|
Bismuth(III) Oxide
Bismuth(III) oxide is a compound of bismuth, and a common starting point for bismuth chemistry. It is found naturally as the mineral bismite (monoclinic) and sphaerobismoite (tetragonal, much more rare), but it is usually obtained as a by-product of the smelting of copper and lead ores. Dibismuth trioxide is commonly used to produce the " Dragon's eggs" effect in fireworks, as a replacement of red lead. Structure The structures adopted by differ substantially from those of arsenic(III) oxide, , and antimony(III) oxide, .Wells, A.F. (1984) ''Structural Inorganic Chemistry''. 5th. London, England: Oxford University Press. p.890 Bismuth oxide, has five crystallographic polymorphs. The room temperature phase, α- has a monoclinic crystal structure. There are three high temperature phases, a tetragonal β-phase, a body-centred cubic γ-phase, a cubic δ- phase and an ε-phase. The room temperature α-phase has a complex structure with layers of oxygen atoms with layers ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Bismite
Bismite is a bismuth oxide mineral, bismuth trioxide or Bi2O3. It is a monoclinic mineral, but the typical form of occurrence is massive and clay-like with no macroscopic crystals. The color varies from green to yellow. It has a Mohs hardness of 4 to 5 and a specific gravity of 8.5 to 9.5, quite high for a nonmetallic mineral. Bismite is a secondary oxidation zone mineral which forms from primary bismuth minerals. It was first described from Goldfield, Nevada in 1868, and later from the Schneeberg District, Ore Mountains, Saxony, Germany. See also * Bismuth trioxide – details on the chemistry of this substance * Bismutite * Bismuthinite * Bismoclite * Eulytine References Mindat localities Bismuth minerals Oxide minerals Goldfield, Nevada Monoclinic minerals Minerals in space group 14 {{oxide-mineral-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Red Lead
Red is the color at the long wavelength end of the visible spectrum of light, next to orange and opposite violet. It has a dominant wavelength of approximately 625–750 nanometres. It is a primary color in the RGB color model and a secondary color (made from magenta and yellow) in the CMYK color model, and is the complementary color of cyan. Reds range from the brilliant yellow-tinged scarlet and vermillion to bluish-red crimson, and vary in shade from the pale red pink to the dark red burgundy. Red pigment made from ochre was one of the first colors used in prehistoric art. The Ancient Egyptians and Mayans colored their faces red in ceremonies; Roman generals had their bodies colored red to celebrate victories. It was also an important color in China, where it was used to color early pottery and later the gates and walls of palaces. In the Renaissance, the brilliant red costumes for the nobility and wealthy were dyed with kermes and cochineal. The 19th century brought ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Vacancy Defect
In crystallography, a vacancy is a type of point defect in a crystal where an atom is missing from one of the lattice sites.Ehrhart, P. (1991) "Properties and interactions of atomic defects in metals and alloys", chapter 2, p. 88 in ''Landolt-Börnstein, New Series III'', Vol. 25, Springer, Berlin Crystals inherently possess imperfections, sometimes referred to as crystallographic defects. Vacancies occur naturally in all crystalline materials. At any given temperature, up to the melting point of the material, there is an equilibrium concentration (ratio of vacant lattice sites to those containing atoms). At the melting point of some metals the ratio can be approximately 1:1000. This temperature dependence can be modelled by :N_ = N \exp\left(\frac\right) where is the vacancy concentration, is the energy required for vacancy formation, is the Boltzmann constant, is the absolute temperature, and is the concentration of atomic sites i.e. : N = \frac where is density, t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Phases Of Matter
In the outline of physical science, physical sciences, a phase is a region of material that is chemically uniform, physically distinct, and (often) mechanically separable. In a system consisting of ice and water in a glass jar, the ice cubes are one phase, the water is a second phase, and the humid air is a third phase over the ice and water. The glass of the jar is a different material, in its own separate phase. (See .) More precisely, a phase is a region of space (a thermodynamic system), throughout which all physical properties of a material are essentially uniform. Examples of physical properties include density, refractive index, index of refraction, magnetization and chemical composition. The term ''phase'' is sometimes used as a synonym for state of matter, but there can be several Miscibility, immiscible phases of the same state of matter (as where oil and water separate into distinct phases, both in the liquid state). Types of phases Distinct phases may be des ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Space Group
In mathematics, physics and chemistry, a space group is the symmetry group of a repeating pattern in space, usually in three dimensions. The elements of a space group (its symmetry operations) are the rigid transformations of the pattern that leave it unchanged. In three dimensions, space groups are classified into 219 distinct types, or 230 types if chiral copies are considered distinct. Space groups are discrete cocompact groups of isometries of an oriented Euclidean space in any number of dimensions. In dimensions other than 3, they are sometimes called Bieberbach groups. In crystallography, space groups are also called the crystallographic or Fedorov groups, and represent a description of the symmetry of the crystal. A definitive source regarding 3-dimensional space groups is the ''International Tables for Crystallography'' . History Space groups in 2 dimensions are the 17 wallpaper groups which have been known for several centuries, though the proof that the list ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chiral
Chirality () is a property of asymmetry important in several branches of science. The word ''chirality'' is derived from the Greek language, Greek (''kheir''), "hand", a familiar chiral object. An object or a system is ''chiral'' if it is distinguishable from its mirror image; that is, it cannot be wikt:superpose, superposed (not to be confused with wikt:superimpose, superimposed) onto it. Conversely, a mirror image of an ''achiral'' object, such as a sphere, cannot be distinguished from the object. A chiral object and its mirror image are called ''enantiomorphs'' (Greek, "opposite forms") or, when referring to molecules, ''enantiomers''. A non-chiral object is called ''achiral'' (sometimes also ''amphichiral'') and can be superposed on its mirror image. The term was first used by Lord Kelvin in 1893 in the second Robert Boyle Lecture at the Oxford University Junior Scientific Club which was published in 1894: Human hands are perhaps the most recognized example of chirality ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
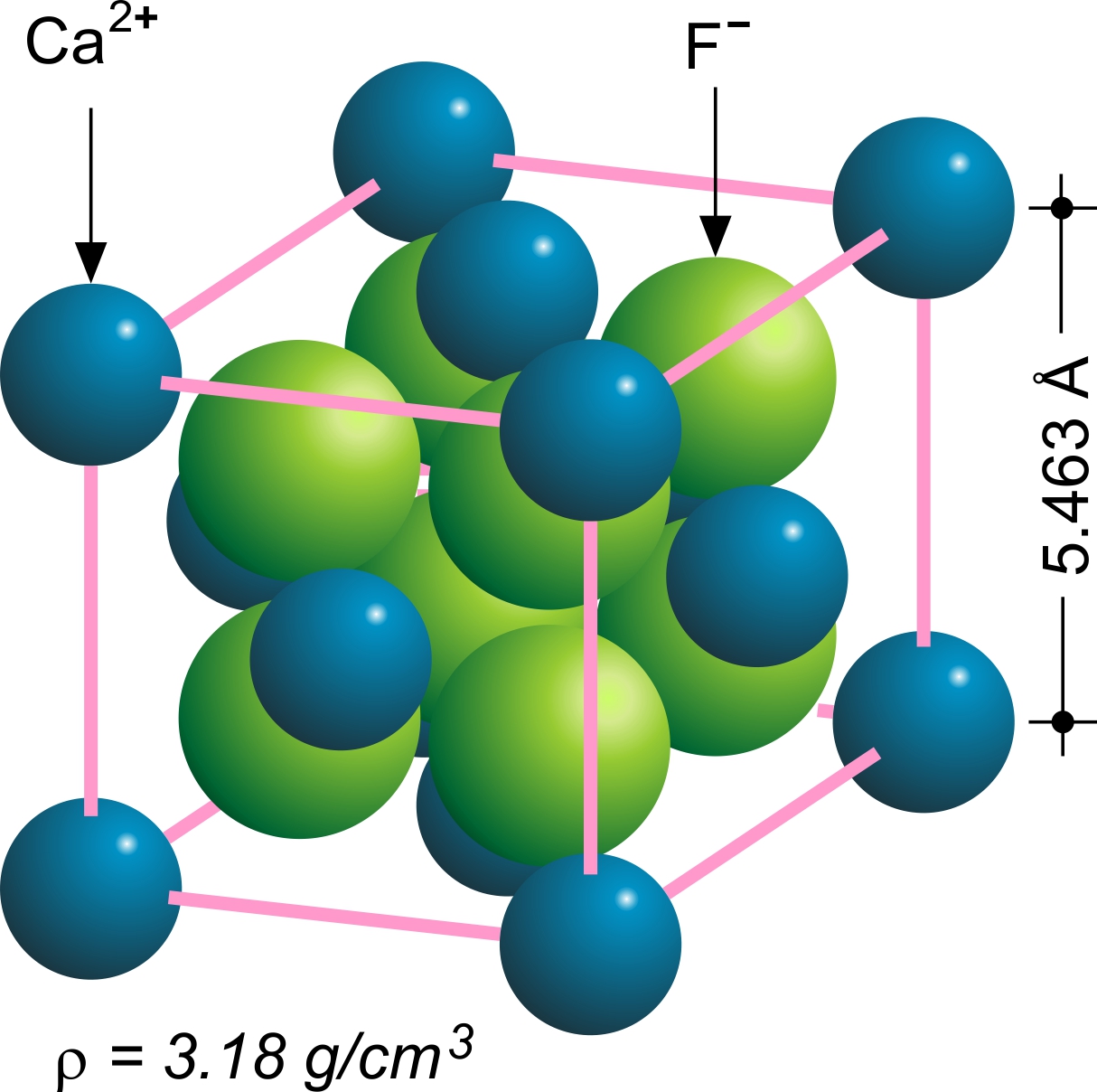
Fluorite
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon. The Mohs scale of mineral hardness, based on scratch hardness comparison, defines value 4 as fluorite. Pure fluorite is colourless and transparent, both in visible and ultraviolet light, but impurities usually make it a colorful mineral and the stone has ornamental and lapidary uses. Industrially, fluorite is used as a flux for smelting, and in the production of certain glasses and enamels. The purest grades of fluorite are a source of fluoride for hydrofluoric acid manufacture, which is the intermediate source of most fluorine-containing fine chemicals. Optically clear transparent fluorite has anomalous partial dispersion, that is, its refractive index varies with the wavelength of light in a manner that differs from that of commonly used glasses, ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Cubic Crystal System
In crystallography, the cubic (or isometric) crystal system is a crystal system where the unit cell is in the shape of a cube. This is one of the most common and simplest shapes found in crystals and minerals. There are three main varieties of these crystals: *Primitive cubic (abbreviated ''cP'' and alternatively called simple cubic) *Body-centered cubic (abbreviated ''cI'' or bcc) *Face-centered cubic (abbreviated ''cF'' or fcc) Note: the term fcc is often used in synonym for the ''cubic close-packed'' or ccp structure occurring in metals. However, fcc stands for a face-centered cubic Bravais lattice, which is not necessarily close-packed when a motif is set onto the lattice points. E.g. the diamond and the zincblende lattices are fcc but not close-packed. Each is subdivided into other variants listed below. Although the ''unit cells'' in these crystals are conventionally taken to be cubes, the primitive unit cells often are not. Bravais lattices The three Bravais latices ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tetragonal
In crystallography, the tetragonal crystal system is one of the 7 crystal systems. Tetragonal crystal lattices result from stretching a cubic lattice along one of its lattice vectors, so that the Cube (geometry), cube becomes a rectangular Prism (geometry), prism with a square base (''a'' by ''a'') and height (''c'', which is different from ''a''). Bravais lattices There are two tetragonal Bravais lattices: the primitive tetragonal and the body-centered tetragonal. The body-centered tetragonal lattice is equivalent to the primitive tetragonal lattice with a smaller unit cell, while the face-centered tetragonal lattice is equivalent to the body-centered tetragonal lattice with a smaller unit cell. Crystal classes The point groups that fall under this crystal system are listed below, followed by their representations in international notation, Schoenflies notation, orbifold notation, Coxeter notation and mineral examples.Hurlbut, Cornelius S.; Klein, Cornelis, 1985, ''Ma ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
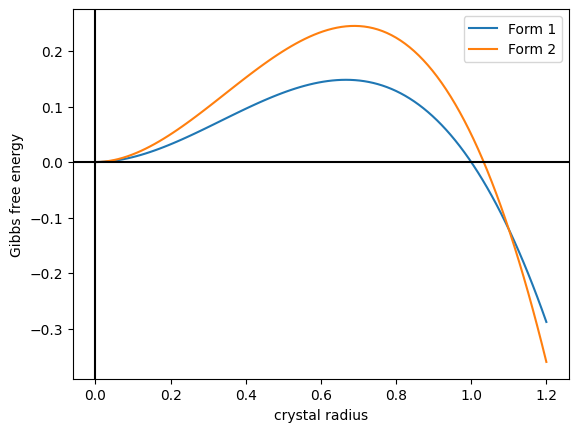
Polymorphism (materials Science)
In crystallography, polymorphism is the phenomenon where a compound or element can crystallize into more than one crystal structure. The preceding definition has evolved over many years and is still under discussion today. Discussion of the defining characteristics of polymorphism involves distinguishing among types of transitions and structural changes occurring in polymorphism versus those in other phenomena. Overview Phase transitions (phase changes) that help describe polymorphism include polymorphic transitions as well as melting and vaporization transitions. According to IUPAC, a polymorphic transition is "A reversible transition of a solid crystalline phase at a certain temperature and pressure (the inversion point) to another phase of the same chemical composition with a different crystal structure." Additionally, Walter McCrone described the phases in polymorphic matter as "different in crystal structure but identical in the liquid or vapor states." McCrone also def ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |