|
5-hydroxymethylfurfural
Hydroxymethylfurfural (HMF), also known as 5-(hydroxymethyl)furfural, is an organic compound formed by the dehydration of reducing sugars. It is a white low-melting solid (although commercial samples are often yellow) which is highly soluble in both water and organic solvents. The molecule consists of a furan ring, containing both aldehyde and alcohol functional groups. HMF can form in sugar-containing food, particularly as a result of heating or cooking. Its formation has been the topic of significant study as HMF was regarded as being potentially carcinogenic to humans. However, so far in vivo genotoxicity was negative. No relevance for humans concerning carcinogenic and genotoxic effects can be derived. HMF is classified as a food improvement agent and is primarily being used in the food industry in form of a food additive as a biomarker as well as a flavoring agent for food products. It is also produced industrially on a modest scale as a carbon-neutral feedstock for the prod ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2,5-Furandicarboxylic Acid
2,5-Furandicarboxylic acid (FDCA) is an organic chemical compound consisting of two carboxylic acid groups attached to a central furan ring. It was first reported as ''dehydromucic acid'' by Rudolph Fittig and Heinzelmann in 1876, who produced it via the action of concentrated hydrobromic acid upon mucic acid. It can be produced from certain carbohydrates and as such is a renewable resource, it was identified by the US Department of Energy as one of 12 priority chemicals for establishing the “green” chemistry industry of the future. Furan-2,5-dicarboxylic acid (FDCA) has been suggested as an important renewable building block because it can substitute for terephthalic acid (PTA) in the production of polyesters and other current polymers containing an aromatic moiety.T. Werpy, G. Petersen: ''Top Value Added Chemicals from Biomass. Volume I – Results of Screening for Potential Candidates from Sugars and Synthesis Gas.'' Produced by the Staff at Pacific Northwest National Labora ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Royal Society Of Chemistry
The Royal Society of Chemistry (RSC) is a learned society and professional association in the United Kingdom with the goal of "advancing the chemistry, chemical sciences". It was formed in 1980 from the amalgamation of the Chemical Society, the Royal Institute of Chemistry, the Faraday Society, and the Society for Analytical Chemistry with a new Royal Charter and the dual role of learned society and professional body. At its inception, the Society had a combined membership of 49,000 in the world. The headquarters of the Society are at Burlington House, Piccadilly, London. It also has offices in Thomas Graham House in Cambridge (named after Thomas Graham (chemist), Thomas Graham, the first president of the Chemical Society) where ''RSC Publishing'' is based. The Society has offices in the United States, on the campuses of The University of Pennsylvania and Drexel University, at the University City Science Center in Philadelphia, Pennsylvania, in both Beijing and Shanghai, People' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydrochloric Acid
Hydrochloric acid, also known as muriatic acid or spirits of salt, is an aqueous solution of hydrogen chloride (HCl). It is a colorless solution with a distinctive pungency, pungent smell. It is classified as a acid strength, strong acid. It is a component of the gastric acid in the digestive systems of most animal species, including humans. Hydrochloric acid is an important laboratory reagent and industrial chemical. Etymology Because it was produced from halite, rock salt according to the methods of Johann Rudolph Glauber, hydrochloric acid was historically called by European alchemists ''spirits of salt'' or ''acidum salis'' (salt acid). Both names are still used, especially in other languages, such as , , , , , , , , , , (''ensan''), zh, 盐酸 (''yánsuān''), and (''yeomsan''). Gaseous HCl was called ''marine acid air''. The name ''muriatic acid'' has the same origin (''muriatic'' means "pertaining to brine or salt", hence ''muriate'' means hydrochloride), and this ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
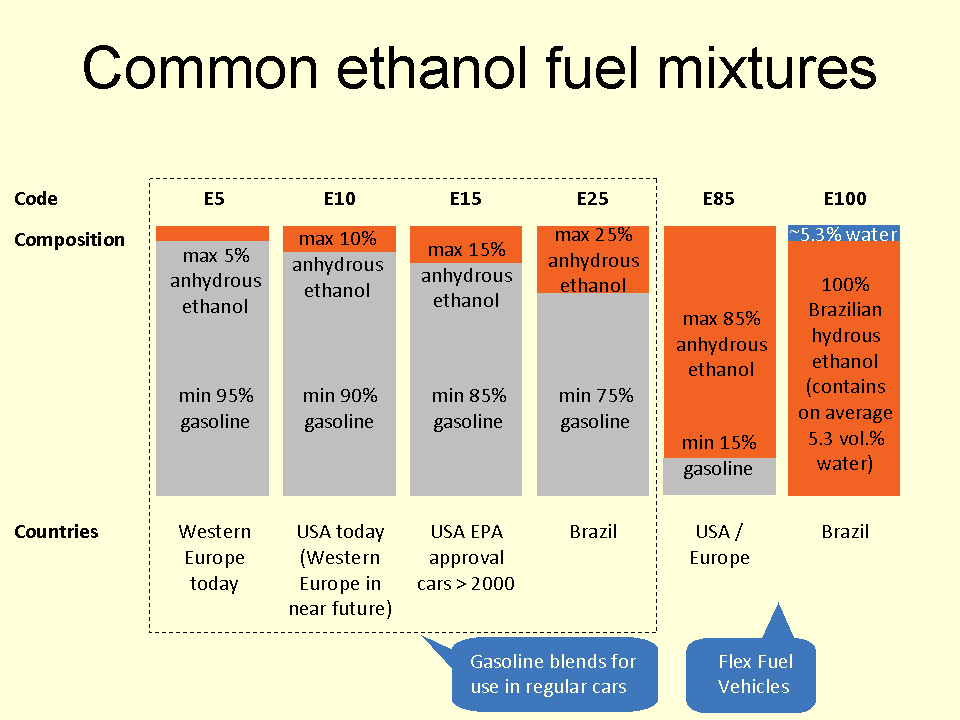
Bioethanol
Ethanol fuel is fuel containing ethyl alcohol, the same type of alcohol as found in alcoholic beverages. It is most often used as a motor fuel, mainly as a biofuel additive for gasoline. Several common ethanol fuel mixtures are in use around the world. The use of pure hydrous or anhydrous ethanol in internal combustion engines (ICEs) is possible only if the engines are designed or modified for that purpose. Anhydrous ethanol can be blended with :gasoline (petrol) for use in gasoline engines, but with a high ethanol content only after engine modifications to meter increased fuel volume since pure ethanol contains only 2/3 the energy of an equivalent volume of pure gasoline. High percentage ethanol mixtures are used in some racing engine applications since the very high octane rating of ethanol is compatible with very high compression ratios. The first production car running entirely on ethanol was the Fiat 147, introduced in 1978 in Brazil by Fiat. Ethanol is commonly m ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2,5-dimethylfuran
2,5-Dimethylfuran is a heterocyclic compound with the chemical formula, formula (CH3)2C4H2O. Although often abbreviated DMF, it should not be confused with dimethylformamide. A derivative of furan, this simple compound is a potential biofuel, being derivable from cellulose. Production Fructose can be converted into 2,5-dimethylfuran in a Catalysis, catalytic biomass-to-liquid process. The conversion of fructose to DMF proceeds via hydroxymethylfurfural. Fructose is obtainable from glucose, a building block in cellulose. Potential as a biofuel DMF has a number of attractions as a biofuel. It has an energy density 40% greater than that of ethanol, making it comparable to gasoline (petrol). It is also chemically stable and, being soluble, insoluble in water, does not absorb moisture from the atmosphere. evaporation, Evaporating dimethylfuran during the production process also requires around one third less energy than the evaporation of ethanol, although it has a boiling point some ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Polyesters
Polyester is a category of polymers that contain one or two ester linkages in every repeat unit of their main chain. As a specific material, it most commonly refers to a type called polyethylene terephthalate (PET). Polyesters include some naturally occurring chemicals, such as those found in plants and insects. Natural polyesters and a few synthetic ones are biodegradable, but most synthetic polyesters are not. Synthetic polyesters are used extensively in clothing. Polyester fibers are sometimes spun together with natural fibers to produce a cloth with blended properties. Cotton-polyester blends can be strong, wrinkle- and tear-resistant, and reduce shrinking. Synthetic fibers using polyester have high water, wind, and environmental resistance compared to plant-derived fibers. They are less Fireproofing, fire-resistant and can melt when ignited. Liquid crystalline polyesters are among the first industrially used liquid crystal polymers. They are used for their mechanical propert ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Terephthalic Acid
