|
Biopharmaceutics Classification System
The Biopharmaceutics Classification System (BCS) is a system to differentiate drugs on the basis of their solubility and permeability. This system restricts the prediction using the parameters solubility and intestinal permeability. The solubility classification is based on a United States Pharmacopoeia (USP) aperture. The intestinal permeability classification is based on a comparison to the intravenous injection. All those factors are highly important because 85% of the most sold drugs in the United States and Europe Europe is a continent located entirely in the Northern Hemisphere and mostly in the Eastern Hemisphere. It is bordered by the Arctic Ocean to the north, the Atlantic Ocean to the west, the Mediterranean Sea to the south, and Asia to the east ... are orally administered. Classes According to the Biopharmaceutics Classification System (BCS) drug substances are classified to four classes upon their solubility and permeability: * Class I – high perme ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
|
Solubility
In chemistry, solubility is the ability of a chemical substance, substance, the solute, to form a solution (chemistry), solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution. The extent of the solubility of a substance in a specific solvent is generally measured as the concentration of the solute in a wikt:saturated#Chemistry, saturated solution, one in which no more solute can be dissolved. At this point, the two substances are said to be at the solubility equilibrium. For some solutes and solvents, there may be no such limit, in which case the two substances are said to be "miscibility, miscible in all proportions" (or just "miscible"). The solute can be a solid, a liquid, or a gas, while the solvent is usually solid or liquid. Both may be pure substances, or may themselves be solutions. Gases are always miscible in all proportions, except in very extreme situations,J. de Swaan Arons and G. A. ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
|
In Vivo
Studies that are ''in vivo'' (Latin for "within the living"; often not italicized in English) are those in which the effects of various biological entities are tested on whole, living organisms or cells, usually animals, including humans, and plants, as opposed to a tissue extract or dead organism. Examples of investigations ''in vivo'' include: the pathogenesis of disease by comparing the effects of bacterial infection with the effects of purified bacterial toxins; the development of non-antibiotics, antiviral drugs, and new drugs generally; and new surgical procedures. Consequently, animal testing and clinical trials are major elements of ''in vivo'' research. ''In vivo'' testing is often employed over ''in vitro'' because it is better suited for observing the overall effects of an experiment on a living subject. In drug discovery, for example, verification of efficacy ''in vivo'' is crucial, because ''in vitro'' assays can sometimes yield misleading results with drug c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
|
IVIVC
An in-vitro in-vivo correlation (IVIVC) has been defined by the U.S. Food and Drug Administration The United States Food and Drug Administration (FDA or US FDA) is a List of United States federal agencies, federal agency of the United States Department of Health and Human Services, Department of Health and Human Services. The FDA is respo ... (FDA) as "a predictive mathematical model describing the relationship between an in-vitro property of a dosage form and an in-vivo response". Generally, the in-vitro property is the rate or extent of drug dissolution or release while the in-vivo response is the plasma drug concentration or amount of drug absorbed. The United States Pharmacopoeia (USP) also defines IVIVC as "the establishment of a relationship between a biological property, or a parameter derived from a biological property produced from a dosage form, and a physicochemical property of the same dosage form". Typically, the parameter derived from the biological property is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
|
Polar Surface Area
