|
2C-D
2C-D, also known as 2C-M or as 2,5-dimethoxy-4-methylphenethylamine, is a psychedelic drug of the 2C family that is sometimes used as an entheogen. It was first synthesized in 1970 by a team from the Texas Research Institute of Mental Sciences, and its activity was subsequently investigated in humans by Alexander Shulgin. Use and effects Not much information is known about the toxicity of 2C-D, as no major studies have been conducted. In his book ''PiHKAL'', Shulgin lists the dosage range as being from 20 to 60 mg. Lower doses of 10 mg or less have been explored for microdosing. According to Shulgin, the effects of 2C-D typically last for 4–6 hours. Shulgin himself referred to this substance as a “pharmacological tofu,” meaning that when mixed with other substances, it can extend or potentiate their effects without coloring the experience too much, in a manner similar to how tofu absorbs the flavors of sauces or spices it is cooked with. Hanscarl Leuner, working in Ge ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
2C (psychedelics)
2C (2C-''x'') is a general name for the family of psychedelic drug, psychedelic substituted phenethylamine, phenethylamines containing Methoxy, methoxy groups on the 2 and 5 carbon, positions of a benzene ring. Most of these compounds also carry lipophilic substituents at the 4 position, usually resulting in more potent and more metabolism, metabolically stable and longer acting compounds. Most of the currently known 2C compounds were first synthesized by Alexander Shulgin in the 1970s and 1980s and published in his book ''PiHKAL'' (''Phenethylamines i Have Known And Loved''). Shulgin also coined the term 2C, being an acronym for the 2 carbon atoms between the benzene ring and the amino group. 2C-B is the most popular of the 2C drugs. Use The 2C drugs are oral administration, orally active, are used at oral doses of 6 to 150mg depending on the drug, and have duration of action, durations of 3 to 48hours depending on the drug. However, many have doses in the range of 10 to 60mg an ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
PiHKAL
''PiHKAL: A Chemical Love Story'' is a book by Alexander Shulgin and Ann Shulgin published in 1991. The subject of the work is Psychoactive drug, psychoactive phenethylamine Derivative (chemistry), chemical derivatives, notably those that act as psychedelic drug, psychedelics and/or empathogen-entactogens. The main title, PiHKAL, is an acronym that stands for "Phenethylamines I Have Known and Loved". The book is arranged into two parts, the first part being a fictionalized autobiography of the couple and the second part describing 179 different psychedelic compounds (most of which Shulgin discovered himself), including detailed synthesis instructions, bioassays, dosages, and other commentary. The second part was made freely available by Shulgin on Erowid while the first part is available only in the printed text. While the reactions described are beyond the ability of people with a basic chemistry education, some tend to emphasize techniques that do not require difficult-to-ob ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Oral Administration
Oral administration is a route of administration whereby a substance is taken through the Human mouth, mouth, swallowed, and then processed via the digestive system. This is a common route of administration for many medications. Oral administration can be easier and less painful than other routes of administration, such as Injection (medicine), injection. However, the onset of action is relatively low, and the effectiveness is reduced if it is not absorbed properly in the digestive system, or if it is broken down by digestive enzymes before it can reach the bloodstream. Some medications may cause gastrointestinal side effects, such as nausea or vomiting, when taken orally. Oral administration can also only be applied to conscious patients, and patients able to swallow. Terminology ''Per os'' (; ''P.O.'') is an adverbial phrase meaning literally from Latin "through the mouth" or "by mouth". The expression is used in medicine to describe a treatment that is taken orally (but not ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
MAO-A
Monoamine oxidase A, also known as MAO-A, is an enzyme ( E.C. 1.4.3.4) that in humans is encoded by the ''MAOA'' gene. This gene is one of two neighboring gene family members that encode mitochondrial enzymes which catalyze the oxidative deamination of amines, such as norepinephrine, serotonin and tyramine. A mutation of this gene results in Brunner syndrome. This gene has also been associated with a variety of other psychiatric disorders, including antisocial behavior. Alternatively spliced transcript variants encoding multiple isoforms have been observed. Structures Gene Monoamine oxidase A, also known as MAO-A, is an enzyme that in humans is encoded by the ''MAOA'' gene. The promoter of ''MAOA'' contains conserved binding sites for Sp1, GATA2, and TBP. This gene is adjacent to a related gene ('' MAOB'') on the opposite strand of the X chromosome. In humans, there is a 30-base repeat sequence repeated several different numbers of times in the promoter region of ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
5-HT1B Receptor
5-hydroxytryptamine receptor 1B also known as the 5-HT1B receptor is a protein that in humans is encoded by the ''HTR1B'' gene. The 5-HT1B receptor is a 5-HT receptor subtype. Tissue distribution and function 5-HT1B receptors are widely distributed throughout the central nervous system with the highest concentrations found in the frontal cortex, basal ganglia, striatum, and the hippocampus. The function of the 5-HT1B receptor differs depending upon its location. In the frontal cortex, it is believed to act as a terminal receptor inhibiting the release of dopamine. In the basal ganglia and the striatum, evidence suggests 5-HT signaling acts on an autoreceptor, inhibiting the release of serotonin and decreasing glutamatergic transmission by reducing miniature excitatory postsynaptic potential (mEPSP) frequency, respectively. In the hippocampus, a recent study has demonstrated that activation of postsynaptic 5-HT1B heteroreceptors produces a facilitation in excitatory synapt ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
5-HT1A Receptor
The serotonin 1A receptor (or 5-HT1A receptor) is a subtype of serotonin receptors, or 5-HT receptors, that binds serotonin, also known as 5-HT, a neurotransmitter. 5-HT1A is expressed in the brain, spleen, and neonatal kidney. It is a G protein-coupled receptor (GPCR), coupled to the Gi protein, and its activation in the brain mediates hyperpolarization and reduction of firing rate of the postsynaptic neuron. In humans, the serotonin 1A receptor is encoded by the HTR1A gene. Distribution The 5-HT1A receptor is the most widespread of all the 5-HT receptors. In the central nervous system, 5-HT1A receptors exist in the cerebral cortex, hippocampus, Septum pellucidum, septum, amygdala, and Raphe nuclei, raphe nucleus in high densities, while low amounts also exist in the basal ganglia and thalamus. The 5-HT1A receptors in the raphe nucleus are largely somatodendritic autoreceptors, whereas those in other areas such as the hippocampus are postsynaptic receptors. Function Neur ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Affinity (pharmacology)
In biochemistry and pharmacology, a ligand is a substance that forms a complex with a biomolecule to serve a biological purpose. The etymology stems from Latin ''ligare'', which means 'to bind'. In protein-ligand binding, the ligand is usually a molecule which produces a signal by binding to a site on a target protein. The binding typically results in a change of conformational isomerism (conformation) of the target protein. In DNA-ligand binding studies, the ligand can be a small molecule, ion, or protein which binds to the DNA double helix. The relationship between ligand and binding partner is a function of charge, hydrophobicity, and molecular structure. Binding occurs by intermolecular forces, such as ionic bonds, hydrogen bonds and Van der Waals forces. The association or docking is actually reversible through dissociation. Measurably irreversible covalent bonding between a ligand and target molecule is atypical in biological systems. In contrast to the definition o ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Biological Target
A biological target is anything within a living organism to which some other entity (like an endogenous ligand or a drug) is directed and/or binds, resulting in a change in its behavior or function. Examples of common classes of biological targets are proteins and nucleic acids. The definition is context-dependent, and can refer to the biological target of a pharmacologically active drug compound, the receptor target of a hormone (like insulin), or some other target of an external stimulus. Biological targets are most commonly proteins such as enzymes, ion channels, and receptors. Mechanism The external stimulus (''i.e.'', the drug or ligand) physically binds to ("hits") the biological target. The interaction between the substance and the target may be: * noncovalent – A relatively weak interaction between the stimulus and the target where no chemical bond is formed between the two interacting partners and hence the interaction is completely reversible. * reversible c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Toxicity
Toxicity is the degree to which a chemical substance or a particular mixture of substances can damage an organism. Toxicity can refer to the effect on a whole organism, such as an animal, bacteria, bacterium, or plant, as well as the effect on a substructure of the organism, such as a cell (biology), cell (cytotoxicity) or an organ such as the liver (hepatotoxicity). Sometimes the word is more or less synonymous with poison#Poisoning, poisoning in everyday usage. A central concept of toxicology is that the effects of a toxicant are Dose (biochemistry), dose-dependent; even water can lead to water intoxication when taken in too high a dose, whereas for even a very toxic substance such as snake venom there is a dose below which there is no detectable toxic effect. Toxicity is species-specific, making cross-species analysis problematic. Newer paradigms and metrics are evolving to bypass animal testing, while maintaining the concept of toxicity endpoints. Etymology In Ancient G ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Overdose
A drug overdose (overdose or OD) is the ingestion or application of a drug or other substance in quantities much greater than are recommended. Retrieved on September 20, 2014."Stairway to Recovery: Glossary of Terms" . Retrieved on March 19, 2021 Typically the term is applied for cases when a risk to health is a potential result. An overdose may result in a toxicity, toxic state or death. Classification The word "overdose" implies that there is a common safe dosage and usage for the drug; therefore, the term is commonly applied only to drugs, not poisons, even though many poisons as well are harmless at a low enough dosage. Drug overdose is sometimes used as a means to commit suicide, as the result of intentional or unintentional misuse of medi ...[...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Selegiline
Selegiline, also known as L-deprenyl and sold under the brand names Eldepryl, Zelapar, and Emsam among others, is a medication which is used in the treatment of Parkinson's disease and major depressive disorder. It has also been studied and used off-label for a variety of other indications, but has not been formally approved for any other use. The medication, in the form licensed for depression, has modest effectiveness for this condition that is similar to that of other antidepressants. Selegiline is provided as a swallowed tablet or capsule or an orally disintegrating tablet (ODT) for Parkinson's disease and as a patch applied to skin for depression. Side effects of selegiline occurring more often than with placebo include insomnia, dry mouth, dizziness, anxiety, abnormal dreams, and application site reactions (with the patch form), among others. At high doses, selegiline has the potential for dangerous food and drug interactions, such as tyramine-related hypertens ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
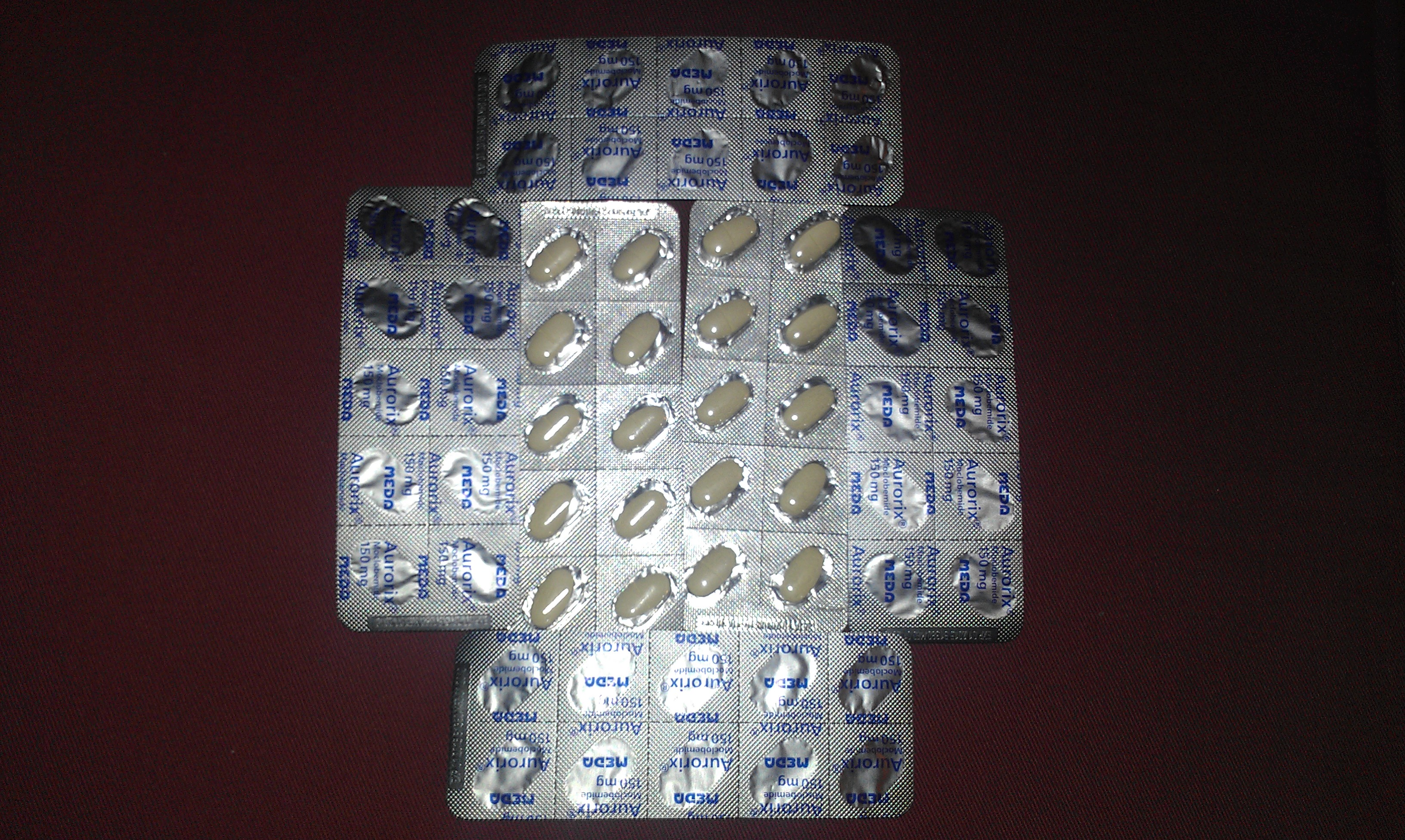
Moclobemide
Moclobemide, sold under the brand names Amira, Aurorix, Clobemix, Depnil and Manerix among others, is a reversible inhibitor of monoamine oxidase A (RIMA) drug primarily used to treat Clinical depression, depression and social anxiety. It is not approved for use in the United States, but is approved in other Western countries such as Canada, the United Kingdom, UK and Australia. It is produced by affiliates of the Hoffmann–La Roche pharmaceutical company. Initially, Aurorix was also marketed by Roche in South Africa, but was withdrawn after its patent rights expired and Cipla Medpro's Depnil and Pharma Dynamic's Clorix became available at half the cost. No significant rise in blood pressure occurs when moclobemide is combined with amines such as tyramine-containing foods or pressor amine drugs, unlike with the older irreversible and non-selective monoamine oxidase inhibitors (MAOIs), which cause a severe rise in blood pressure with such combination. Due to the lack of anticholi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |