Sulfate ion on:
[Wikipedia]
[Google]
[Amazon]
The sulfate or sulphate ion is a polyatomic anion with the empirical formula . Salts, acid derivatives, and
 The first description of the bonding in modern terms was by Gilbert Lewis in his groundbreaking paper of 1916, where he described the bonding in terms of electron octets around each atom. There are two double bonds, and there is a formal charge of +2 on the sulfur atom and -1 on each oxygen atom.
Later,
The first description of the bonding in modern terms was by Gilbert Lewis in his groundbreaking paper of 1916, where he described the bonding in terms of electron octets around each atom. There are two double bonds, and there is a formal charge of +2 on the sulfur atom and -1 on each oxygen atom.
Later,
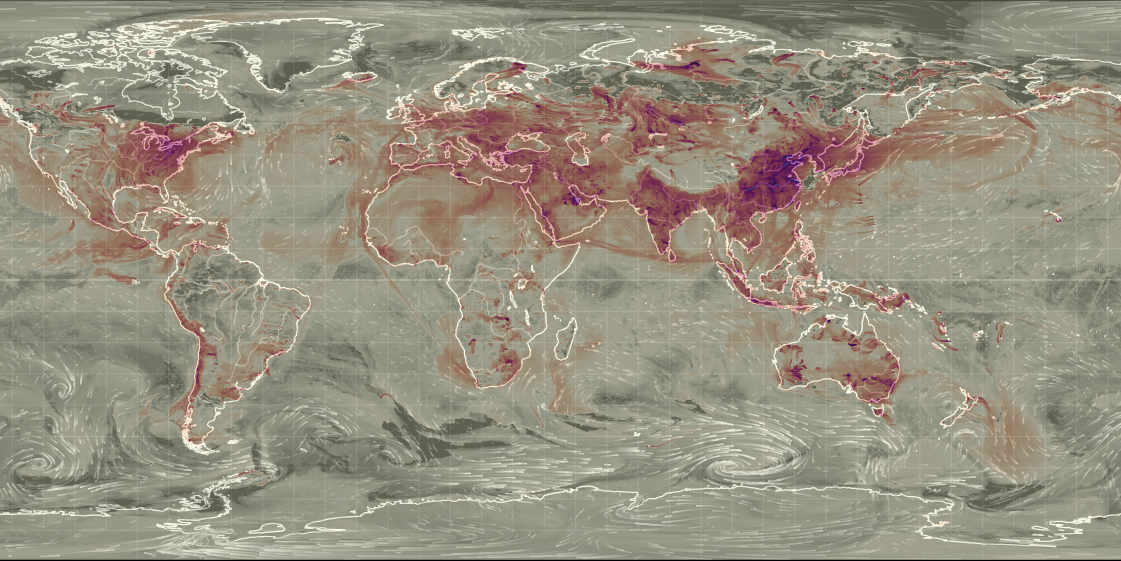
 As the real world had shown the importance of sulfate aerosol concentrations to the global climate, research into the subject accelerated. Formation of the aerosols and their effects on the atmosphere can be studied in the lab, with methods like ion-chromatography and
As the real world had shown the importance of sulfate aerosol concentrations to the global climate, research into the subject accelerated. Formation of the aerosols and their effects on the atmosphere can be studied in the lab, with methods like ion-chromatography and
Current global map of aerosol optical thickness
Particulates Sulfur oxyanions
peroxide
In chemistry, peroxides are a group of Chemical compound, compounds with the structure , where the R's represent a radical (a portion of a complete molecule; not necessarily a free radical) and O's are single oxygen atoms. Oxygen atoms are joined ...
s of sulfate are widely used in industry. Sulfates occur widely in everyday life. Sulfates are salt
In common usage, salt is a mineral composed primarily of sodium chloride (NaCl). When used in food, especially in granulated form, it is more formally called table salt. In the form of a natural crystalline mineral, salt is also known as r ...
s of sulfuric acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid (English in the Commonwealth of Nations, Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen, ...
and many are prepared from that acid.
Spelling
"Sulfate" is the spelling recommended byIUPAC
The International Union of Pure and Applied Chemistry (IUPAC ) is an international federation of National Adhering Organizations working for the advancement of the chemical sciences, especially by developing nomenclature and terminology. It is ...
, but "sulphate" was traditionally used in British English
British English is the set of Variety (linguistics), varieties of the English language native to the United Kingdom, especially Great Britain. More narrowly, it can refer specifically to the English language in England, or, more broadly, to ...
.
Structure
The sulfate anion consists of a centralsulfur
Sulfur ( American spelling and the preferred IUPAC name) or sulphur ( Commonwealth spelling) is a chemical element; it has symbol S and atomic number 16. It is abundant, multivalent and nonmetallic. Under normal conditions, sulfur atoms ...
atom surrounded by four equivalent oxygen
Oxygen is a chemical element; it has chemical symbol, symbol O and atomic number 8. It is a member of the chalcogen group (periodic table), group in the periodic table, a highly reactivity (chemistry), reactive nonmetal (chemistry), non ...
atoms in a tetrahedral
In geometry, a tetrahedron (: tetrahedra or tetrahedrons), also known as a triangular pyramid, is a polyhedron composed of four triangular Face (geometry), faces, six straight Edge (geometry), edges, and four vertex (geometry), vertices. The tet ...
arrangement. The symmetry of the isolated anion is the same as that of methane. The sulfur atom is in the +6 oxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical Electrical charge, charge of an atom if all of its Chemical bond, bonds to other atoms are fully Ionic bond, ionic. It describes the degree of oxidation (loss of electrons ...
while the four oxygen atoms are each in the −2 state. The sulfate ion carries an overall charge of −2 and it is the conjugate base
A conjugate acid, within the Brønsted–Lowry acid–base theory, is a chemical compound formed when an acid gives a proton () to a base—in other words, it is a base with a hydrogen ion added to it, as it loses a hydrogen ion in the reve ...
of the bisulfate (or hydrogensulfate) ion, , which is in turn the conjugate base of , sulfuric acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid (English in the Commonwealth of Nations, Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen, ...
. Organic sulfate ester
In organosulfur chemistry, organosulfates are a class of organic compounds sharing a common functional group with the structure . The core is a sulfate group and the R group is any Organyl group, organic residue. All organosulfates are formally ...
s, such as dimethyl sulfate
Dimethyl sulfate (DMS) is a chemical compound with formula (CH3O)2SO2. As the diester of methanol and sulfuric acid, its formula is often written as ( CH3)2 SO4 or Me2SO4, where CH3 or Me is methyl. Me2SO4 is mainly used as a methylating agen ...
, are covalent compounds and ester
In chemistry, an ester is a compound derived from an acid (either organic or inorganic) in which the hydrogen atom (H) of at least one acidic hydroxyl group () of that acid is replaced by an organyl group (R). These compounds contain a distin ...
s of sulfuric acid. The tetrahedral molecular geometry
In a tetrahedral molecular geometry, a central atom is located at the center with four substituents that are located at the corners of a tetrahedron. The bond angles are arccos(−) = 109.4712206...° ≈ 109.5° when all four substituents are ...
of the sulfate ion is as predicted by VSEPR theory
Valence shell electron pair repulsion (VSEPR) theory ( , ) is a conceptual model, model used in chemistry to predict the geometry of individual molecules from the number of electron pairs surrounding their central atoms. It is also named the Gill ...
.
Bonding
Linus Pauling
Linus Carl Pauling ( ; February 28, 1901August 19, 1994) was an American chemist and peace activist. He published more than 1,200 papers and books, of which about 850 dealt with scientific topics. ''New Scientist'' called him one of the 20 gre ...
used valence bond theory
In chemistry, valence bond (VB) theory is one of the two basic theories, along with molecular orbital (MO) theory, that were developed to use the methods of quantum mechanics to explain chemical bonding. It focuses on how the atomic orbitals of ...
to propose that the most significant resonance canonicals had two pi bond
In chemistry, pi bonds (π bonds) are covalent chemical bonds, in each of which two lobes of an orbital on one atom overlap with two lobes of an orbital on another atom, and in which this overlap occurs laterally. Each of these atomic orbital ...
s involving d orbitals. His reasoning was that the charge on sulfur was thus reduced, in accordance with his principle of electroneutrality. The S−O bond length of 149 pm is shorter than the bond lengths in sulfuric acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid (English in the Commonwealth of Nations, Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen, ...
of 157 pm for S−OH. The double bonding was taken by Pauling to account for the shortness of the S−O bond.
Pauling's use of d orbitals provoked a debate on the relative importance of pi bond
In chemistry, pi bonds (π bonds) are covalent chemical bonds, in each of which two lobes of an orbital on one atom overlap with two lobes of an orbital on another atom, and in which this overlap occurs laterally. Each of these atomic orbital ...
ing and bond polarity ( electrostatic attraction) in causing the shortening of the S−O bond. The outcome was a broad consensus that d orbitals play a role, but are not as significant as Pauling had believed.
A widely accepted description involving pπ – dπ bonding was initially proposed by Durward William John Cruickshank. In this model, fully occupied p orbitals on oxygen overlap with empty sulfur d orbitals (principally the d''z''2 and d''x''2–''y''2). However, in this description, despite there being some π character to the S−O bonds, the bond has significant ionic character. For sulfuric acid, computational analysis (with natural bond orbitals) confirms a clear positive charge on sulfur (theoretically +2.45) and a low 3d occupancy. Therefore, the representation with four single bonds is the optimal Lewis structure rather than the one with two double bonds (thus the Lewis model, not the Pauling model).
In this model, the structure obeys the octet rule
The octet rule is a chemical rule of thumb that reflects the theory that main-group elements tend to bond in such a way that each atom has eight electrons in its valence shell, giving it the same electronic configuration as a noble gas. The ru ...
and the charge distribution is in agreement with the electronegativity
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the ...
of the atoms. The discrepancy between the S−O bond length in the sulfate ion and the S−OH bond length in sulfuric acid is explained by donation of p-orbital electrons from the terminal S=O bonds in sulfuric acid into the antibonding S−OH orbitals, weakening them resulting in the longer bond length of the latter.
However, Pauling's representation for sulfate and other main group compounds with oxygen is still a common way of representing the bonding in many textbooks. The apparent contradiction can be clarified if one realizes that the covalent
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atom ...
double bonds in the Lewis structure actually represent bonds that are strongly polarized by more than 90% towards the oxygen atom. On the other hand, in the structure with a dipolar bond, the charge is localized as a lone pair
In chemistry, a lone pair refers to a pair of valence electrons that are not shared with another atom in a covalent bondIUPAC ''Gold Book'' definition''lone (electron) pair''/ref> and is sometimes called an unshared pair or non-bonding pair. Lone ...
on the oxygen.
Preparation
Typically metal sulfates are prepared by treating metal oxides, metal carbonates, or the metal itself withsulfuric acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid (English in the Commonwealth of Nations, Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen, ...
:
:
:
:
Although written with simple anhydrous formulas, these conversions generally are conducted in the presence of water. Consequently the product sulfates are hydrated, corresponding to zinc sulfate
Zinc sulfate is an inorganic compound with the formula ZnSO4. It forms hydrates ZnSO4·''n''H2O, where ''n'' can range from 0 to 7. All are colorless solids. The most common form includes water of crystallization as the heptahydrate, with the che ...
, copper(II) sulfate , and cadmium sulfate .
Some metal sulfide
Sulfide (also sulphide in British English) is an inorganic anion of sulfur with the chemical formula S2− or a compound containing one or more S2− ions. Solutions of sulfide salts are corrosive. ''Sulfide'' also refers to large families o ...
s can be oxidized to give metal sulfates.
Properties
There are numerous examples of ionic sulfates, many of which are highlysoluble
In chemistry, solubility is the ability of a substance, the solute, to form a solution with another substance, the solvent. Insolubility is the opposite property, the inability of the solute to form such a solution.
The extent of the solubi ...
in water
Water is an inorganic compound with the chemical formula . It is a transparent, tasteless, odorless, and Color of water, nearly colorless chemical substance. It is the main constituent of Earth's hydrosphere and the fluids of all known liv ...
. Exceptions include calcium sulfate
Calcium sulfate (or calcium sulphate) is an inorganic salt with the chemical formula . It occurs in several hydrated forms; the anhydrous state (known as anhydrite) is a white crystalline solid often found in evaporite deposits. Its dihydrate ...
, strontium sulfate, lead(II) sulfate, barium sulfate
Barium sulfate (or sulphate) is the inorganic compound with the chemical formula Ba SO4. It is a white crystalline solid that is odorless and insoluble in water. It occurs in nature as the mineral barite, which is the main commercial source of ...
, silver sulfate, and mercury sulfate, which are poorly soluble. Radium sulfate is the most insoluble sulfate known. The barium derivative is useful in the gravimetric analysis
Gravimetric analysis describes a set of methods used in analytical chemistry for the quantitative determination of an analyte (the ion being analyzed) based on its mass. The principle of this type of analysis is that once an ion's mass has been ...
of sulfate: if one adds a solution of most barium salts, for instance barium chloride
Barium chloride is an inorganic compound with the formula . It is one of the most common water-soluble salts of barium. Like most other water-soluble barium salts, it is a white powder, highly toxic, and imparts a yellow-green coloration to a flam ...
, to a solution containing sulfate ions, barium sulfate will precipitate out of solution as a whitish powder. This is a common laboratory test to determine if sulfate anions are present.
The sulfate ion can act as a ligand attaching either by one oxygen (monodentate) or by two oxygens as either a chelate or a bridge. An example is the complex or the neutral metal complex where the sulfate ion is acting as a bidentate ligand. The metal–oxygen bonds in sulfate complexes can have significant covalent character.
Uses and occurrence
Commercial applications
Sulfates are widely used industrially. Major compounds include: *Gypsum
Gypsum is a soft sulfate mineral composed of calcium sulfate Hydrate, dihydrate, with the chemical formula . It is widely mined and is used as a fertilizer and as the main constituent in many forms of plaster, drywall and blackboard or sidewalk ...
, the natural mineral form of hydrated calcium sulfate
Calcium sulfate (or calcium sulphate) is an inorganic salt with the chemical formula . It occurs in several hydrated forms; the anhydrous state (known as anhydrite) is a white crystalline solid often found in evaporite deposits. Its dihydrate ...
, is used to produce plaster
Plaster is a building material used for the protective or decorative coating of walls and ceilings and for moulding and casting decorative elements. In English, "plaster" usually means a material used for the interiors of buildings, while "re ...
. About 100 million tonnes per year are used by the construction industry.
* Copper sulfate Copper sulfate may refer to:
* Copper(II) sulfate, CuSO4, a common, greenish blue compound used as a fungicide and herbicide
* Copper(I) sulfate, Cu2SO4, an unstable white solid which is uncommonly used
{{chemistry index
Copper compounds ...
, a common fungicide
Fungicides are pesticides used to kill parasitic fungi or their spores. Fungi can cause serious damage in agriculture, resulting in losses of yield and quality. Fungicides are used both in agriculture and to fight fungal infections in animals, ...
, the more stable pentahydrate form ( ) is used for Bordeaux mixture in agriculture, galvanic cells as electrolyte and pigment.
* Iron(II) sulfate
Iron(II) sulfate or ferrous sulfate (British English: sulphate instead of sulfate) denotes a range of salts with the formula Fe SO4·''x''H2O. These compounds exist most commonly as the heptahydrate (''x'' = 7), but several values for ...
, a common form of iron in mineral supplements for humans, animals, and soil for plants.
* Magnesium sulfate
Magnesium sulfate or magnesium sulphate is a chemical compound, a salt with the formula , consisting of magnesium cations (20.19% by mass) and sulfate anions . It is a white crystalline solid, soluble in water but not in ethanol.
Magnesi ...
(commonly known as Epsom salts), used in therapeutic baths.
* Lead(II) sulfate, produced on both plates during the discharge of a lead–acid battery
The lead–acid battery is a type of rechargeable battery first invented in 1859 by French physicist Gaston Planté. It was the first type of rechargeable battery to be invented. Compared to modern rechargeable batteries, lead–acid batteries ha ...
.
* Sodium laureth sulfate, or SLES, a common detergent
A detergent is a surfactant or a mixture of surfactants with Cleanliness, cleansing properties when in Concentration, dilute Solution (chemistry), solutions. There are a large variety of detergents. A common family is the alkylbenzene sulfonate ...
in shampoo formulations.
* Polyhalite
Polyhalite is an evaporite mineral, a hydrated sulfate of potassium, calcium and magnesium with formula: . Polyhalite crystallizes in the triclinic system, although crystals are very rare. The normal habit is massive to fibrous. It is typica ...
, , used as fertiliser
A fertilizer or fertiliser is any material of natural or synthetic origin that is applied to soil or to plant tissues to supply plant nutrition, plant nutrients. Fertilizers may be distinct from Liming (soil), liming materials or other non- ...
.
Occurrence in nature
Sulfate-reducing bacteria
Sulfate-reducing microorganisms (SRM) or sulfate-reducing prokaryotes (SRP) are a group composed of sulfate-reducing bacteria (SRB) and sulfate-reducing archaea (SRA), both of which can perform anaerobic respiration utilizing sulfate () as termina ...
, some anaerobic microorganisms, such as those living in sediment or near deep sea thermal vents, use the reduction of sulfates coupled with the oxidation of organic compounds or hydrogen as an energy source for chemosynthesis.
History
Some sulfates were known to alchemists. The vitriol salts, from the Latin ''vitreolum'', glassy, were so-called because they were some of the first transparent crystals known. Green vitriol isiron
Iron is a chemical element; it has symbol Fe () and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, forming much of Earth's o ...
(II) sulfate heptahydrate, ; blue vitriol is copper
Copper is a chemical element; it has symbol Cu (from Latin ) and atomic number 29. It is a soft, malleable, and ductile metal with very high thermal and electrical conductivity. A freshly exposed surface of pure copper has a pinkish-orang ...
(II) sulfate pentahydrate, and white vitriol is zinc sulfate heptahydrate, . Alum
An alum () is a type of chemical compound, usually a hydrated double salt, double sulfate salt (chemistry), salt of aluminium with the general chemical formula, formula , such that is a valence (chemistry), monovalent cation such as potassium ...
, a double sulfate of potassium
Potassium is a chemical element; it has Symbol (chemistry), symbol K (from Neo-Latin ) and atomic number19. It is a silvery white metal that is soft enough to easily cut with a knife. Potassium metal reacts rapidly with atmospheric oxygen to ...
and aluminium
Aluminium (or aluminum in North American English) is a chemical element; it has chemical symbol, symbol Al and atomic number 13. It has a density lower than that of other common metals, about one-third that of steel. Aluminium has ...
with the formula , figured in the development of the chemical industry.
Environmental effects
Sulfates occur as microscopic particles (aerosols
An aerosol is a suspension of fine solid particles or liquid droplets in air or another gas. Aerosols can be generated from natural or human causes. The term ''aerosol'' commonly refers to the mixture of particulates in air, and not to t ...
) resulting from fossil fuel
A fossil fuel is a flammable carbon compound- or hydrocarbon-containing material formed naturally in the Earth's crust from the buried remains of prehistoric organisms (animals, plants or microplanktons), a process that occurs within geolog ...
and biomass
Biomass is a term used in several contexts: in the context of ecology it means living organisms, and in the context of bioenergy it means matter from recently living (but now dead) organisms. In the latter context, there are variations in how ...
combustion. They increase the acidity of the atmosphere
An atmosphere () is a layer of gases that envelop an astronomical object, held in place by the gravity of the object. A planet retains an atmosphere when the gravity is great and the temperature of the atmosphere is low. A stellar atmosph ...
and form acid rain
Acid rain is rain or any other form of Precipitation (meteorology), precipitation that is unusually acidic, meaning that it has elevated levels of hydrogen ions (low pH). Most water, including drinking water, has a neutral pH that exists b ...
. The anaerobic sulfate-reducing bacteria
Sulfate-reducing microorganisms (SRM) or sulfate-reducing prokaryotes (SRP) are a group composed of sulfate-reducing bacteria (SRB) and sulfate-reducing archaea (SRA), both of which can perform anaerobic respiration utilizing sulfate () as termina ...
'' Desulfovibrio desulfuricans'' and '' D. vulgaris'' can remove the black sulfate crust that often tarnishes buildings.
Main effects on climate

Reversal and accelerated warming
Hydrological cycle
Solar geoengineering
 As the real world had shown the importance of sulfate aerosol concentrations to the global climate, research into the subject accelerated. Formation of the aerosols and their effects on the atmosphere can be studied in the lab, with methods like ion-chromatography and
As the real world had shown the importance of sulfate aerosol concentrations to the global climate, research into the subject accelerated. Formation of the aerosols and their effects on the atmosphere can be studied in the lab, with methods like ion-chromatography and mass spectrometry
Mass spectrometry (MS) is an analytical technique that is used to measure the mass-to-charge ratio of ions. The results are presented as a ''mass spectrum'', a plot of intensity as a function of the mass-to-charge ratio. Mass spectrometry is used ...
Samples of actual particles can be recovered from the stratosphere
The stratosphere () is the second-lowest layer of the atmosphere of Earth, located above the troposphere and below the mesosphere. The stratosphere is composed of stratified temperature zones, with the warmer layers of air located higher ...
using balloons or aircraft, and remote satellite
A satellite or an artificial satellite is an object, typically a spacecraft, placed into orbit around a celestial body. They have a variety of uses, including communication relay, weather forecasting, navigation ( GPS), broadcasting, scient ...
s were also used for observation. This data is fed into the climate model
Numerical climate models (or climate system models) are mathematical models that can simulate the interactions of important drivers of climate. These drivers are the atmosphere, oceans, land surface and ice. Scientists use climate models to st ...
s, as the necessity of accounting for aerosol cooling to truly understand the rate and evolution of warming had long been apparent, with the IPCC Second Assessment Report being the first to include an estimate of their impact on climate, and every major model able to simulate them by the time IPCC Fourth Assessment Report was published in 2007. Many scientists also see the other side of this research, which is learning how to cause the same effect artificially. While discussed around the 1990s, if not earlier, stratospheric aerosol injection as a solar geoengineering method is best associated with Paul Crutzen
Paul Jozef Crutzen (; 3 December 1933 – 28 January 2021) was a Dutch meteorologist and atmospheric chemistry, atmospheric chemist. In 1995, he was awarded the Nobel Prize in Chemistry alongside Mario Molina and F. Sherwood Rowland, Frank Sherw ...
's detailed 2006 proposal. Deploying in the stratosphere ensures that the aerosols are at their most effective, and that the progress of clean air measures would not be reversed: more recent research estimated that even under the highest-emission scenario RCP 8.5, the addition of stratospheric sulfur required to avoid relative to now (and relative to the preindustrial) would be effectively offset by the future controls on tropospheric sulfate pollution, and the amount required would be even less for less drastic warming scenarios. This spurred a detailed look at its costs and benefits, but even with hundreds of studies into the subject completed by the early 2020s, some notable uncertainties remain.
Hydrogensulfate (bisulfate)
The hydrogensulfate ion (), also called the bisulfate ion, is theconjugate base
A conjugate acid, within the Brønsted–Lowry acid–base theory, is a chemical compound formed when an acid gives a proton () to a base—in other words, it is a base with a hydrogen ion added to it, as it loses a hydrogen ion in the reve ...
of sulfuric acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid (English in the Commonwealth of Nations, Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen, ...
(). Sulfuric acid is classified as a strong acid; in aqueous solutions it ionizes completely to form hydronium
In chemistry, hydronium (hydroxonium in traditional British English) is the cation , also written as , the type of oxonium ion produced by protonation of water. It is often viewed as the positive ion present when an Arrhenius acid is dissolved ...
() and hydrogensulfate () ions. In other words, the sulfuric acid behaves as a Brønsted–Lowry acid and is deprotonated to form hydrogensulfate ion. Hydrogensulfate has a valency of 1. An example of a salt containing the ion is sodium bisulfate, . In dilute solutions the hydrogensulfate ions also dissociate, forming more hydronium ions and sulfate ions ().
Other sulfur oxyanions
See also
*Sulfonate
In organosulfur chemistry, a sulfonate is a salt, anion or ester of a sulfonic acid. Its formula is , containing the functional group , where R is typically an organyl group, amino group or a halogen atom. Sulfonates are the conjugate bases of ...
* Sulfation and desulfation of lead–acid batteries
* Sulfate-reducing microorganism
Notes
References
{{SulfatesExternal links
Current global map of aerosol optical thickness
Particulates Sulfur oxyanions