|
Band Of Stability
Stable nuclides are isotopes of a chemical element whose nucleons are in a configuration that does not permit them the surplus energy required to produce a radioactive emission. The nuclei of such isotopes are not radioactive and unlike radionuclides do not spontaneously undergo radioactive decay. When these nuclides are referred to in relation to specific elements they are usually called that element's stable isotopes. The 80 elements with one or more stable isotopes comprise a total of 251 nuclides that have not been shown to decay using current equipment. Of these 80 elements, 26 have only one stable isotope and are called monoisotopic. The other 56 have more than one stable isotope. Tin has ten stable isotopes, the largest number of any element. Definition of stability, and naturally occurring nuclides Most naturally occurring nuclides are stable (about 251; see list at the end of this article), and about 35 more (total of 286) are known to be radioactive with long enou ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Table Isotopes En
Table may refer to: * Table (database), how the table data arrangement is used within the databases * Table (furniture), a piece of furniture with a flat surface and one or more legs * Table (information), a data arrangement with rows and columns * Table (landform), a flat area of land * Table (parliamentary procedure) * Table (sports), a ranking of the teams in a sports league * Tables (board game) * Mathematical table * Tables of the skull, a term for the flat bones * Table, surface of the sound board (music) of a string instrument * ''Al-Ma'ida'', the fifth ''surah'' of the Qur'an, occasionally translated as “The Table” * Calligra Tables, a spreadsheet application * Water table See also * Spreadsheet, a computer application * Table cut, a type of diamond cut * The Table (other) * Table Mountain (other) * Table Rock (other) Table Rock may refer to: Canada * Table Rock, Niagara Falls, a former rock formation ** Table Rock Welcome Centre, a reta ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Formation Of The Solar System
There is evidence that the formation of the Solar System began about 4.6 billion years ago with the gravitational collapse of a small part of a giant molecular cloud. Most of the collapsing mass collected in the center, forming the Sun, while the rest flattened into a protoplanetary disk out of which the planets, moons, asteroids, and other small Solar System bodies formed. This model, known as the nebular hypothesis, was first developed in the 18th century by Emanuel Swedenborg, Immanuel Kant, and Pierre-Simon Laplace. Its subsequent development has interwoven a variety of scientific disciplines including astronomy, chemistry, geology, physics, and planetary science. Since the dawn of the Space Age in the 1950s and the discovery of exoplanets in the 1990s, the model has been both challenged and refined to account for new observations. The Solar System has evolved considerably since its initial formation. Many moons have formed from circling discs of gas and dust around thei ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boron-10
Boron is a chemical element; it has symbol B and atomic number 5. In its crystalline form it is a brittle, dark, lustrous metalloid; in its amorphous form it is a brown powder. As the lightest element of the boron group it has three valence electrons for forming covalent bonds, resulting in many compounds such as boric acid, the mineral sodium borate, and the ultra-hard crystals of boron carbide and boron nitride. Boron is synthesized entirely by cosmic ray spallation and supernovas and not by stellar nucleosynthesis, so it is a low-abundance element in the Solar System and in the Earth's crust. It constitutes about 0.001 percent by weight of Earth's crust. It is concentrated on Earth by the water-solubility of its more common naturally occurring compounds, the borate minerals. These are mined industrially as evaporites, such as borax and kernite. The largest known deposits are in Turkey, the largest producer of boron minerals. Elemental boron is found in small amoun ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Lithium-6
Naturally occurring lithium (3Li) is composed of two stable isotope ratio, stable isotopes, lithium-6 (6Li) and lithium-7 (7Li), with the latter being far more abundant on Earth. Both of the natural isotopes have an unexpectedly low nuclear binding energy per nucleon ( for 6Li and for 7Li) when compared with the adjacent lighter and heavier elements, helium ( for helium-4) and beryllium ( for beryllium-9). The longest-lived radionuclide, radioisotope of lithium is 8Li, which has a half-life of just . 9Li has a half-life of , and 11Li has a half-life of . All of the remaining isotopes of lithium have half-lives that are shorter than 10 nanoseconds. The shortest-lived known isotope of lithium is 4Li, which decays by proton emission with a half-life of about (), although the half-life of 3Li is yet to be determined, and is likely to be much shorter, like 2He (helium-2, diproton) which undergoes proton emission within s. Both 7Li and 6Li are two of the primordial nuclides that we ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Deuterium
Deuterium (hydrogen-2, symbol H or D, also known as heavy hydrogen) is one of two stable isotopes of hydrogen; the other is protium, or hydrogen-1, H. The deuterium nucleus (deuteron) contains one proton and one neutron, whereas the far more common H has no neutrons. The name ''deuterium'' comes from Greek '' deuteros'', meaning "second". American chemist Harold Urey discovered deuterium in 1931. Urey and others produced samples of heavy water in which the H had been highly concentrated. The discovery of deuterium won Urey a Nobel Prize in 1934. Nearly all deuterium found in nature was synthesized in the Big Bang 13.8 billion years ago, forming the primordial ratio of H to H (~26 deuterium nuclei per 10 hydrogen nuclei). Deuterium is subsequently produced by the slow stellar proton–proton chain, but rapidly destroyed by exothermic fusion reactions. The deuterium–deuterium reaction has the second-lowest energy threshold, and is the most astrophysically acce ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isobar (nuclide)
Isobars are atoms (nuclides) of different chemical elements that have the same number of nucleons. Correspondingly, isobars differ in atomic number (or number of protons) but have the same mass number. An example of a series of isobars is 40S, 40Cl, 40Ar, 40K, and 40Ca. While the nuclei of these nuclides all contain 40 nucleons, they contain varying numbers of protons and neutrons. The term "isobars" (originally "isobares") for nuclides was suggested by British chemist Alfred Walter Stewart in 1918. It is derived . Mass The same mass number implies neither the same mass of nuclei, nor equal atomic masses of corresponding nuclides. From the Weizsäcker formula for the mass of a nucleus: : m(A,Z) = Z m_p + N m_n - a_ A + a_ A^ + a_ \frac + a_ \frac - \delta(A,Z) where mass number equals to the sum of atomic number and number of neutrons , and , , , , , are constants, one can see that the mass depends on and non-linearly, even for a constant ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Age Of The Universe
In physical cosmology, the age of the universe is the cosmological time, time elapsed since the Big Bang: 13.79 billion years. Astronomers have two different approaches to determine the age of the universe. One is based on a particle physics model of the early universe called Lambda-CDM model, Lambda-CDM, matched to measurements of the distant, and thus old features, like the cosmic microwave background. The other is based on the distance and relative velocity of a series or "ladder" of different kinds of stars, making it depend on local measurements late in the history of the universe. These two methods give slightly different values for the Hubble constant, which is then used in a formula to calculate the age. The range of the estimate is also within the range of the estimate for the List of oldest stars, oldest observed star in the universe. History In the 18th century, the concept that the age of Earth was millions, if not billions, of years began to appear. Nonetheless ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Double Beta Decay
In nuclear physics, double beta decay is a type of radioactive decay in which two neutrons are simultaneously transformed into two protons, or vice versa, inside an atomic nucleus. As in single beta decay, this process allows the atom to move closer to the optimal ratio of protons and neutrons. As a result of this transformation, the nucleus emits two detectable beta particles, which are electrons or positrons. The literature distinguishes between two types of double beta decay: ''ordinary'' double beta decay and ''neutrinoless'' double beta decay. In ordinary double beta decay, which has been observed in several isotopes, two electrons and two electron antineutrinos are emitted from the decaying nucleus. In neutrinoless double beta decay, a hypothesized process that has never been observed, only electrons would be emitted. History The idea of double beta decay was first proposed by Maria Goeppert Mayer in 1935. In 1937, Ettore Majorana demonstrated that all results of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Nuclear Shell Model
In nuclear physics, atomic physics, and nuclear chemistry, the nuclear shell model utilizes the Pauli exclusion principle to model the structure of atomic nuclei in terms of energy levels. The first shell model was proposed by Dmitri Ivanenko (together with E. Gapon) in 1932. The model was developed in 1949 following independent work by several physicists, most notably Maria Goeppert Mayer and J. Hans D. Jensen, who received the 1963 Nobel Prize in Physics for their contributions to this model, and Eugene Wigner, who received the Nobel Prize alongside them for his earlier groundlaying work on the atomic nuclei. The nuclear shell model is partly analogous to the atomic shell model, which describes the arrangement of electrons in an atom, in that a filled shell results in better stability. When adding nucleons (protons and neutrons) to a nucleus, there are certain points where the binding energy of the next nucleon is significantly less than the last one. This observation th ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
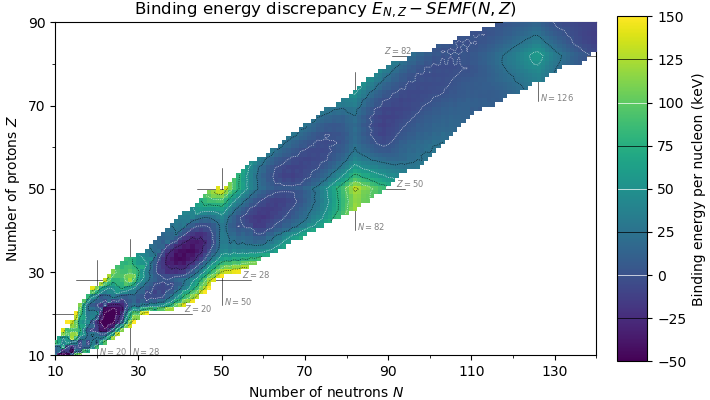
Magic Number (physics)
In nuclear physics, a magic number is a number of nucleons (either protons or neutrons, separately) such that they are arranged into complete shells within the atomic nucleus. As a result, atomic nuclei with a "magic" number of protons or neutrons are much more stable than other nuclei. The seven most widely recognized magic numbers as of 2019 are 2, 8, 20, 28, 50, 82, and 126. For protons, this corresponds to the elements helium, oxygen, calcium, nickel, tin, lead, and the hypothetical unbihexium, although 126 is so far only known to be a magic number for neutrons. Atomic nuclei consisting of such a magic number of nucleons have a higher average binding energy per nucleon than one would expect based upon predictions such as the semi-empirical mass formula and are hence more stable against nuclear decay. The unusual stability of isotopes having magic numbers means that transuranium elements could theoretically be created with extremely large nuclei and yet not be subject ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Promethium
Promethium is a chemical element; it has Symbol (chemistry), symbol Pm and atomic number 61. All of its isotopes are Radioactive decay, radioactive; it is extremely rare, with only about 500–600 grams naturally occurring in the Earth's crust at any given time. Promethium is one of the only two radioactive elements that are both preceded and followed in the periodic table by elements with stable forms, the other being technetium. Chemically, promethium is a lanthanide. Promethium shows only one stable oxidation state of +3. In 1902 Bohuslav Brauner suggested that there was a then-unknown element with properties intermediate between those of the known elements neodymium (60) and samarium (62); this was confirmed in 1914 by Henry Moseley, who, having measured the atomic numbers of all the elements then known, found that the element with atomic number 61 was missing. In 1926, two groups (one Italian and one American) claimed to have isolated a sample of element 61; both "discoverie ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Technetium
Technetium is a chemical element; it has Symbol (chemistry), symbol Tc and atomic number 43. It is the lightest element whose isotopes are all radioactive. Technetium and promethium are the only radioactive elements whose neighbours in the sense of atomic number are both stable. All available technetium is produced as a synthetic element. Naturally occurring technetium is a spontaneous fission product in uranium ore and thorium ore (the most common source), or the product of neutron capture in molybdenum ores. This silvery gray, crystalline transition metal lies between manganese and rhenium in group 7 element, group 7 of the periodic table, and its chemical properties are intermediate between those of both adjacent elements. The most common naturally occurring isotope is 99Tc, in traces only. Many of technetium's properties had been predicted by Dmitri Mendeleev before it was discovered; Mendeleev noted a gap in his periodic table and gave the undiscovered element the provis ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |