Energy conversion efficiency on:
[Wikipedia]
[Google]
[Amazon]

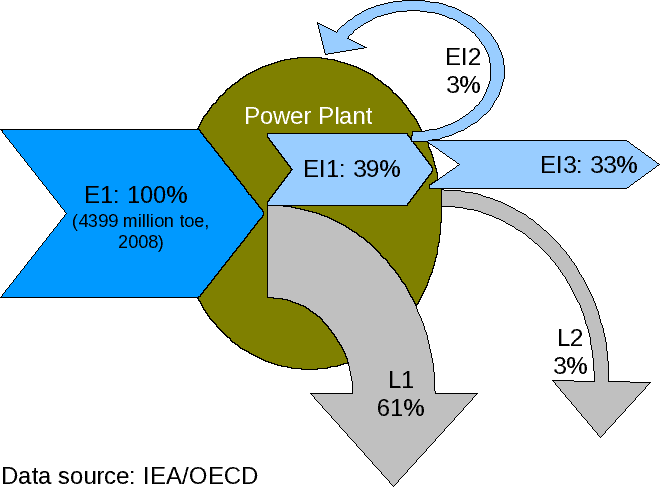 Energy conversion efficiency (''η'') is the
Energy conversion efficiency (''η'') is the
 In optical systems such as lighting and
In optical systems such as lighting and
Does it make sense to switch to LED?
{{DEFAULTSORT:Energy Conversion Efficiency Building engineering Dimensionless numbers of thermodynamics Energy conservation Energy conversion Energy efficiency

 Energy conversion efficiency (''η'') is the
Energy conversion efficiency (''η'') is the ratio
In mathematics, a ratio shows how many times one number contains another. For example, if there are eight oranges and six lemons in a bowl of fruit, then the ratio of oranges to lemons is eight to six (that is, 8:6, which is equivalent to the ...
between the useful output of an energy conversion machine and the input, in energy
In physics, energy (from Ancient Greek: ἐνέργεια, ''enérgeia'', “activity”) is the quantitative property that is transferred to a body or to a physical system, recognizable in the performance of work and in the form of ...
terms. The input, as well as the useful output may be chemical, electric power
Electric power is the rate at which electrical energy is transferred by an electric circuit. The SI unit of power is the watt, one joule per second. Standard prefixes apply to watts as with other SI units: thousands, millions and billions ...
, mechanical work, light
Light or visible light is electromagnetic radiation that can be perceived by the human eye. Visible light is usually defined as having wavelengths in the range of 400–700 nanometres (nm), corresponding to frequencies of 750–420 t ...
(radiation), or heat
In thermodynamics, heat is defined as the form of energy crossing the boundary of a thermodynamic system by virtue of a temperature difference across the boundary. A thermodynamic system does not ''contain'' heat. Nevertheless, the term is ...
. The resulting value, ''η'' (eta), ranges between 0 and 1.
Overview
Energy conversion efficiency depends on the usefulness of the output. All or part of the heat produced from burning a fuel may become rejectedwaste heat
Waste heat is heat that is produced by a machine, or other process that uses energy, as a byproduct of doing work. All such processes give off some waste heat as a fundamental result of the laws of thermodynamics. Waste heat has lower utilit ...
if, for example, work is the desired output from a thermodynamic cycle
A thermodynamic cycle consists of a linked sequence of thermodynamic processes that involve transfer of heat and work into and out of the system, while varying pressure, temperature, and other state variables within the system, and that eventu ...
. Energy converter is an example of an energy transformation. For example, a light bulb falls into the categories energy converter.
Even though the definition includes the notion of usefulness, efficiency
Efficiency is the often measurable ability to avoid wasting materials, energy, efforts, money, and time in doing something or in producing a desired result. In a more general sense, it is the ability to do things well, successfully, and without ...
is considered a technical
Technical may refer to:
* Technical (vehicle), an improvised fighting vehicle
* Technical analysis, a discipline for forecasting the future direction of prices through the study of past market data
* Technical drawing, showing how something is co ...
or physical
Physical may refer to:
* Physical examination, a regular overall check-up with a doctor
* ''Physical'' (Olivia Newton-John album), 1981
** "Physical" (Olivia Newton-John song)
* ''Physical'' (Gabe Gurnsey album)
* "Physical" (Alcazar song) (2004)
* ...
term. Goal or mission oriented terms include effectiveness
Effectiveness is the capability of producing a desired result or the ability to produce desired output. When something is deemed effective, it means it has an intended or expected outcome, or produces a deep, vivid impression.
Etymology
The ori ...
and efficacy
Efficacy is the ability to perform a task to a satisfactory or expected degree. The word comes from the same roots as ''effectiveness'', and it has often been used synonymously, although in pharmacology a distinction is now often made between ...
.
Generally, energy conversion efficiency is a dimensionless number between 0 and 1.0, or 0% to 100%. Efficiencies may not exceed 100%, e.g., for a perpetual motion
Perpetual motion is the motion of bodies that continues forever in an unperturbed system. A perpetual motion machine is a hypothetical machine that can do work infinitely without an external energy source. This kind of machine is impossible, a ...
machine. However, other effectiveness measures that can exceed 1.0 are used for heat pumps and other devices that move heat rather than convert it.
When talking about the efficiency of heat engines and power stations the convention should be stated, i.e., HHV ( Gross Heating Value, etc.) or LCV (a.k.a. Net Heating value), and whether gross output (at the generator terminals) or net output (at the power station fence) are being considered. The two are separate but both must be stated. Failure to do so causes endless confusion.
Related, more specific terms include
* Electrical efficiency, useful power output per electrical power consumed;
* Mechanical efficiency, where one form of mechanical energy (e.g. potential energy of water) is converted to mechanical energy ( work);
* Thermal efficiency or Fuel efficiency
Fuel efficiency is a form of thermal efficiency, meaning the ratio of effort to result of a process that converts chemical potential energy contained in a carrier (fuel) into kinetic energy or work. Overall fuel efficiency may vary per device ...
, useful heat
In thermodynamics, heat is defined as the form of energy crossing the boundary of a thermodynamic system by virtue of a temperature difference across the boundary. A thermodynamic system does not ''contain'' heat. Nevertheless, the term is ...
and/or work output per input energy such as the fuel
A fuel is any material that can be made to react with other substances so that it releases energy as thermal energy or to be used for work. The concept was originally applied solely to those materials capable of releasing chemical energy b ...
consumed;
*'Total efficiency', e.g., for cogeneration
Cogeneration or combined heat and power (CHP) is the use of a heat engine or power station to generate electricity and useful heat at the same time.
Cogeneration is a more efficient use of fuel or heat, because otherwise- wasted heat from elec ...
, useful electric power
Electric power is the rate at which electrical energy is transferred by an electric circuit. The SI unit of power is the watt, one joule per second. Standard prefixes apply to watts as with other SI units: thousands, millions and billions ...
and heat output per fuel energy consumed. Same as the thermal efficiency.
* Luminous efficiency, that portion of the emitted electromagnetic radiation is usable for human vision.
Chemical conversion efficiency
The change of Gibbs energy of a defined chemical transformation at a particular temperature is the minimum theoretical quantity of energy required to make that change occur (if the change in Gibbs energy between reactants and products is positive) or the maximum theoretical energy that might be obtained from that change (if the change in Gibbs energy between reactants and products is negative). The energy efficiency of a process involving chemical change may be expressed relative to these theoretical minima or maxima.The difference between the change of enthalpy and the change of Gibbs energy of a chemical transformation at a particular temperature indicates the heat input required or the heat removal (cooling) required to maintain that temperature. A fuel cell may be considered to be the reverse of electrolysis. For example, an ideal fuel cell operating at a temperature of 25 °C having gaseous hydrogen and gaseous oxygen as inputs and liquid water as the output could produce a theoretical maximum amount of electrical energy of 237.129 kJ (0.06587 kWh) per gram mol (18.0154 gram) of water produced and would require 48.701 kJ (0.01353 kWh) per gram mol of water produced of heat energy to be removed from the cell to maintain that temperature.D. D. Wagman, W. H. Evans, Vivian B. Parker, Richard H. Schumm, Iva Harlow, Sylvia M. Bailey, Kenneth L. Churney, and Ralph L. Nutall. "The NBS Tables of Chemical Thermodynamic Properties" Journal of Physical and Chemical Reference Data Volume 10, 1982 Supplement No. 2 An ideal electrolysis unit operating at a temperature of 25 °C having liquid water as the input and gaseous hydrogen and gaseous oxygen as products would require a theoretical minimum input of electrical energy of 237.129 kJ (0.06587 kWh) per gram mol (18.0154 gram) of water consumed and would require 48.701 kJ (0.01353 kWh) per gram mol of water consumed of heat energy to be added to the unit to maintain that temperature. It would operate at a cell voltage of 1.24 V. For a water electrolysis unit operating at a constant temperature of 25 °C without the input of any additional heat energy, electrical energy would have to be supplied at a rate equivalent of the enthalpy (heat) of reaction or 285.830 kJ (0.07940 kWh) per gram mol of water consumed. It would operate at a cell voltage of 1.48 V. The electrical energy input of this cell is 1.20 times greater than the theoretical minimum so the energy efficiency is 0.83 compared to the ideal cell. A water electrolysis unit operating with a higher voltage that 1.48 V and at a temperature of 25 °C would have to have heat energy removed in order to maintain a constant temperature and the energy efficiency would be less than 0.83. The large entropy difference between liquid water and gaseous hydrogen plus gaseous oxygen accounts for the significant difference between the Gibbs energy of reaction and the enthalpy (heat) of reaction.Fuel heating values and efficiency
In Europe the usable energy content of a fuel is typically calculated using the lower heating value (LHV) of that fuel, the definition of which assumes that thewater vapor
(99.9839 °C)
, -
, Boiling point
,
, -
, specific gas constant
, 461.5 J/( kg·K)
, -
, Heat of vaporization
, 2.27 MJ/kg
, -
, Heat capacity
, 1.864 kJ/(kg·K)
Water vapor, water vapour or aqueous vapor is the gaseous p ...
produced during fuel combustion
Combustion, or burning, is a high-temperature exothermic redox chemical reaction between a fuel (the reductant) and an oxidant, usually atmospheric oxygen, that produces oxidized, often gaseous products, in a mixture termed as smoke. Combus ...
(oxidation) remains gaseous, and is not condensed to liquid water so the latent heat of vaporization of that water is not usable. Using the LHV, a condensing boiler
Condensing boilers are water heaters typically used for heating systems that are fueled by gas or oil. When operated in the correct circumstances, a heating system can achieve high efficiency (greater than 90% on the higher heating value) by cond ...
can achieve a "heating efficiency" in excess of 100% (this does not violate the first law of thermodynamics
The first law of thermodynamics is a formulation of the law of conservation of energy, adapted for thermodynamic processes. It distinguishes in principle two forms of energy transfer, heat and thermodynamic work for a system of a constant am ...
as long as the LHV convention is understood, but does cause confusion). This is because the apparatus recovers part of the heat of vaporization
The enthalpy of vaporization (symbol ), also known as the (latent) heat of vaporization or heat of evaporation, is the amount of energy (enthalpy) that must be added to a liquid substance to transform a quantity of that substance into a gas. T ...
, which is not included in the definition of the lower heating value of a fuel. In the U.S. and elsewhere, the higher heating value (HHV) is used, which includes the latent heat for condensing the water vapor, and thus the thermodynamic maximum of 100% efficiency cannot be exceeded.
Wall-plug efficiency, luminous efficiency, and efficacy
laser
A laser is a device that emits light through a process of optical amplification based on the stimulated emission of electromagnetic radiation. The word "laser" is an acronym for "light amplification by stimulated emission of radiation". The ...
s, the energy conversion efficiency is often referred to as wall-plug efficiency. The wall-plug efficiency is the measure of output radiative-energy, in watt
The watt (symbol: W) is the unit of power or radiant flux in the International System of Units (SI), equal to 1 joule per second or 1 kg⋅m2⋅s−3. It is used to quantify the rate of energy transfer. The watt is named after James ...
s (joule
The joule ( , ; symbol: J) is the unit of energy in the International System of Units (SI). It is equal to the amount of work done when a force of 1 newton displaces a mass through a distance of 1 metre in the direction of the force appli ...
s per second), per total input electrical energy in watts. The output energy is usually measured in terms of absolute Absolute may refer to:
Companies
* Absolute Entertainment, a video game publisher
* Absolute Radio, (formerly Virgin Radio), independent national radio station in the UK
* Absolute Software Corporation, specializes in security and data risk manag ...
irradiance and the wall-plug efficiency is given as a percentage of the total input energy, with the inverse percentage representing the losses.
The wall-plug efficiency differs from the ''luminous efficiency'' in that wall-plug efficiency describes the direct output/input conversion of energy (the amount of work that can be performed) whereas luminous efficiency takes into account the human eye's varying sensitivity to different wavelengths (how well it can illuminate a space). Instead of using watts, the power of a light source to produce wavelengths proportional to human perception is measured in lumens. The human eye is most sensitive to wavelengths of 555 nanometers (greenish-yellow) but the sensitivity decreases dramatically to either side of this wavelength, following a Gaussian
Carl Friedrich Gauss (1777–1855) is the eponym of all of the topics listed below.
There are over 100 topics all named after this German mathematician and scientist, all in the fields of mathematics, physics, and astronomy. The English eponym ...
power-curve and dropping to zero sensitivity at the red and violet ends of the spectrum. Due to this the eye does not usually see all of the wavelengths emitted by a particular light-source, nor does it see all of the wavelengths within the visual spectrum equally. Yellow and green, for example, make up more than 50% of what the eye perceives as being white, even though in terms of radiant energy white-light is made from equal portions of all colors (i.e.: a 5 mW green laser appears brighter than a 5 mW red laser, yet the red laser stands-out better against a white background). Therefore, the radiant intensity of a light source may be much greater than its luminous intensity, meaning that the source emits more energy than the eye can use. Likewise, the lamp's wall-plug efficiency is usually greater than its luminous efficiency. The effectiveness of a light source to convert electrical energy into wavelengths of visible light, in proportion to the sensitivity of the human eye, is referred to as luminous efficacy, which is measured in units of lumens per watt (lm/w) of electrical input-energy.
Unlike efficacy (effectiveness), which is a unit of measurement, efficiency is a unitless
A dimensionless quantity (also known as a bare quantity, pure quantity, or scalar quantity as well as quantity of dimension one) is a quantity to which no physical dimension is assigned, with a corresponding SI unit of measurement of one (or 1) ...
number expressed as a percentage
In mathematics, a percentage (from la, per centum, "by a hundred") is a number or ratio expressed as a fraction of 100. It is often denoted using the percent sign, "%", although the abbreviations "pct.", "pct" and sometimes "pc" are also use ...
, requiring only that the input and output units be of the same type. The luminous efficiency of a light source is thus the percentage of luminous efficacy per theoretical maximum efficacy at a specific wavelength. The amount of energy carried by a photon of light is determined by its wavelength. In lumens, this energy is offset by the eye's sensitivity to the selected wavelengths. For example, a green laser pointer can have greater than 30 times the apparent brightness of a red pointer of the same power output. At 555 nm in wavelength, 1 watt of radiant energy is equivalent to 685 lumens, thus a monochromatic light source at this wavelength, with a luminous efficacy of 685 lm/w, has a luminous efficiency of 100%. The theoretical-maximum efficacy lowers for wavelengths at either side of 555 nm. For example, low-pressure sodium lamps produce monochromatic light at 589 nm with a luminous efficacy of 200 lm/w, which is the highest of any lamp. The theoretical-maximum efficacy at that wavelength is 525 lm/w, so the lamp has a luminous efficiency of 38.1%. Because the lamp is monochromatic, the luminous efficiency nearly matches the wall-plug efficiency of < 40%.
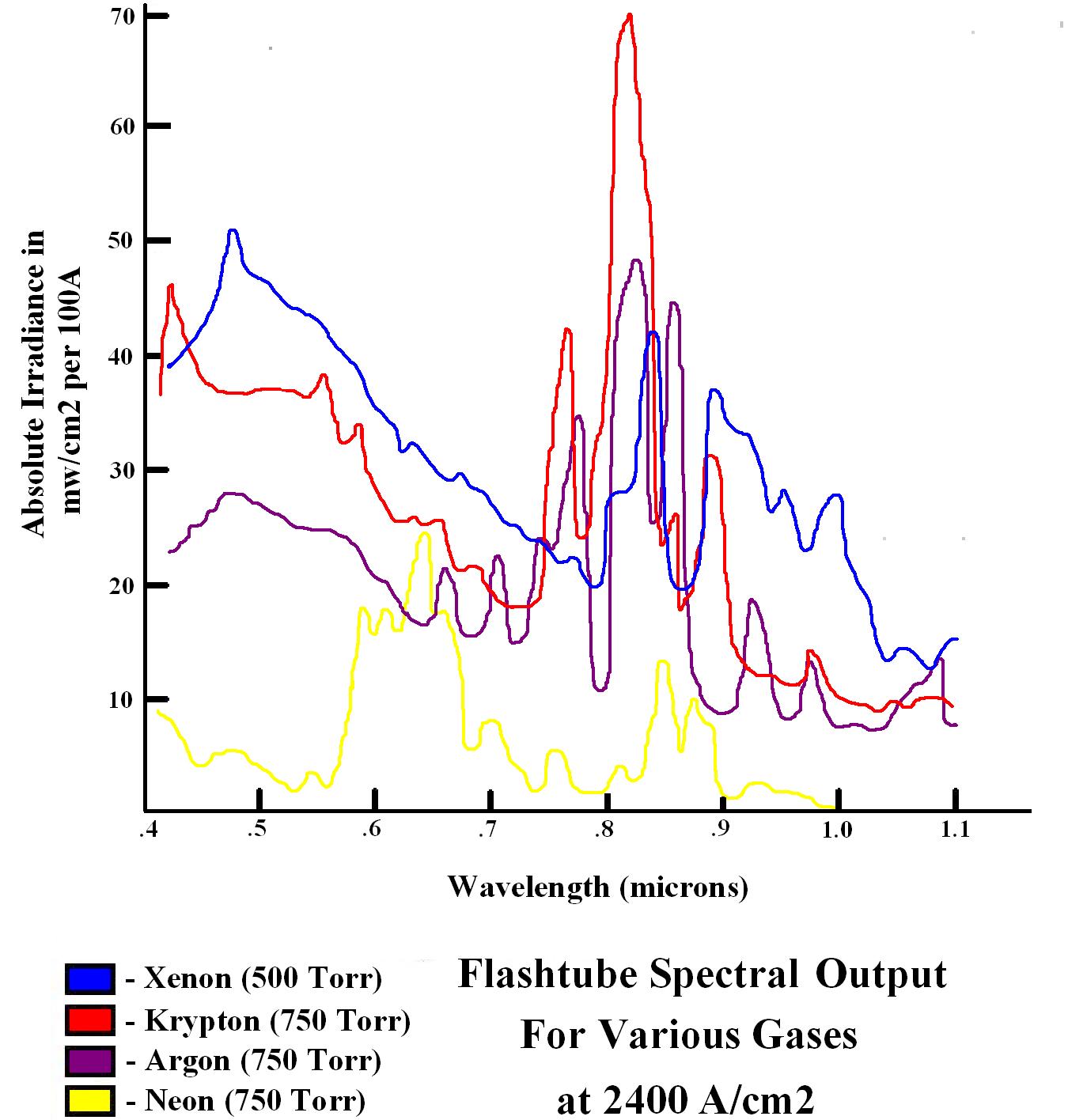
Calculations for luminous efficiency become more complex for lamps that produce white light or a mixture of spectral lines. Fluorescent lamps have higher wall-plug efficiencies than low-pressure sodium lamps, but only have half the luminous efficacy of ~ 100 lm/w, thus the luminous efficiency of fluorescents is lower than sodium lamps. A xenon flashtube
A flashtube (flashlamp) is an electric arc lamp designed to produce extremely intense, incoherent, full-spectrum white light for a very short time. A flashtube is a glass tube with an electrode at each end and is filled with a gas that, when tr ...
has a typical wall-plug efficiency of 50–70%, exceeding that of most other forms of lighting. Because the flashtube emits large amounts of infrared and ultraviolet radiation, only a portion of the output energy is used by the eye. The luminous efficacy is therefore typically around 50 lm/w. However, not all applications for lighting involve the human eye nor are restricted to visible wavelengths. For laser pumping, the efficacy is not related to the human eye so it is not called "luminous" efficacy, but rather simply "efficacy" as it relates to the absorption lines of the laser medium. Krypton flashtubes are often chosen for pumping Nd:YAG lasers, even though their wall-plug efficiency is typically only ~ 40%. Krypton
Krypton (from grc, κρυπτός, translit=kryptos 'the hidden one') is a chemical element with the symbol Kr and atomic number 36. It is a colorless, odorless, tasteless noble gas that occurs in trace amounts in the atmosphere and is of ...
's spectral lines better match the absorption lines of the neodymium
Neodymium is a chemical element with the symbol Nd and atomic number 60. It is the fourth member of the lanthanide series and is considered to be one of the rare-earth metals. It is a hard, slightly malleable, silvery metal that quickly tarn ...
- doped crystal, thus the efficacy of krypton for this purpose is much higher than xenon
Xenon is a chemical element with the symbol Xe and atomic number 54. It is a dense, colorless, odorless noble gas found in Earth's atmosphere in trace amounts. Although generally unreactive, it can undergo a few chemical reactions such as the ...
; able to produce up to twice the laser output for the same electrical input. All of these terms refer to the amount of energy and lumens as they exit the light source, disregarding any losses that might occur within the lighting fixture or subsequent output optics. ''Luminaire efficiency'' refers to the total lumen-output from the fixture per the lamp output.
With the exception of a few light sources, such as incandescent light bulb
An incandescent light bulb, incandescent lamp or incandescent light globe is an electric light with a wire filament heated until it glows. The filament is enclosed in a glass bulb with a vacuum or inert gas to protect the filament from oxi ...
s, most light sources have multiple stages of energy conversion between the "wall plug" (electrical input point, which may include batteries, direct wiring, or other sources) and the final light-output, with each stage producing a loss. Low-pressure sodium lamps initially convert the electrical energy using an electrical ballast, to maintain the proper current and voltage, but some energy is lost in the ballast. Similarly, fluorescent lamps also convert the electricity using a ballast (electronic efficiency). The electricity is then converted into light energy by the electrical arc
An electric arc, or arc discharge, is an electrical breakdown of a gas that produces a prolonged electrical discharge. The current through a normally nonconductive medium such as air produces a plasma; the plasma may produce visible light. A ...
(electrode efficiency and discharge efficiency). The light is then transferred to a fluorescent coating that only absorbs suitable wavelengths, with some losses of those wavelengths due to reflection off and transmission through the coating (transfer efficiency). The number of photons absorbed by the coating will not match the number then reemitted as fluorescence
Fluorescence is the emission of light by a substance that has absorbed light or other electromagnetic radiation. It is a form of luminescence. In most cases, the emitted light has a longer wavelength, and therefore a lower photon energy, tha ...
(quantum efficiency
The term quantum efficiency (QE) may apply to incident photon to converted electron (IPCE) ratio of a photosensitive device, or it may refer to the TMR effect of a Magnetic Tunnel Junction.
This article deals with the term as a measurement of ...
). Finally, due to the phenomenon of the Stokes shift
__NOTOC__
Stokes shift is the difference (in energy, wavenumber or frequency units) between positions of the band maxima of the absorption and emission spectra ( fluorescence and Raman being two examples) of the same electronic transition. I ...
, the re-emitted photons will have a longer wavelength (thus lower energy) than the absorbed photons (fluorescence efficiency). In very similar fashion, lasers also experience many stages of conversion between the wall plug and the output aperture
In optics, an aperture is a hole or an opening through which light travels. More specifically, the aperture and focal length of an optical system determine the cone angle of a bundle of rays that come to a focus in the image plane.
An ...
. The terms "wall-plug efficiency" or "energy conversion efficiency" are therefore used to denote the overall efficiency of the energy-conversion device, deducting the losses from each stage, although this may exclude external components needed to operate some devices, such as coolant pumps.''Handbook of Luminescent Semiconductor Materials'' by Leah Bergman, Jeanne L. McHale -- CRC Press 2012 Page 270
Example of energy conversion efficiency
See also
* Cost of electricity by source * Energy efficiency (disambiguation) * EROEI *Exergy efficiency Exergy efficiency (also known as the second-law efficiency or rational efficiency) computes the effectiveness of a system relative to its performance in reversible conditions. It is defined as the ratio of the thermal efficiency of an actual system ...
* Figure of merit
*Heat of combustion
The heating value (or energy value or calorific value) of a substance, usually a fuel or food (see food energy), is the amount of heat released during the combustion of a specified amount of it.
The ''calorific value'' is the total energy rele ...
*International Electrotechnical Commission
The International Electrotechnical Commission (IEC; in French: ''Commission électrotechnique internationale'') is an international standards organization that prepares and publishes international standards for all electrical, electronic and ...
*Perpetual motion
Perpetual motion is the motion of bodies that continues forever in an unperturbed system. A perpetual motion machine is a hypothetical machine that can do work infinitely without an external energy source. This kind of machine is impossible, a ...
* Sensitivity (electronics)
* Solar cell efficiency
*Coefficient of performance
The coefficient of performance or COP (sometimes CP or CoP) of a heat pump, refrigerator or air conditioning system is a ratio of useful heating or cooling provided to work (energy) required. Higher COPs equate to higher efficiency, lower energy ( ...
References
External links
Does it make sense to switch to LED?
{{DEFAULTSORT:Energy Conversion Efficiency Building engineering Dimensionless numbers of thermodynamics Energy conservation Energy conversion Energy efficiency