The polar surface area (PSA) or topological polar surface area (TPSA) of a molecule is defined as the surface sum over all polar atoms or molecules, primarily oxygen and nitrogen, also including their attached hydrogen atoms. PSA is a commonly used medicinal chemistry metric for the optimization of a drug's ability to permeate cells. Molecules with a polar surface area of greater than 140 angstroms squared (Å2) tend to be poor at permeating cell membranes. For molecules to penetrate the blood–brain barrier (and thus act on receptors in the central nervous system), a PSA less than 90 Å2 is usually needed. TPSA is a valuable tool in drug discovery and development. By analyzing a drug candidate's TPSA, scientists can predict its potential for oral bioavailability and ability to reach target sites within the body. This prediction hinges on a drug's ability to permeate biological barriers. Permeating these barriers, such as the Blood-Brain Barrier (BBB), the Placental Barrier (P ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
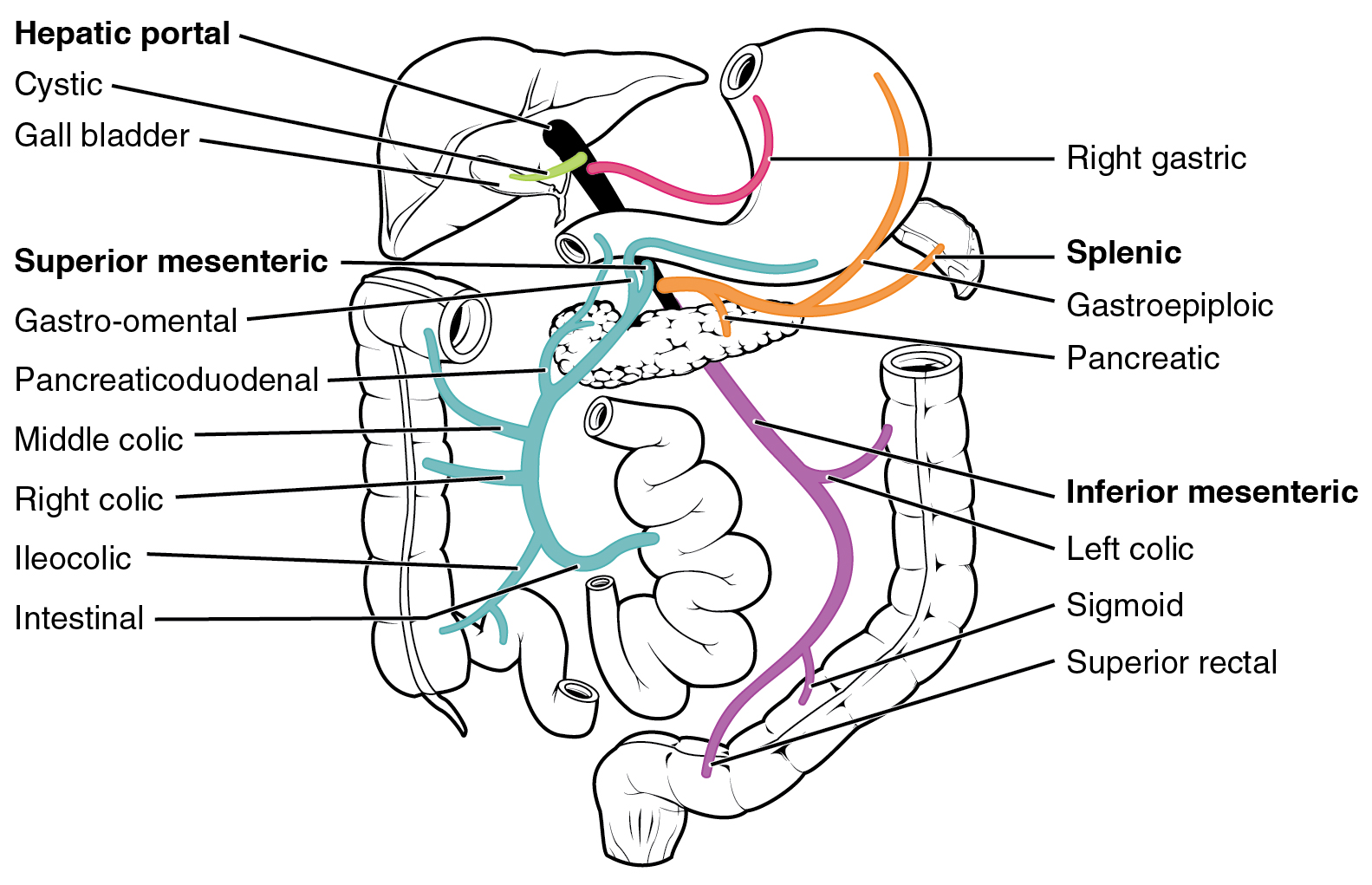 |
First Pass Effect
The first pass effect (also known as first-pass metabolism or presystemic metabolism) is a phenomenon of drug metabolism at a specific location in the body which leads to a reduction in the concentration of the active drug before it reaches the site of action or systemic circulation. The effect is most associated with orally administered medications, but some drugs still undergo first-pass metabolism even when delivered via an alternate route (e.g., IV, IM, etc.). During this metabolism, drug is lost during the process of absorption which is generally related to the liver and gut wall. The liver is the major site of first pass effect; however, it can also occur in the lungs, vasculature or other metabolically active tissues in the body. Notable drugs that experience a significant first pass effect are buprenorphine, chlorpromazine, cimetidine, diazepam, ethanol (drinking alcohol), imipramine, insulin, lidocaine, midazolam, morphine, pethidine, propranolol, and tetrahydroca ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
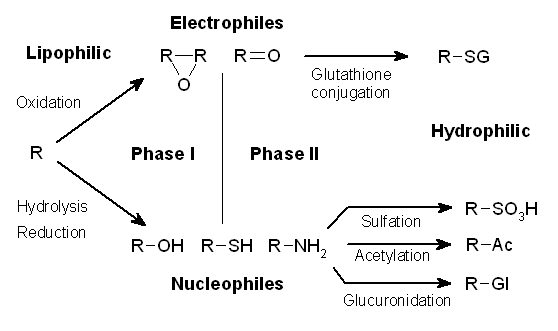 |
Drug Metabolism
Drug metabolism is the metabolic breakdown of drugs by living organisms, usually through specialized enzymatic systems. More generally, xenobiotic metabolism (from the Greek xenos "stranger" and biotic "related to living beings") is the set of metabolic pathways that modify the chemical structure of xenobiotics, which are compounds foreign to an organism's normal biochemistry, such as any drug or poison. These pathways are a form of biotransformation present in all major groups of organisms and are considered to be of ancient origin. These reactions often act to detoxify poisonous compounds (although in some cases the intermediates in xenobiotic metabolism can themselves cause toxic effects). The study of drug metabolism is the object of pharmacokinetics. Metabolism is one of the stages (see ADME) of the drug's transit through the body that involves the breakdown of the drug so that it can be excreted by the body. The metabolism of pharmaceutical drugs is an important as ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
Bioavailability
In pharmacology, bioavailability is a subcategory of absorption and is the fraction (%) of an administered drug that reaches the systemic circulation. By definition, when a medication is administered intravenously, its bioavailability is 100%. However, when a medication is administered via routes other than intravenous, its bioavailability is lower due to intestinal epithelium absorption and first-pass metabolism. Thereby, mathematically, bioavailability equals the ratio of comparing the area under the plasma drug concentration curve versus time (AUC) for the extravascular formulation to the AUC for the intravascular formulation. AUC is used because AUC is proportional to the dose that has entered the systemic circulation. Bioavailability of a drug is an average value; to take population variability into account, deviation range is shown as ±. To ensure that the drug taker who has poor absorption is dosed appropriately, the bottom value of the deviation range is employed ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
 |
Partition Coefficient
In the physical sciences, a partition coefficient (''P'') or distribution coefficient (''D'') is the ratio of concentrations of a chemical compound, compound in a mixture of two immiscible solvents at partition equilibrium, equilibrium. This ratio is therefore a comparison of the solubilities of the solute in these two liquids. The partition coefficient generally refers to the concentration ratio of ionization, un-ionized species of compound, whereas the distribution coefficient refers to the concentration ratio of all species of the compound (ionized plus un-ionized). In the chemistry, chemical and pharmaceutical sciences, both phases usually are solvents. Most commonly, one of the solvents is water, while the second is hydrophobic, such as 1-octanol. Hence the partition coefficient measures how hydrophilic ("water-loving") or hydrophobic ("water-fearing") a chemical substance is. Partition coefficients are useful in estimating the distribution (pharmacology), distribution of dru ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
ADME
ADME is the four-letter abbreviation (acronym) for absorption (pharmacokinetics), ''absorption'', distribution (pharmacology), ''distribution'', ''metabolism'', and ''excretion'', and is mainly used in fields such as pharmacokinetics and pharmacology. The four letter stands for descriptors quantifying how a given drug interacts within body over time. The term ADME was first introduced in the 1960s, and has become a standard term widely used in scientific literature, teaching, drug regulations, and clinical practice. ADME, describes the Drug Disposition, disposition of a pharmaceutical Chemical compound, compound within an organism. The four criteria all influence the drug metabolism, drug levels and kinetics of drug exposure to the tissues and hence influence the performance and pharmacological activity of the compound as a drug. Sometimes, liberation (pharmacology), liberation and/or toxicity are also considered, yielding LADME, ADMET, or LADMET. Components Absorption/admini ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
 |
Intravenous
Intravenous therapy (abbreviated as IV therapy) is a medical technique that administers fluids, medications and nutrients directly into a person's vein. The intravenous route of administration is commonly used for rehydration or to provide nutrients for those who cannot, or will not—due to reduced mental states or otherwise—consume food or water per os, by mouth. It may also be used to administer pharmaceutical drug, medications or other medical therapy such as blood transfusion, blood products or electrolytes to correct electrolyte imbalances. Attempts at providing intravenous therapy have been recorded as early as the 1400s, but the practice did not become widespread until the 1900s after the development of techniques for safe, effective use. The intravenous route is the fastest way to deliver medications and fluid replacement throughout the body as they are introduced directly into the circulatory system and thus quickly distributed. For this reason, the intravenous route ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
Bioequivalence
Bioequivalence is a term in pharmacokinetics used to assess the expected in vivo biological equivalence of two proprietary preparations of a drug. If two products are said to be bioequivalent it means that they would be expected to be, for all intents and purposes, the same. One article defined bioequivalence by stating that, "two pharmaceutical products are bioequivalent if they are pharmaceutically equivalent and their bioavailabilities (rate and extent of availability) after administration in the same molar dose are similar to such a degree that their effects, with respect to both efficacy and safety, can be expected to be essentially the same. Pharmaceutical equivalence implies the same amount of the same active substance(s), in the same dosage form, for the same route of administration and meeting the same or comparable standards." For The World Health Organization (WHO) "two pharmaceutical products are bioequivalent if they are pharmaceutically equivalent or pharmaceut ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |
|
|
Bifonazole
Bifonazole (trade name Canespor among others) is an imidazole antifungal drug used in form of ointments. It was patented in 1974 and approved for medical use in 1983. There are also combinations with carbamide for the treatment of onychomycosis. Adverse effects The most common side effect is a burning sensation at the application site. Other reactions, such as itching, eczema or skin dryness, are rare. Bifonazole is a potent aromatase inhibitor ''in vitro''. Pharmacology Mechanism of action Bifonazole has a dual mode of action. It inhibits fungal ergosterol biosynthesis at two points, via transformation of 24-methylendihydrolanosterol to desmethylsterol, together with inhibition of HMG-CoA. This enables fungicidal properties against dermatophytes and distinguishes bifonazole from other antifungal drugs. Pharmacokinetics Six hours after application, bifonazole concentrations range from 1000 Microgram, μg/Cubic centimetre, cm3 in the stratum corneum to 5 μg/cm3 in t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] [Amazon] |