Terephthalic acid is an organic compound with formula C6H4(CO2H)2. This white solid is a commodity chemical, used principally as a precursor to the polyester PET, used to make clothing and plastic bottles. Several million tons are produced annually. The common name is derived from the turpentine-producing tree ''Pistacia terebinthus'' and phthalic acid. Terephthalic acid is also used in the production of PBT plastic (polybutylene terephthalate). History Terephthalic acid was first isolated (from turpentine) by the French chemist Amédée Cailliot (1805–1884) in 1846. Terephthalic acid became industrially important after World War II. Terephthalic acid was produced by oxidation of ''p''-xylene with 30-40% nitric acid. Air oxidation of ''p''-xylene gives ''p''-toluic acid, which resists further air-oxidation. Esterification of ''p''-toluic acid to methyl p-toluate (CH3C6H4CO2CH3) opens the way for further oxidation to monomethyl terephthalate. In the Dynamit−Nobel process t ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Acid Catalysis
In acid catalysis and base catalysis, a chemical reaction is catalyzed by an acid or a base. By Brønsted–Lowry acid–base theory, the acid is the proton (hydrogen ion, H+) donor and the base is the proton acceptor. Typical reactions catalyzed by proton transfer are esterifications and aldol reactions. In these reactions, the conjugate acid of the carbonyl group is a better electrophile than the neutral carbonyl group itself. Depending on the chemical species that act as the acid or base, catalytic mechanisms can be classified as either specific catalysis and general catalysis. Many enzymes operate by general catalysis. Applications and examples Brønsted acids Acid catalysis is mainly used for organic chemical reactions. Many acids can function as sources for the protons. Acid used for acid catalysis include hydrofluoric acid (in the alkylation process), phosphoric acid, toluenesulfonic acid, polystyrene sulfonate, heteropoly acids, zeolites. Strong acids catalyze the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Reactive Distillation
Reactive distillation is a process where the chemical reactor is also the still. Separation of the product from the reaction mixture does not need a separate distillation step which saves energy (for heating) and materials. This technique can be useful for equilibrium-limited reactions such as esterification and ester hydrolysis reactions. Conversion can be increased beyond what is expected by the equilibrium due to the continuous removal of reaction products from the reactive zone. This approach can also reduce capital and investment costs. The conditions in the reactive column are suboptimal both as a chemical reactor and as a distillation column, since the reactive column combines these. The introduction of an in situ separation process in the reaction zone or vice versa leads to complex interactions between vapor–liquid equilibrium, mass transfer rates, diffusion and chemical kinetics, which poses a great challenge for design and synthesis of these systems. Side reactors, wh ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ionic Liquids
An ionic liquid (IL) is a salt in the liquid state at ambient conditions. In some contexts, the term has been restricted to salts whose melting point is below a specific temperature, such as . While ordinary liquids such as water and gasoline are predominantly made of electrically neutral molecules, ionic liquids are largely made of ions. These substances are variously called liquid electrolytes, ionic melts, ionic fluids, fused salts, liquid salts, or ionic glasses. Ionic liquids have many potential applications. They are powerful solvents and can be used as electrolytes. Salts that are liquid at near-ambient temperature are important for electric battery applications, and have been considered as sealants due to their very low vapor pressure. Any salt that melts without decomposing or vaporizing usually yields an ionic liquid. Sodium chloride (NaCl), for example, melts at into a liquid that consists largely of sodium cations () and chloride anions (). Conversely, when an ioni ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |