|
Cysteine Metabolism
Cysteine metabolism refers to the biological pathways that consume or create cysteine. The pathways of different amino acids and other metabolites interweave and overlap to creating complex systems. Human cysteine metabolism In human cysteine metabolism, L-cysteine is consumed in several ways as shown below. L-Cysteine is also consumed in pantothenate/coenzyme A biosynthesis. L-Cysteine is the product of several processes as well. In addition to the reactions below, L-cysteine is also a product of glycine, serine, and threonine Threonine (symbol Thr or T) is an amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated −NH form when dissolved in water), a carboxyl group (which is in the deprotonated −COO− ... metabolism. References See also * D-cysteine desulfhydrase * Sulfur metabolism {{DEFAULTSORT:Cysteine Metabolism Alpha-Amino acids Sulfur metabolism Sulfur amino acids ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cysteine
Cysteine (; symbol Cys or C) is a semiessential proteinogenic amino acid with the chemical formula, formula . The thiol side chain in cysteine enables the formation of Disulfide, disulfide bonds, and often participates in enzymatic reactions as a nucleophile. Cysteine is chiral, but both D and L-cysteine are found in nature. LCysteine is a protein monomer in all biota, and D-cysteine acts as a signaling molecule in mammalian nervous systems. Cysteine is named after its discovery in urine, which comes from the urinary bladder or cyst, from Ancient Greek, Greek κύστις ''kýstis'', "bladder". The thiol is susceptible to oxidation to give the disulfide bond, disulfide derivative cystine, which serves an important structural role in many proteins. In this case, the symbol Cyx is sometimes used. The deprotonated form can generally be described by the symbol Cym as well. When used as a food additive, cysteine has the E number E920. Cysteine is Genetic code, encoded by the codo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
3-mercapto-pyruvate
3-Mercaptopyruvic acid is an intermediate in cysteine metabolism. It has been studied as a potential treatment for cyanide poisoning, but its half-life is too short for it to be clinically effective. Instead, prodrug A prodrug is a pharmacologically inactive medication or compound that, after intake, is metabolized (i.e., converted within the body) into a pharmacologically active drug. Instead of administering a drug directly, a corresponding prodrug can be ...s, such as sulfanegen, are being evaluated to compensate for the short half-life of 3-mercaptopyruvic acid. See also * 3-mercaptopyruvate sulfurtransferase References Carboxylic acids Thiols Alpha-keto acids {{OrganicAcid-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
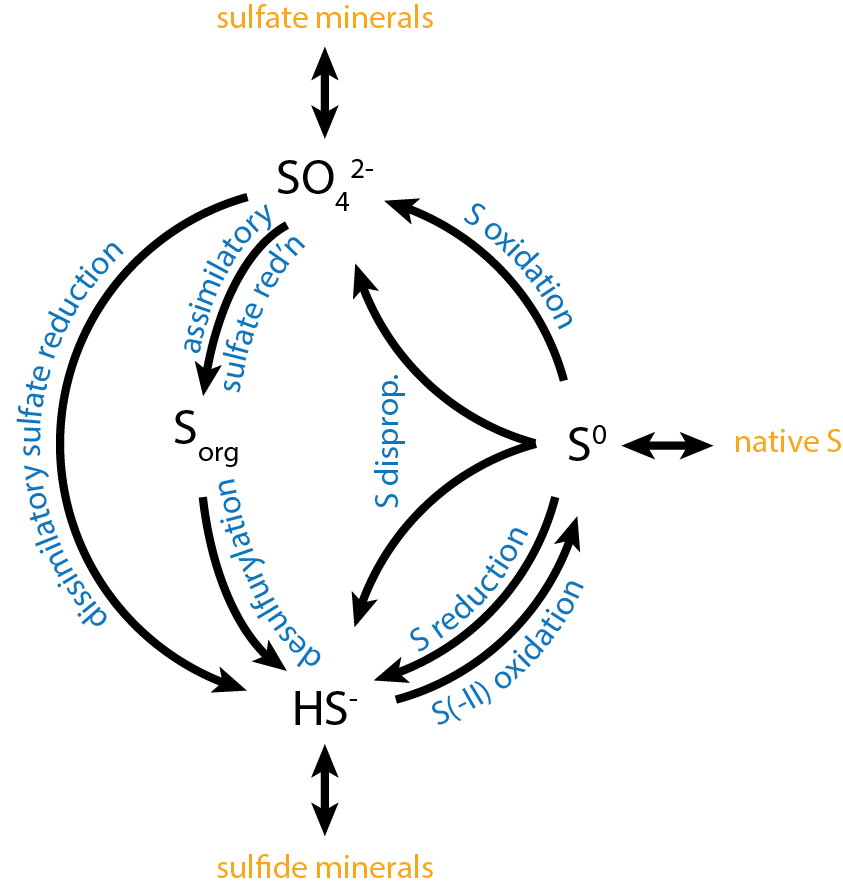
Sulfur Metabolism
Sulfur is metabolized by all organisms, from bacteria and archaea to plants and animals. Sulfur can have an oxidation state from −2 to +6 and is reduced or oxidized by a diverse range of organisms. The element is present in proteins, sulfate esters of polysaccharides, steroids, phenols, and sulfur-containing coenzymes. Oxidation Reduced sulfur compounds are oxidized by most organisms, including higher animals and higher plants. Some organisms can conserve energy (i.e., produce ATP) from the oxidation of sulfur and it can serve as the sole energy source for some lithotrophic bacteria and archaea. Sulfur oxidizers use enzymes such as Sulfide:quinone reductase, sulfur dioxygenase and sulfite oxidase to oxidize sulfur compounds to sulfate. Sulfur-oxidizing microorganisms Reduced sulfur compounds, such as hydrogen sulfide, elemental sulfur, sulfite, thiosulfate, and various polythionates (e.g., tetrathionate), are oxidized by chemotrophic, phototrophic, and mixotrophic bacteria for ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
D-cysteine Desulfhydrase
The enzyme D-cysteine desulfhydrase (EC 4.4.1.15) catalyzes the chemical reaction :D-cysteine + H2O \rightleftharpoons sulfide + NH3 + pyruvate This enzyme belongs to the family of lyases, specifically the class of carbon-sulfur lyases. The systematic name of this enzyme class is D-cysteine sulfide-lyase (deaminating; pyruvate-forming). Other names in common use include D-cysteine lyase, and D-cysteine sulfide-lyase (deaminating). This enzyme participates in cysteine metabolism Cysteine metabolism refers to the biological pathways that consume or create cysteine. The pathways of different amino acids and other metabolites interweave and overlap to creating complex systems. Human cysteine metabolism In human cysteine metab .... References * * * EC 4.4.1 Enzymes of unknown structure {{lyase-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cystathionine
Cystathionine is an intermediate in the synthesis of cysteine from homocysteine. It is produced by the transsulfuration pathway and is converted into cysteine by cystathionine gamma-lyase (CTH). Biosynthetically, cystathionine is generated from homocysteine and serine by cystathionine beta synthase (upper reaction in the diagram below). It is then cleaved into cysteine and α-ketobutyrate by cystathionine gamma-lyase (lower reaction). An excess of cystathionine in the urine is called cystathioninuria. Cysteine dioxygenase (CDO), and sulfinoalanine decarboxylase can turn cysteine into hypotaurine and then taurine. Alternately, the cysteine from the cystathionine gamma-lyase can be used by the enzymes glutamate–cysteine ligase (GCL) and glutathione synthetase (GSS) to produce glutathione Glutathione (GSH, ) is an organic compound with the chemical formula . It is an antioxidant in plants, animals, fungi, and some bacteria and archaea. Glutathione is capable of preventi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cysteine Synthase
In enzymology, a cysteine synthase () is an enzyme that catalyzes the chemical reaction :''O''3-acetyl-L-serine + hydrogen sulfide \rightleftharpoons L-cysteine + acetate Thus, the two substrates of this enzyme are ''O''3-acetyl-L-serine and hydrogen sulfide, whereas its two products are L-cysteine and acetate. This enzyme belongs to the family of transferases, specifically those transferring aryl or alkyl groups other than methyl groups. The systematic name of this enzyme class is O3-acetyl-L-serine:hydrogen-sulfide 2-amino-2-carboxyethyltransferase. Other names in common use include O-acetyl-L-serine sulfhydrylase, O-acetyl-L-serine sulfohydrolase, O-acetylserine (thiol)-lyase, O-acetylserine (thiol)-lyase A, O-acetylserine sulfhydrylase, O3-acetyl-L-serine acetate-lyase (adding hydrogen-sulfide), acetylserine sulfhydrylase, cysteine synthetase, S-sulfocysteine synthase, 3-O-acetyl-L-serine:hydrogen-sulfide, and 2-amino-2-carboxyethyltransferase. This enzyme participates i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
O-Acetylserine
''O''-Acetylserine is an α-amino acid with the chemical formula HO2CCH(NH2)CH2OC(O)CH3. It is an intermediate in the biosynthesis of the common amino acid cysteine in bacteria and plants. ''O''-Acetylserine is biosynthesized by acetylation of the serine by the enzyme An enzyme () is a protein that acts as a biological catalyst by accelerating chemical reactions. The molecules upon which enzymes may act are called substrate (chemistry), substrates, and the enzyme converts the substrates into different mol ... serine transacetylase. The enzyme ''O''-acetylserine (thiol)-lyase, using sulfide sources, converts this ester into cysteine, releasing acetate: :HO2CCH(NH2)CH2OH → HO2CCH(NH2)CH2OC(O)CH3 :HO2CCH(NH2)CH2OC(O)CH3 → HO2CCH(NH2)CH2SH References {{DEFAULTSORT:Acetyserine, O- Alpha-Amino acids Acetate esters ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Threonine
Threonine (symbol Thr or T) is an amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated −NH form when dissolved in water), a carboxyl group (which is in the deprotonated −COO− form when dissolved in water), and a side chain containing a hydroxyl group, making it a polar, uncharged amino acid. It is essential in humans, meaning the body cannot synthesize it: it must be obtained from the diet. Threonine is synthesized from aspartate in bacteria such as ''E. coli''. It is encoded by all the codons starting AC (ACU, ACC, ACA, and ACG). Threonine sidechains are often hydrogen bonded; the most common small motifs formed are based on interactions with serine: ST turns, ST motifs (often at the beginning of alpha helices) and ST staples (usually at the middle of alpha helices). Modifications The threonine residue is susceptible to numerous posttranslational modifications. The hydroxyl side-chain can und ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Serine
Serine (symbol Ser or S) is an α-amino acid that is used in the biosynthesis of proteins. It contains an α- amino group (which is in the protonated − form under biological conditions), a carboxyl group (which is in the deprotonated − form under biological conditions), and a side chain consisting of a hydroxymethyl group, classifying it as a polar amino acid. It can be synthesized in the human body under normal physiological circumstances, making it a nonessential amino acid. It is encoded by the codons UCU, UCC, UCA, UCG, AGU and AGC. Occurrence This compound is one of the proteinogenic amino acids. Only the L- stereoisomer appears naturally in proteins. It is not essential to the human diet, since it is synthesized in the body from other metabolites, including glycine. Serine was first obtained from silk protein, a particularly rich source, in 1865 by Emil Cramer. Its name is derived from the Latin for silk, '' sericum''. Serine's structure was established in ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glycine
Glycine (symbol Gly or G; ) is an amino acid that has a single hydrogen atom as its side chain. It is the simplest stable amino acid. Glycine is one of the proteinogenic amino acids. It is encoded by all the codons starting with GG (GGU, GGC, GGA, GGG). Glycine disrupts the formation of alpha-helices in secondary protein structure. Its small side chain causes it to favor random coils instead. Glycine is also an inhibitory neurotransmitter – interference with its release within the spinal cord (such as during a '' Clostridium tetani'' infection) can cause spastic paralysis due to uninhibited muscle contraction. It is the only achiral proteinogenic amino acid. It can fit into both hydrophilic and hydrophobic environments, due to its minimal side chain of only one hydrogen atom. History and etymology Glycine was discovered in 1820 by French chemist Henri Braconnot when he hydrolyzed gelatin by boiling it with sulfuric acid. He originally called it "sugar of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Glutamate–cysteine Ligase
Glutamate–cysteine ligase (GCL) ), previously known as γ-glutamylcysteine synthetase (GCS), is the first enzyme of the cellular glutathione (GSH) biosynthetic pathway that catalyzes the chemical reaction: L-glutamate + L-cysteine + ATP \rightleftharpoons γ-glutamyl cysteine + ADP + Pi GSH, and by extension GCL, is critical to cell survival. Nearly every eukaryotic cell, from plants to yeast to humans, expresses a form of the GCL protein for the purpose of synthesizing GSH. To further highlight the critical nature of this enzyme, genetic knockout of GCL results in embryonic lethality. Furthermore, dysregulation of GCL enzymatic function and activity is known to be involved in the vast majority of human diseases, such as diabetes, Parkinson's disease, Alzheimer's disease, COPD, HIV/AIDS, and cancer. This typically involves impaired function leading to decreased GSH biosynthesis, reduced cellular antioxidant capacity, and the induction of oxidative stress. However, in cancer, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |