|
Colligative
In chemistry, colligative properties are those properties of solutions that depend on the ratio of the number of solute particles to the number of solvent particles in a solution, and not on the nature of the chemical species present. The number ratio can be related to the various units for concentration of a solution such as molarity, molality, normality (chemistry), etc. The assumption that solution properties are independent of nature of solute particles is exact only for ideal solutions, which are solutions that exhibit thermodynamic properties analogous to those of an ideal gas, and is approximate for dilute real solutions. In other words, colligative properties are a set of solution properties that can be reasonably approximated by the assumption that the solution is ideal. Only properties which result from the dissolution of a nonvolatile solute in a volatile liquid solvent are considered.KL Kapoor ''Applications of Thermodynamics'' Volume 3 They are essentially solvent p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boiling Point Elevation
Boiling-point elevation describes the phenomenon that the boiling point of a liquid (a solvent) will be higher when another compound is added, meaning that a solution has a higher boiling point than a pure solvent. This happens whenever a non-volatile solute, such as a salt, is added to a pure solvent, such as water. The boiling point can be measured accurately using an ebullioscope. Explanation The ''boiling point elevation'' is a colligative property, which means that it is dependent on the presence of dissolved particles and their number, but not their identity. It is an effect of the dilution of the solvent in the presence of a solute. It is a phenomenon that happens for all solutes in all solutions, even in ideal solutions, and does not depend on any specific solute–solvent interactions. The boiling point elevation happens both when the solute is an electrolyte, such as various salts, and a nonelectrolyte. In thermodynamic terms, the origin of the boiling point elevati ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Freezing-point Depression
Freezing-point depression is a drop in the minimum temperature at which a substance freezes, caused when a smaller amount of another, non- volatile substance is added. Examples include adding salt into water (used in ice cream makers and for de-icing roads), alcohol in water, ethylene or propylene glycol in water (used in antifreeze in cars), adding copper to molten silver (used to make solder that flows at a lower temperature than the silver pieces being joined), or the mixing of two solids such as impurities into a finely powdered drug. In all cases, the substance added/present in smaller amounts is considered the solute, while the original substance present in larger quantity is thought of as the solvent. The resulting liquid solution or solid-solid mixture has a lower freezing point than the pure solvent or solid because the chemical potential of the solvent in the mixture is lower than that of the pure solvent, the difference between the two being proportional to t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ideal Solution
In chemistry, an ideal solution or ideal mixture is a solution that exhibits thermodynamic properties analogous to those of a mixture of ideal gases. The enthalpy of mixing is zero as is the volume change on mixing by definition; the closer to zero the enthalpy of mixing is, the more "ideal" the behavior of the solution becomes. The vapor pressures of the solvent and solute obey Raoult's law and Henry's law, respectively, and the activity coefficient (which measures deviation from ideality) is equal to one for each component. The concept of an ideal solution is fundamental to chemical thermodynamics and its applications, such as the explanation of colligative properties. Physical origin Ideality of solutions is analogous to ideality for gases, with the important difference that intermolecular interactions in liquids are strong and cannot simply be neglected as they can for ideal gases. Instead we assume that the mean strength of the interactions are the same between all th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Solution (chemistry)
In chemistry, a solution is a special type of homogeneous mixture composed of two or more substances. In such a mixture, a solute is a substance dissolved in another substance, known as a solvent. If the attractive forces between the solvent and solute particles are greater than the attractive forces holding the solute particles together, the solvent particles pull the solute particles apart and surround them. These surrounded solute particles then move away from the solid solute and out into the solution. The mixing process of a solution happens at a scale where the effects of chemical polarity are involved, resulting in interactions that are specific to solvation. The solution usually has the state of the solvent when the solvent is the larger fraction of the mixture, as is commonly the case. One important parameter of a solution is the concentration, which is a measure of the amount of solute in a given amount of solution or solvent. The term " aqueous solution" is used ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Osmotic Pressure
Osmotic pressure is the minimum pressure which needs to be applied to a solution to prevent the inward flow of its pure solvent across a semipermeable membrane. It is also defined as the measure of the tendency of a solution to take in a pure solvent by osmosis. Potential osmotic pressure is the maximum osmotic pressure that could develop in a solution if it were separated from its pure solvent by a semipermeable membrane. Osmosis occurs when two solutions containing different concentrations of solute are separated by a selectively permeable membrane. Solvent molecules pass preferentially through the membrane from the low-concentration solution to the solution with higher solute concentration. The transfer of solvent molecules will continue until equilibrium is attained. Theory and measurement Jacobus van 't Hoff found a quantitative relationship between osmotic pressure and solute concentration, expressed in the following equation: :\Pi = icRT where \Pi is osmotic ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molar Mass
In chemistry, the molar mass of a chemical compound is defined as the mass of a sample of that compound divided by the amount of substance which is the number of moles in that sample, measured in moles. The molar mass is a bulk, not molecular, property of a substance. The molar mass is an ''average'' of many instances of the compound, which often vary in mass due to the presence of isotopes. Most commonly, the molar mass is computed from the standard atomic weights and is thus a terrestrial average and a function of the relative abundance of the isotopes of the constituent atoms on Earth. The molar mass is appropriate for converting between the mass of a substance and the amount of a substance for bulk quantities. The molecular mass and formula mass are commonly used as a synonym of molar mass, particularly for molecular compounds; however, the most authoritative sources define it differently. The difference is that molecular mass is the mass of one specific particle or m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Van 't Hoff Factor
The van 't Hoff factor (named after Dutch chemist Jacobus Henricus van 't Hoff) is a measure of the effect of a solute on colligative properties such as osmotic pressure, relative lowering in vapor pressure, boiling-point elevation and freezing-point depression. The van 't Hoff factor is the ratio between the actual concentration of particles produced when the substance is dissolved and the concentration of a substance as calculated from its mass. For most non- electrolytes dissolved in water, the van 't Hoff factor is essentially 1. For most ionic compounds dissolved in water, the van 't Hoff factor is equal to the number of discrete ions in a formula unit of the substance. This is true for ideal solutions only, as occasionally ion pairing occurs in solution. At a given instant a small percentage of the ions are paired and count as a single particle. Ion pairing occurs to some extent in all electrolyte solutions. This causes the measured van 't Hoff factor t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molality
Molality is a measure of the number of moles of solute in a solution corresponding to 1 kg or 1000 g of solvent. This contrasts with the definition of molarity which is based on a specified volume of solution. A commonly used unit for molality in chemistry is mol/ kg. A solution of concentration 1 mol/kg is also sometimes denoted as ''1 molal''. The unit ''mol/kg'' requires that molar mass be expressed in ''kg/mol'', instead of the usual ''g/mol'' or ''kg/kmol''. Definition The molality (''b''), of a solution is defined as the amount of substance (in moles) of solute, ''n''solute, divided by the mass (in kg) of the solvent, ''m''solvent: :b = \frac In the case of solutions with more than one solvent, molality can be defined for the mixed solvent considered as a pure pseudo-solvent. Instead of mole solute per kilogram solvent as in the binary case, units are defined as mole solute per kilogram mixed solvent. Origin The term ''molality'' is formed in analogy to ''mo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
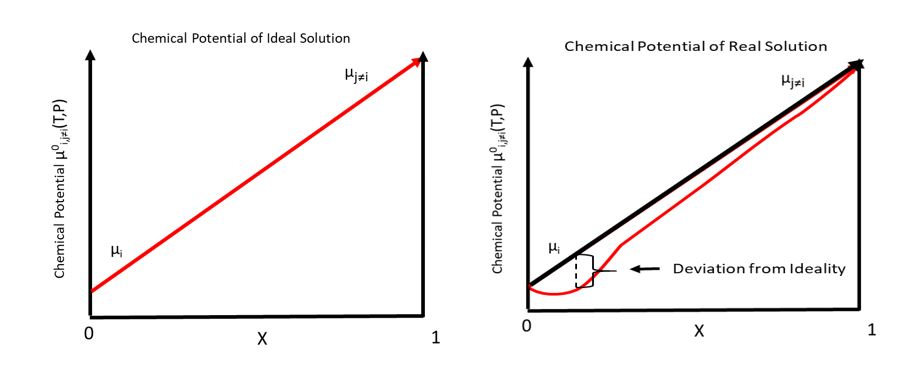
Chemical Potential
In thermodynamics, the chemical potential of a species is the energy that can be absorbed or released due to a change of the particle number of the given species, e.g. in a chemical reaction or phase transition. The chemical potential of a species in a mixture is defined as the rate of change of free energy of a thermodynamic system with respect to the change in the number of atoms or molecules of the species that are added to the system. Thus, it is the partial derivative of the free energy with respect to the amount of the species, all other species' concentrations in the mixture remaining constant. When both temperature and pressure are held constant, and the number of particles is expressed in moles, the chemical potential is the partial molar Gibbs free energy. At chemical equilibrium or in phase equilibrium, the total sum of the product of chemical potentials and stoichiometric coefficients is zero, as the free energy is at a minimum. In a system in diffusion equilibrium, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Dissociation (chemistry)
Dissociation in chemistry is a general process in which molecules (or ionic compounds such as salts, or complexes) separate or split into other things such as atoms, ions, or radicals, usually in a reversible manner. For instance, when an acid dissolves in water, a covalent bond between an electronegative atom and a hydrogen atom is broken by heterolytic fission, which gives a proton (H+) and a negative ion. Dissociation is the opposite of association or recombination. Dissociation constant For reversible dissociations in a chemical equilibrium :AB A + B the dissociation constant ''K''d is the ratio of dissociated to undissociated compound :K_d = \mathrm where the brackets denote the equilibrium concentrations of the species. Dissociation degree The dissociation degree \alpha is the fraction of original solute molecules that have dissociated. It is usually indicated by the Greek symbol α. More accurately, degree of dissociation refers to the amount of solute dissoci ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mole Fraction
In chemistry, the mole fraction or molar fraction (''xi'' or ) is defined as unit of the amount of a constituent (expressed in moles), ''ni'', divided by the total amount of all constituents in a mixture (also expressed in moles), ''n''tot. This expression is given below: :x_i = \frac The sum of all the mole fractions is equal to 1: :\sum_^ n_i = n_\mathrm ; \ \sum_^ x_i = 1. The same concept expressed with a denominator of 100 is the mole percent, molar percentage or molar proportion (mol%). The mole fraction is also called the amount fraction. It is identical to the number fraction, which is defined as the number of molecules of a constituent ''Ni'' divided by the total number of all molecules ''N''tot. The mole fraction is sometimes denoted by the lowercase Greek letter (chi) instead of a Roman ''x''. For mixtures of gases, IUPAC recommends the letter ''y''. The National Institute of Standards and Technology of the United States prefers the term amount-of-substance fract ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |