|
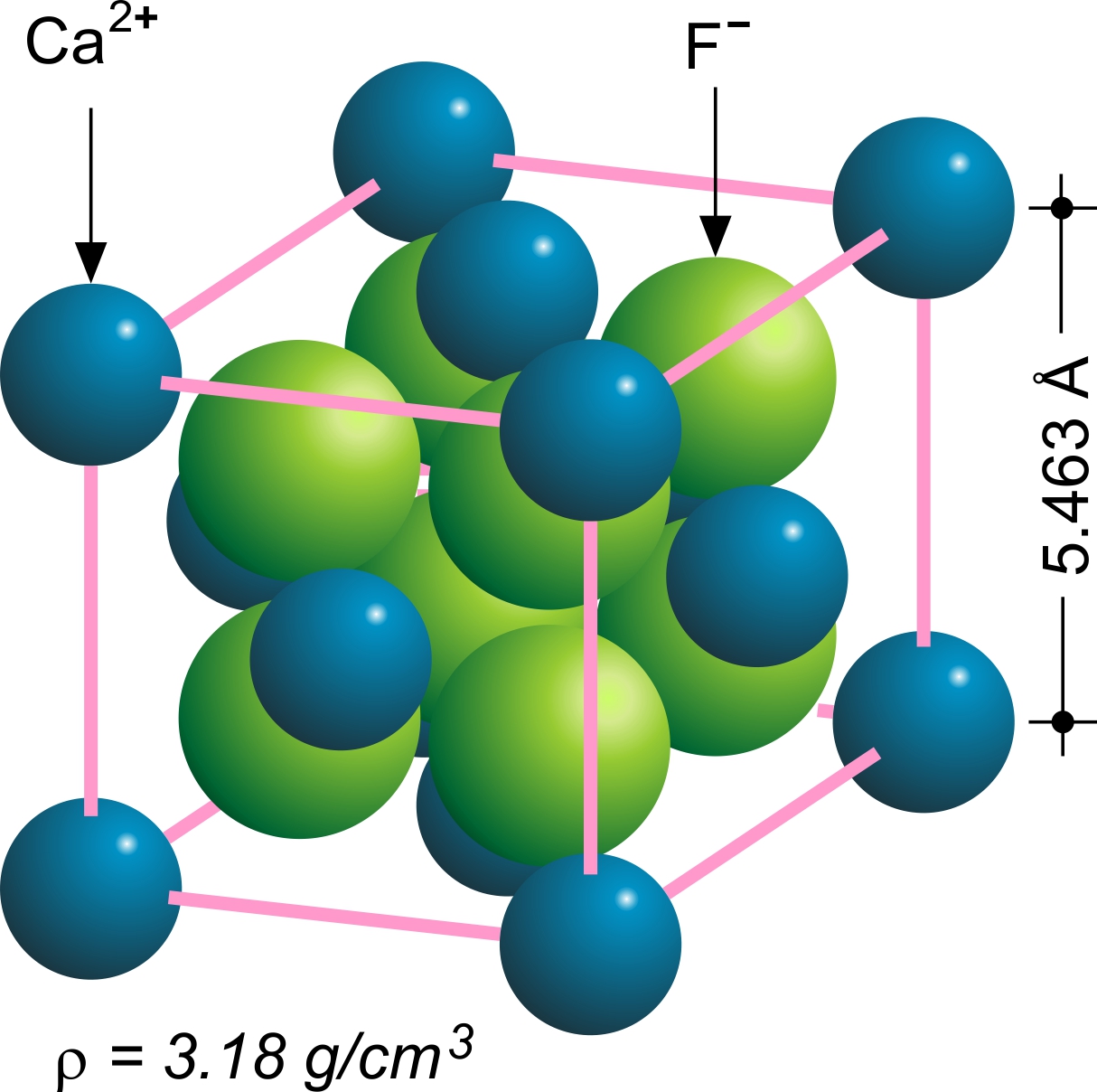
Fluorite Crystal Structure
The fluorite structure refers to a common motif for compounds with the formula MX2. The X ions occupy the eight tetrahedral interstitial sites whereas M ions occupy the regular sites of a face-centered cubic (FCC) structure. Many compounds, notably the common mineral fluorite (CaF2), adopt this structure. Many compounds with formula M2X have an antifluorite structure. In these the locations of the anions and cations are reversed relative to fluorite (an anti-structure); the anions occupy the FCC regular sites whereas the cations occupy the tetrahedral interstitial sites. For example, magnesium silicide, Mg2Si, has a lattice parameter of 6.338 Å with magnesium cations occupying the tetrahedral interstitial sites, in which each silicide A silicide is a type of chemical compound that combines silicon and a usually more electropositive element. Silicon is more electropositive than carbon. In terms of their physical properties, silicides are structurally closer to borides than t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Interstitial Site
In crystallography, interstitial sites, holes or voids are the empty space that exists between the packing of atoms (spheres) in the crystal structure. The holes are easy to see if you try to Circle packing, pack circles together; no matter how close you get them or how you arrange them, you will have empty space in between. The same is true in a unit cell; no matter how the atoms are arranged, there will be interstitial sites present between the atoms. These sites or holes can be filled with other atoms (interstitial defect). The picture with packed circles is only a 2D representation. In a crystal lattice, the atoms (spheres) would be Sphere packing, packed in a 3D arrangement. This results in different shaped interstitial sites depending on the arrangement of the atoms in the lattice. Close packed A close packed unit cell, both face-centered cubic and hexagonal close packed, can form two different shaped holes. Looking at the three green spheres in the hexagonal packi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cubic Crystal System
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals. There are three main varieties of these crystals: *Primitive cubic (abbreviated ''cP'' and alternatively called simple cubic) *Body-centered cubic (abbreviated ''cI'' or bcc) *Face-centered cubic (abbreviated ''cF'' or fcc) Note: the term fcc is often used in synonym for the ''cubic close-packed'' or ccp structure occurring in metals. However, fcc stands for a face-centered cubic Bravais lattice, which is not necessarily close-packed when a motif is set onto the lattice points. E.g. the diamond and the zincblende lattices are fcc but not close-packed. Each is subdivided into other variants listed below. Although the ''unit cells'' in these crystals are conventionally taken to be cubes, the primitive unit cells often are not. Bravais lattices The three Bravais latices ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluorite
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon. The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 4 as fluorite. Pure fluorite is colourless and transparent, both in visible and ultraviolet light, but impurities usually make it a colorful mineral and the stone has ornamental and lapidary uses. Industrially, fluorite is used as a flux for smelting, and in the production of certain glasses and enamels. The purest grades of fluorite are a source of fluoride for hydrofluoric acid manufacture, which is the intermediate source of most fluorine-containing fine chemicals. Optically clear transparent fluorite has anomalous partial dispersion, that is, its refractive index varies with the wavelength of light in a manner that differs from that of commonly used glasses, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Anti-structure
In crystallography, an anti-structure is obtained from a salt structure by exchanging anion and cation positions. For instance, calcium fluoride, CaF2, crystallizes in a cubic motif called the fluorite structure. The same crystal structure is found in numerous ionic compounds with formula AB2, such as ceria (CeO2), zirconia (cubic ZrO2), uranium dioxide (UO2). In the corresponding ''anti-structure'', called the antifluorite structure, anions and cations are swapped, such as beryllium carbide (Be2C) or lithium oxide (Li2O), potassium sulfate Potassium sulfate (US) or potassium sulphate (UK), also called sulphate of potash (SOP), arcanite, or archaically potash of sulfur, is the inorganic compound with formula K2SO4, a white water-soluble solid. It is commonly used in fertilizers, prov ... (K2SO4). Other anti-structures include: * anti- SnO2: Ti2N * anti- PbCl2: Co2P * anti- CdCl2: Co2N * anti- CdI2: Cs2O * anti- NbS2: Hf2S * anti- ReO3: Cu3N * anti- LaF3: Cu3P, Cu3As Refer ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Magnesium Silicide
Magnesium silicide, Mg2Si, is an inorganic compound consisting of magnesium and silicon. As-grown Mg2Si usually forms black crystals; they are semiconductors with n-type conductivity and have potential applications in thermoelectric generators. Crystal structure Mg2Si crystallizes in the antifluorite structure. In the Cubic crystal system, face-centered cubic lattice Si centers occupy the corners and face-centered positions of the unit cell and Mg centers occupy eight tetrahedral sites in the interior of the unit cell. The coordination numbers of Si and Mg are eight and four, respectively. Synthesis It can be produced by heating silicon dioxide, SiO2, found in sand, with excess magnesium. The process first forms silicon metal and magnesium oxide, and, if an excess of SiO2 is used, then elemental silicon is formed: :2 Mg + SiO2 → 2 MgO + Si If an excess of Mg is present, Mg2Si is formed from the reaction of the remaining magnesium with the silicon: :2 Mg + Si → Mg ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Silicide
A silicide is a type of chemical compound that combines silicon and a usually more electropositive element. Silicon is more electropositive than carbon. In terms of their physical properties, silicides are structurally closer to borides than to carbides. Because of size differences however silicides are not isostructural with borides and carbides. Bonds in silicides range from conductive metal-like structures to covalent or ionic. Silicides of all non-transition metals have been described except beryllium. Silicides are used in interconnects. Structure Silicon atoms in silicides can have many possible organizations: *Isolated silicon atoms: electrically conductive (or semiconductive) CrSi, MnSi, FeSi, CoSi, , , , , , *Si2 pairs: , hafnium and thorium silicides *Si4 tetrahedra: KSi, RbSi, CsSi *Si''n'' chains: USi, , CaSi, SrSi, YSi *Planar hexagonal graphite-like Si layers: β-USi2, silicides of other lanthanoids and actinoids *Corrugated hexagonal Si layers: C ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Uranium Dioxide
Uranium dioxide or uranium(IV) oxide (), also known as urania or uranous oxide, is an oxide of uranium, and is a black, radioactive, crystalline powder that naturally occurs in the mineral uraninite. It is used in nuclear fuel rods in nuclear reactors. A mixture of uranium and plutonium dioxides is used as MOX fuel. It has been used as an orange, yellow, green, and black color in ceramic glazes and glass. Production Uranium dioxide is produced by reducing uranium trioxide with hydrogen. This reaction often creates triuranium octoxide as an intermediate. :UO3 + H2 → UO2 + H2O at 700 °C (973 K) This reaction plays an important part in the creation of nuclear fuel through nuclear reprocessing and uranium enrichment. Chemistry Structure The solid is isostructural with (has the same structure as) fluorite ( calcium fluoride), where each U is surrounded by eight O nearest neighbors in a cubic arrangement. In addition, the dioxides of cerium, thorium, and the tran ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cubic Zirconia
Cubic zirconia (CZ) is the cubic crystalline form of zirconium dioxide (ZrO2). The synthesized material is hard and usually colorless, but may be made in a variety of different colors. It should not be confused with zircon, which is a zirconium silicate (ZrSiO4). It is sometimes erroneously called ''cubic zirconium''. Because of its low cost, durability, and close visual likeness to diamond, synthetic cubic zirconia has remained the most gemologically and economically important competitor for diamonds since commercial production began in 1976. Its main competitor as a synthetic gemstone is a more recently cultivated material, synthetic moissanite. Technical aspects Cubic zirconia is crystallographically isometric, an important attribute of a would-be diamond simulant. During synthesis zirconium oxide naturally forms monoclinic crystals, which are stable under normal atmospheric conditions. A stabilizer is required for cubic crystals (taking on the fluorite structure) ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cubic Minerals
Cubic may refer to: Science and mathematics * Cube (algebra) In arithmetic and algebra, the cube of a number is its third exponentiation, power, that is, the result of multiplying three instances of together. The cube of a number is denoted , using a superscript 3, for example . The cube Mathematical ..., "cubic" measurement * Cube, a three-dimensional solid object bounded by six square faces, facets or sides, with three meeting at each vertex ** Cubic crystal system, a crystal system where the unit cell is in the shape of a cube * Cubic function, a polynomial function of degree three * Cubic equation, a polynomial equation (reducible to ''ax''3 + ''bx''2 + ''cx'' + ''d'' = 0) * Cubic form, a homogeneous polynomial of degree 3 * Cubic graph (mathematics - graph theory), a graph where all vertices have degree 3 * Cubic plane curve (mathematics), a plane algebraic curve ''C'' defined by a cubic equation * Cubic reciprocity (mathematics - number theory), a theorem ana ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Minerals In Space Group 225
In geology and mineralogy, a mineral or mineral species is, broadly speaking, a solid substance with a fairly well-defined chemical composition and a specific crystal structure that occurs naturally in pure form.John P. Rafferty, ed. (2011): Minerals'; p. 1. In the series ''Geology: Landforms, Minerals, and Rocks''. Rosen Publishing Group. The geological definition of mineral normally excludes compounds that occur only in living organisms. However, some minerals are often biogenic (such as calcite) or organic compounds in the sense of chemistry (such as mellite). Moreover, living organisms often synthesize inorganic minerals (such as hydroxylapatite) that also occur in rocks. The concept of mineral is distinct from rock, which is any bulk solid geologic material that is relatively homogeneous at a large enough scale. A rock may consist of one type of mineral or may be an aggregate of two or more different types of minerals, spacially segregated into distinct phases. Som ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluorite
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon. The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 4 as fluorite. Pure fluorite is colourless and transparent, both in visible and ultraviolet light, but impurities usually make it a colorful mineral and the stone has ornamental and lapidary uses. Industrially, fluorite is used as a flux for smelting, and in the production of certain glasses and enamels. The purest grades of fluorite are a source of fluoride for hydrofluoric acid manufacture, which is the intermediate source of most fluorine-containing fine chemicals. Optically clear transparent fluorite has anomalous partial dispersion, that is, its refractive index varies with the wavelength of light in a manner that differs from that of commonly used glasses, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Fluorine Minerals
Fluorine is a chemical element; it has symbol F and atomic number 9. It is the lightest halogen and exists at standard conditions as pale yellow diatomic gas. Fluorine is extremely reactive as it reacts with all other elements except for the light noble gases. It is highly toxic. Among the elements, fluorine ranks 24th in cosmic abundance and 13th in crustal abundance. Fluorite, the primary mineral source of fluorine, which gave the element its name, was first described in 1529; as it was added to metal ores to lower their melting points for smelting, the Latin verb meaning gave the mineral its name. Proposed as an element in 1810, fluorine proved difficult and dangerous to separate from its compounds, and several early experimenters died or sustained injuries from their attempts. Only in 1886 did French chemist Henri Moissan isolate elemental fluorine using low-temperature electrolysis, a process still employed for modern production. Industrial production of fluorine gas fo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |