Fissionable materials include those (such as uranium-238) for which fission can be induce ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Natural Uranium
Natural uranium (NU or Unat) is uranium with the same isotopic ratio as found in nature. It contains 0.711% uranium-235, 99.284% uranium-238, and a trace of uranium-234 by weight (0.0055%). Approximately 2.2% of its radioactivity comes from uranium-235, 48.6% from uranium-238, and 49.2% from uranium-234. Natural uranium can be used to fuel both low- and high-power nuclear reactors. Historically, graphite-moderated reactors and heavy water-moderated reactors have been fueled with natural uranium in the pure metal (U) or uranium dioxide (UO2) ceramic forms. However, experimental fuelings with uranium trioxide (UO3) and triuranium octaoxide (U3O8) have shown promise. The 0.72% uranium-235 is not sufficient to produce a self-sustaining critical chain reaction in light water reactors or nuclear weapons; these applications must use enriched uranium. Nuclear weapons take a concentration of 90% uranium-235, and light water reactors require a concentration of roughly 3% uranium-235 ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Primordial Nuclide
In geochemistry, geophysics and nuclear physics, primordial nuclides, also known as primordial isotopes, are nuclides found on Earth that have existed in their current form since before Earth was formed. Primordial nuclides were present in the interstellar medium from which the Solar System was formed, and were formed in, or after, the Big Bang, by nucleosynthesis in stars and supernovae followed by mass ejection, by cosmic ray spallation, and potentially from other processes. They are the stable nuclides plus the long-lived fraction of radionuclides surviving in the primordial solar nebula through planet accretion until the present; 286 such nuclides are known. Stability All of the known 251 stable nuclides, plus another 35 nuclides that have half-lives long enough to have survived from the formation of the Earth, occur as primordial nuclides. These 35 primordial radionuclides represent isotopes of 28 separate elements. Cadmium, tellurium, xenon, neodymium, samarium, osmi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Neutron
The neutron is a subatomic particle, symbol or , that has no electric charge, and a mass slightly greater than that of a proton. The Discovery of the neutron, neutron was discovered by James Chadwick in 1932, leading to the discovery of nuclear fission in 1938, the first self-sustaining nuclear reactor (Chicago Pile-1, 1942) and the first nuclear weapon (Trinity (nuclear test), Trinity, 1945). Neutrons are found, together with a similar number of protons in the atomic nucleus, nuclei of atoms. Atoms of a chemical element that differ only in neutron number are called isotopes. Free neutrons are produced copiously in nuclear fission and nuclear fusion, fusion. They are a primary contributor to the nucleosynthesis of chemical elements within stars through fission, fusion, and neutron capture processes. Neutron stars, formed from massive collapsing stars, consist of neutrons at the density of atomic nuclei but a total mass more than the Sun. Neutron properties and interactions ar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nuclear Power Plant
A nuclear power plant (NPP), also known as a nuclear power station (NPS), nuclear generating station (NGS) or atomic power station (APS) is a thermal power station in which the heat source is a nuclear reactor. As is typical of thermal power stations, heat is used to generate steam that drives a steam turbine connected to a electric generator, generator that produces electricity. , the International Atomic Energy Agency reported that there were 410 nuclear power reactors in operation in 32 countries around the world, and 57 nuclear power reactors under construction. Most nuclear power plants use thermal reactors with enriched uranium in a Nuclear fuel cycle#Once-through nuclear fuel cycle, once-through fuel cycle. Fuel is removed when the percentage of neutron poison, neutron absorbing atoms becomes so large that a nuclear chain reaction, chain reaction can no longer be sustained, typically three years. It is then cooled for several years in on-site spent fuel pools be ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |