Hydrogen is a
chemical element
A chemical element is a chemical substance whose atoms all have the same number of protons. The number of protons is called the atomic number of that element. For example, oxygen has an atomic number of 8: each oxygen atom has 8 protons in its ...
; it has
symbol
A symbol is a mark, Sign (semiotics), sign, or word that indicates, signifies, or is understood as representing an idea, physical object, object, or wikt:relationship, relationship. Symbols allow people to go beyond what is known or seen by cr ...
H and
atomic number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of its atomic nucleus. For ordinary nuclei composed of protons and neutrons, this is equal to the proton number (''n''p) or the number of pro ...
1. It is the lightest and
most abundant chemical element in the
universe
The universe is all of space and time and their contents. It comprises all of existence, any fundamental interaction, physical process and physical constant, and therefore all forms of matter and energy, and the structures they form, from s ...
, constituting about 75% of all
normal matter. Under
standard conditions, hydrogen is a
gas of
diatomic molecule
Diatomic molecules () are molecules composed of only two atoms, of the same or different chemical elements. If a diatomic molecule consists of two atoms of the same element, such as hydrogen () or oxygen (), then it is said to be homonuclear mol ...
s with the
formula
In science, a formula is a concise way of expressing information symbolically, as in a mathematical formula or a ''chemical formula''. The informal use of the term ''formula'' in science refers to the general construct of a relationship betwe ...
, called dihydrogen, or sometimes hydrogen gas, molecular hydrogen, or simply hydrogen. Dihydrogen is colorless, odorless, non-toxic, and highly
combustible
A combustible material is a material that can burn (i.e., sustain a flame) in air under certain conditions. A material is flammable if it ignites easily at ambient temperatures. In other words, a combustible material ignites with some effort a ...
.
Stars
A star is a luminous spheroid of plasma held together by self-gravity. The nearest star to Earth is the Sun. Many other stars are visible to the naked eye at night; their immense distances from Earth make them appear as fixed points of ...
, including the
Sun, mainly consist of hydrogen in a
plasma state, while on Earth, hydrogen is found as the gas (dihydrogen) and in
molecular forms, such as in
water
Water is an inorganic compound with the chemical formula . It is a transparent, tasteless, odorless, and Color of water, nearly colorless chemical substance. It is the main constituent of Earth's hydrosphere and the fluids of all known liv ...
and
organic compounds
Some chemical authorities define an organic compound as a chemical compound that contains a carbon–hydrogen or carbon–carbon bond; others consider an organic compound to be any chemical compound that contains carbon. For example, carbon-co ...
. The most common
isotope of hydrogen
Hydrogen (H) has three naturally occurring isotopes: H, H, and H. H and H are stable, while H has a half-life of years. Heavier isotopes also exist; all are synthetic and have a half-life of less than 1 zeptosecond (10 s).
Of these, H is t ...
(H) consists of one
proton
A proton is a stable subatomic particle, symbol , Hydron (chemistry), H+, or 1H+ with a positive electric charge of +1 ''e'' (elementary charge). Its mass is slightly less than the mass of a neutron and approximately times the mass of an e ...
, one
electron
The electron (, or in nuclear reactions) is a subatomic particle with a negative one elementary charge, elementary electric charge. It is a fundamental particle that comprises the ordinary matter that makes up the universe, along with up qua ...
, and no
neutron
The neutron is a subatomic particle, symbol or , that has no electric charge, and a mass slightly greater than that of a proton. The Discovery of the neutron, neutron was discovered by James Chadwick in 1932, leading to the discovery of nucle ...
s.
Hydrogen gas was first produced artificially in the 17th century by the reaction of acids with metals.
Henry Cavendish, in 1766–1781, identified hydrogen gas as a distinct substance and discovered its property of producing water when burned; hence its name means 'water-former' in Greek. Understanding the colors of light absorbed and emitted by hydrogen was a crucial part of developing
quantum mechanics
Quantum mechanics is the fundamental physical Scientific theory, theory that describes the behavior of matter and of light; its unusual characteristics typically occur at and below the scale of atoms. Reprinted, Addison-Wesley, 1989, It is ...
.
Hydrogen, typically
nonmetallic except under
extreme pressure, readily forms
covalent bonds
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atom ...
with most nonmetals, contributing to the formation of compounds like water and various organic substances. Its role is crucial in
acid-base reactions, which mainly involve proton exchange among soluble molecules. In
ionic compounds
In chemistry, a salt or ionic compound is a chemical compound consisting of an assembly of positively charged ions ( cations) and negatively charged ions (anions), which results in a compound with no net electric charge (electrically neutral). ...
, hydrogen can take the form of either a negatively charged
anion
An ion () is an atom or molecule with a net electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by conven ...
, where it is known as
hydride
In chemistry, a hydride is formally the anion of hydrogen (H−), a hydrogen ion with two electrons. In modern usage, this is typically only used for ionic bonds, but it is sometimes (and has been more frequently in the past) applied to all che ...
, or as a positively charged
cation
An ion () is an atom or molecule with a net electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convent ...
, , called a proton. Although tightly bonded to water molecules, protons strongly affect the behavior of
aqueous solution
An aqueous solution is a solution in which the solvent is water. It is mostly shown in chemical equations by appending (aq) to the relevant chemical formula. For example, a solution of table salt, also known as sodium chloride (NaCl), in water ...
s, as reflected in the importance of pH. Hydride, on the other hand, is rarely observed because it tends to deprotonate solvents, yielding .
In the
early universe, neutral hydrogen atoms formed about 370,000 years after the
Big Bang
The Big Bang is a physical theory that describes how the universe expanded from an initial state of high density and temperature. Various cosmological models based on the Big Bang concept explain a broad range of phenomena, including th ...
as the universe expanded and plasma had cooled enough for electrons to remain bound to protons. Once stars formed most of the atoms in the
intergalactic medium re-ionized.
Nearly all
hydrogen production
Hydrogen gas is produced by several industrial methods. Nearly all of the world's current supply of hydrogen is created from fossil fuels. Article in press. Most hydrogen is ''gray hydrogen'' made through steam methane reforming. In this process, ...
is done by transforming fossil fuels, particularly
steam reforming
Steam reforming or steam methane reforming (SMR) is a method for producing syngas (hydrogen and carbon monoxide) by reaction of hydrocarbons with water. Commonly, natural gas is the feedstock. The main purpose of this technology is often hydrogen ...
of
natural gas
Natural gas (also fossil gas, methane gas, and gas) is a naturally occurring compound of gaseous hydrocarbons, primarily methane (95%), small amounts of higher alkanes, and traces of carbon dioxide and nitrogen, hydrogen sulfide and helium ...
. It can also be produced from water or saline by
electrolysis
In chemistry and manufacturing, electrolysis is a technique that uses Direct current, direct electric current (DC) to drive an otherwise non-spontaneous chemical reaction. Electrolysis is commercially important as a stage in the separation of c ...
, but this process is more expensive. Its main industrial uses include
fossil fuel
A fossil fuel is a flammable carbon compound- or hydrocarbon-containing material formed naturally in the Earth's crust from the buried remains of prehistoric organisms (animals, plants or microplanktons), a process that occurs within geolog ...
processing and
ammonia production for fertilizer. Emerging uses for hydrogen include the use of
fuel cell
A fuel cell is an electrochemical cell that converts the chemical energy of a fuel (often hydrogen fuel, hydrogen) and an oxidizing agent (often oxygen) into electricity through a pair of redox reactions. Fuel cells are different from most bat ...
s to generate electricity.
Properties
Atomic hydrogen
Electron energy levels
The
ground state energy level
A quantum mechanics, quantum mechanical system or particle that is bound state, bound—that is, confined spatially—can only take on certain discrete values of energy, called energy levels. This contrasts with classical mechanics, classical pa ...
of the electron in a hydrogen atom is −13.6
eV, equivalent to an
ultraviolet
Ultraviolet radiation, also known as simply UV, is electromagnetic radiation of wavelengths of 10–400 nanometers, shorter than that of visible light, but longer than X-rays. UV radiation is present in sunlight and constitutes about 10% of ...
photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are massless particles that can ...
of roughly 91 nm wavelength. The energy levels of hydrogen are referred to by consecutive
quantum number
In quantum physics and chemistry, quantum numbers are quantities that characterize the possible states of the system.
To fully specify the state of the electron in a hydrogen atom, four quantum numbers are needed. The traditional set of quantu ...
s, with
being the ground state. The
hydrogen spectral series corresponds to emission of light due to transitions from higher to lower energy levels. Each energy level is further split by
spin interactions between the electron and proton into 4
hyperfine levels.
High precision values for the hydrogen atom energy levels are required for definitions of physical constants. Quantum calculations have identified 9 contributions to the energy levels. The eigenvalue from the
Dirac equation
In particle physics, the Dirac equation is a relativistic wave equation derived by British physicist Paul Dirac in 1928. In its free form, or including electromagnetic interactions, it describes all spin-1/2 massive particles, called "Dirac ...
is the largest contribution. Other terms include
relativistic recoil, the
self-energy, and the
vacuum polarization terms.
Isotopes

Hydrogen has three naturally occurring isotopes, denoted , and . Other, highly unstable nuclei ( to ) have been synthesized in the laboratory but not observed in nature.
is the most common hydrogen isotope, with an abundance of >99.98%. Because the
nucleus of this isotope consists of only a single proton, it is given the descriptive but rarely used formal name ''protium''. It is the only stable isotope with no neutrons; see
diproton for a discussion of why others do not exist.
, the other stable hydrogen isotope, is known as
deuterium
Deuterium (hydrogen-2, symbol H or D, also known as heavy hydrogen) is one of two stable isotopes of hydrogen; the other is protium, or hydrogen-1, H. The deuterium nucleus (deuteron) contains one proton and one neutron, whereas the far more c ...
and contains one proton and one
neutron
The neutron is a subatomic particle, symbol or , that has no electric charge, and a mass slightly greater than that of a proton. The Discovery of the neutron, neutron was discovered by James Chadwick in 1932, leading to the discovery of nucle ...
in the nucleus. Nearly all deuterium nuclei in the universe is thought to have been produced at the time of the
Big Bang
The Big Bang is a physical theory that describes how the universe expanded from an initial state of high density and temperature. Various cosmological models based on the Big Bang concept explain a broad range of phenomena, including th ...
, and has endured since then. Deuterium is not radioactive, and is not a significant toxicity hazard. Water enriched in molecules that include deuterium instead of normal hydrogen is called
heavy water
Heavy water (deuterium oxide, , ) is a form of water (molecule), water in which hydrogen atoms are all deuterium ( or D, also known as ''heavy hydrogen'') rather than the common hydrogen-1 isotope (, also called ''protium'') that makes up most o ...
. Deuterium and its compounds are used as a non-radioactive label in chemical experiments and in solvents for -
NMR spectroscopy. Heavy water is used as a
neutron moderator
In nuclear engineering, a neutron moderator is a medium that reduces the speed of fast neutrons, ideally without capturing any, leaving them as thermal neutrons with only minimal (thermal) kinetic energy. These thermal neutrons are immensely ...
and coolant for nuclear reactors. Deuterium is also a potential fuel for commercial
nuclear fusion
Nuclear fusion is a nuclear reaction, reaction in which two or more atomic nuclei combine to form a larger nuclei, nuclei/neutrons, neutron by-products. The difference in mass between the reactants and products is manifested as either the rele ...
.
is known as
tritium
Tritium () or hydrogen-3 (symbol T or H) is a rare and radioactive isotope of hydrogen with a half-life of ~12.33 years. The tritium nucleus (t, sometimes called a ''triton'') contains one proton and two neutrons, whereas the nucleus of the ...
and contains one proton and two neutrons in its nucleus. It is radioactive, decaying into
helium-3 through
beta decay
In nuclear physics, beta decay (β-decay) is a type of radioactive decay in which an atomic nucleus emits a beta particle (fast energetic electron or positron), transforming into an isobar of that nuclide. For example, beta decay of a neutron ...
with a
half-life Half-life is a mathematical and scientific description of exponential or gradual decay.
Half-life, half life or halflife may also refer to:
Film
* Half-Life (film), ''Half-Life'' (film), a 2008 independent film by Jennifer Phang
* ''Half Life: ...
of 12.32 years.
It is radioactive enough to be used in
luminous paint to enhance the visibility of data displays, such as for painting the hands and dial-markers of watches. The watch glass prevents the small amount of radiation from escaping the case.
Small amounts of tritium are produced naturally by cosmic rays striking atmospheric gases; tritium has also been released in
nuclear weapons tests. It is used in nuclear fusion, as a tracer in
isotope geochemistry
Isotope geochemistry is an aspect of geology based upon the study of natural variations in the relative abundances of isotopes of various Chemical element, elements. Variations in isotopic abundance are measured by isotope-ratio mass spectrometry, ...
, and in specialized
self-powered lighting
Tritium radioluminescence is the use of gaseous tritium, a radioactive isotope of hydrogen, to create visible light. Tritium emits electrons through beta decay and, when they interact with a phosphor material, light is emitted through the proc ...
devices. Tritium has also been used in chemical and biological labeling experiments as a
radiolabel.
Unique among the elements, distinct names are assigned to its isotopes in common use. During the early study of radioactivity, heavy radioisotopes were given their own names, but these are mostly no longer used. The symbols D and T (instead of and ) are sometimes used for deuterium and tritium, but the symbol P was already used for
phosphorus
Phosphorus is a chemical element; it has Chemical symbol, symbol P and atomic number 15. All elemental forms of phosphorus are highly Reactivity (chemistry), reactive and are therefore never found in nature. They can nevertheless be prepared ar ...
and thus was not available for protium. In its
nomenclatural guidelines, the
International Union of Pure and Applied Chemistry
The International Union of Pure and Applied Chemistry (IUPAC ) is an international federation of National Adhering Organizations working for the advancement of the chemical sciences, especially by developing nomenclature and terminology. It is ...
(IUPAC) allows any of D, T, , and to be used, though and are preferred.
Antihydrogen () is the
antimatter
In modern physics, antimatter is defined as matter composed of the antiparticles (or "partners") of the corresponding subatomic particle, particles in "ordinary" matter, and can be thought of as matter with reversed charge and parity, or go ...
counterpart to hydrogen. It consists of an
antiproton
The antiproton, , (pronounced ''p-bar'') is the antiparticle of the proton. Antiprotons are stable, but they are typically short-lived, since any collision with a proton will cause both particles to be annihilated in a burst of energy.
The exis ...
with a
positron
The positron or antielectron is the particle with an electric charge of +1''elementary charge, e'', a Spin (physics), spin of 1/2 (the same as the electron), and the same Electron rest mass, mass as an electron. It is the antiparticle (antimatt ...
. Antihydrogen is the only type of antimatter atom to have been produced .
The
exotic atom muonium (symbol Mu), composed of an anti
muon
A muon ( ; from the Greek letter mu (μ) used to represent it) is an elementary particle similar to the electron, with an electric charge of −1 '' e'' and a spin of ''ħ'', but with a much greater mass. It is classified as a ...
and an
electron
The electron (, or in nuclear reactions) is a subatomic particle with a negative one elementary charge, elementary electric charge. It is a fundamental particle that comprises the ordinary matter that makes up the universe, along with up qua ...
, is analogous hydrogen and IUPAC nomenclature incorporates such hypothetical compounds as muonium chloride (MuCl) and sodium muonide (NaMu), analogous to
hydrogen chloride
The Chemical compound, compound hydrogen chloride has the chemical formula and as such is a hydrogen halide. At room temperature, it is a colorless gas, which forms white fumes of hydrochloric acid upon contact with atmospheric water vapor. Hyd ...
and
sodium hydride respectively.
Dihydrogen
Under
standard conditions, hydrogen is a
gas of
diatomic molecule
Diatomic molecules () are molecules composed of only two atoms, of the same or different chemical elements. If a diatomic molecule consists of two atoms of the same element, such as hydrogen () or oxygen (), then it is said to be homonuclear mol ...
s with the
formula
In science, a formula is a concise way of expressing information symbolically, as in a mathematical formula or a ''chemical formula''. The informal use of the term ''formula'' in science refers to the general construct of a relationship betwe ...
, officially called "dihydrogen", but also called "molecular hydrogen",
or simply hydrogen. Dihydrogen is a colorless, odorless, flammable gas.
Combustion
Hydrogen gas is highly flammable, reacting with
oxygen
Oxygen is a chemical element; it has chemical symbol, symbol O and atomic number 8. It is a member of the chalcogen group (periodic table), group in the periodic table, a highly reactivity (chemistry), reactive nonmetal (chemistry), non ...
in air, to produce liquid water:
:
The
amount of heat released per
mole of hydrogen is −286 kJ or 141.865 MJ for a kilogram mass.
Hydrogen gas forms explosive mixtures with air in concentrations from 4–74% and with chlorine at 5–95%. The hydrogen
autoignition temperature, the temperature of spontaneous ignition in air, is . In a high-pressure hydrogen leak, the shock wave from the leak itself can heat air to the autoignition temperature, leading to flaming and possibly explosion.
Hydrogen flames emit faint blue and
ultraviolet
Ultraviolet radiation, also known as simply UV, is electromagnetic radiation of wavelengths of 10–400 nanometers, shorter than that of visible light, but longer than X-rays. UV radiation is present in sunlight and constitutes about 10% of ...
light.
Flame detectors are used to detect hydrogen fires as they are nearly invisible to the naked eye in daylight.
Spin isomers
Molecular exists as two
nuclear isomers that differ in the
spin states of their nuclei.
In the orthohydrogen form, the spins of the two nuclei are parallel, forming a spin
triplet state
In quantum mechanics, a triplet state, or spin triplet, is the quantum state of an object such as an electron, atom, or molecule, having a quantum spin ''S'' = 1. It has three allowed values of the spin's projection along a given axis ''m''S = � ...
having a
total molecular spin ; in the parahydrogen form the spins are antiparallel and form a spin
singlet state
In quantum mechanics, a singlet state usually refers to a system in which all electrons are paired. The term 'singlet' originally meant a linked set of particles whose net angular momentum is zero, that is, whose overall spin quantum number s=0. A ...
having spin
. The equilibrium ratio of ortho- to para-hydrogen depends on temperature. At room temperature or warmer, equilibrium hydrogen gas contains about 25% of the para form and 75% of the ortho form.
The ortho form is an
excited state
In quantum mechanics
Quantum mechanics is the fundamental physical Scientific theory, theory that describes the behavior of matter and of light; its unusual characteristics typically occur at and below the scale of atoms. Reprinted, Add ...
, having higher energy than the para form by 1.455 kJ/mol,
and it converts to the para form over the course of several minutes when cooled to low temperature. The thermal properties of these isomers differ because each has distinct
rotational quantum states.
The ortho-to-para ratio in is an important consideration in the
liquefaction and storage of
liquid hydrogen
Liquid hydrogen () is the liquid state of the element hydrogen. Hydrogen is found naturally in the molecule, molecular H2 form.
To exist as a liquid, H2 must be cooled below its critical point (thermodynamics), critical point of 33 Kelvins, ...
: the conversion from ortho to para is
exothermic and produces sufficient heat to evaporate most of the liquid if not converted first to parahydrogen during the cooling process.
Catalyst
Catalysis () is the increase in rate of a chemical reaction due to an added substance known as a catalyst (). Catalysts are not consumed by the reaction and remain unchanged after it. If the reaction is rapid and the catalyst recycles quick ...
s for the ortho-para interconversion, such as
ferric oxide and
activated carbon
Activated carbon, also called activated charcoal, is a form of carbon commonly used to filter contaminants from water and air, among many other uses. It is processed (activated) to have small, low-volume pores that greatly increase the surface ar ...
compounds, are used during hydrogen cooling to avoid this loss of liquid.
Phases

Liquid hydrogen
Liquid hydrogen () is the liquid state of the element hydrogen. Hydrogen is found naturally in the molecule, molecular H2 form.
To exist as a liquid, H2 must be cooled below its critical point (thermodynamics), critical point of 33 Kelvins, ...
can exist at temperatures below hydrogen's
critical point of 33
K. However, for it to be in a fully liquid state at
atmospheric pressure
Atmospheric pressure, also known as air pressure or barometric pressure (after the barometer), is the pressure within the atmosphere of Earth. The standard atmosphere (symbol: atm) is a unit of pressure defined as , which is equivalent to 1,013. ...
, H
2 needs to be cooled to . Hydrogen was liquefied by
James Dewar in 1898 by using
regenerative cooling and his invention, the
vacuum flask.
Liquid hydrogen becomes
solid hydrogen at
standard pressure below hydrogen's
melting point
The melting point (or, rarely, liquefaction point) of a substance is the temperature at which it changes state of matter, state from solid to liquid. At the melting point the solid and liquid phase (matter), phase exist in Thermodynamic equilib ...
of . Distinct solid phases exist, known as Phase I through Phase V, each exhibiting a characteristic molecular arrangement.
Liquid and solid phases can exist in combination at the
triple point
In thermodynamics, the triple point of a substance is the temperature and pressure at which the three Phase (matter), phases (gas, liquid, and solid) of that substance coexist in thermodynamic equilibrium.. It is that temperature and pressure at ...
, a substance known as
slush hydrogen.
Metallic hydrogen, a phase obtained at extremely high pressures (in excess of ), is an electrical conductor. It is believed to exist deep within
giant planets like
Jupiter
Jupiter is the fifth planet from the Sun and the List of Solar System objects by size, largest in the Solar System. It is a gas giant with a Jupiter mass, mass more than 2.5 times that of all the other planets in the Solar System combined a ...
.
When
ionized, hydrogen becomes a
plasma. This is the form in which hydrogen exists within
star
A star is a luminous spheroid of plasma (physics), plasma held together by Self-gravitation, self-gravity. The List of nearest stars and brown dwarfs, nearest star to Earth is the Sun. Many other stars are visible to the naked eye at night sk ...
s.
Thermal and physical properties
History
18th century
In 1671, Irish scientist
Robert Boyle discovered and described the reaction between
iron
Iron is a chemical element; it has symbol Fe () and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, forming much of Earth's o ...
filings and dilute
acid
An acid is a molecule or ion capable of either donating a proton (i.e. Hydron, hydrogen cation, H+), known as a Brønsted–Lowry acid–base theory, Brønsted–Lowry acid, or forming a covalent bond with an electron pair, known as a Lewis ...
s, which results in the production of hydrogen gas.
Boyle did not note that the gas was inflammable, but hydrogen would play a key role in overturning the
phlogiston theory
The phlogiston theory, a superseded scientific theory, postulated the existence of a fire-like element dubbed phlogiston () contained within combustible bodies and released during combustion. The name comes from the Ancient Greek (''burnin ...
of combustion.
In 1766,
Henry Cavendish was the first to recognize hydrogen gas as a discrete substance, by naming the gas from a
metal-acid reaction "inflammable air". He speculated that "inflammable air" was in fact identical to the hypothetical substance "
phlogiston"
and further finding in 1781 that the gas produces water when burned. He is usually given credit for the discovery of hydrogen as an element.
In 1783,
Antoine Lavoisier
Antoine-Laurent de Lavoisier ( ; ; 26 August 17438 May 1794), When reduced without charcoal, it gave off an air which supported respiration and combustion in an enhanced way. He concluded that this was just a pure form of common air and that i ...
identified the element that came to be known as hydrogen when he and
Laplace reproduced Cavendish's finding that water is produced when hydrogen is burned.
Lavoisier produced hydrogen for his experiments on mass conservation by treating metallic
iron
Iron is a chemical element; it has symbol Fe () and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, forming much of Earth's o ...
with a stream of H
2O through an incandescent iron tube heated in a fire. Anaerobic oxidation of iron by the protons of water at high temperature can be schematically represented by the set of following reactions:
*
*
*
Many metals react similarly with water leading to the production of hydrogen. In some situations, this H
2-producing process is problematic as is the case of zirconium cladding on nuclear fuel rods.
19th century
By 1806 hydrogen was used to fill balloons.
François Isaac de Rivaz built the first
de Rivaz engine, an internal combustion engine powered by a mixture of hydrogen and oxygen in 1806.
Edward Daniel Clarke invented the hydrogen gas blowpipe in 1819. The
Döbereiner's lamp and
limelight were invented in 1823. Hydrogen was
liquefied for the first time by
James Dewar in 1898 by using
regenerative cooling and his invention, the
vacuum flask. He produced
solid hydrogen the next year.
One of the first
quantum
In physics, a quantum (: quanta) is the minimum amount of any physical entity (physical property) involved in an interaction. The fundamental notion that a property can be "quantized" is referred to as "the hypothesis of quantization". This me ...
effects to be explicitly noticed (but not understood at the time) was
James Clerk Maxwell
James Clerk Maxwell (13 June 1831 – 5 November 1879) was a Scottish physicist and mathematician who was responsible for the classical theory of electromagnetic radiation, which was the first theory to describe electricity, magnetism an ...
's observation that the
specific heat capacity
In thermodynamics, the specific heat capacity (symbol ) of a substance is the amount of heat that must be added to one unit of mass of the substance in order to cause an increase of one unit in temperature. It is also referred to as massic heat ...
of unaccountably departs from that of a
diatomic
Diatomic molecules () are molecules composed of only two atoms, of the same or different chemical elements. If a diatomic molecule consists of two atoms of the same element, such as hydrogen () or oxygen (), then it is said to be homonuclear mol ...
gas below room temperature and begins to increasingly resemble that of a monatomic gas at cryogenic temperatures. According to quantum theory, this behavior arises from the spacing of the (quantized) rotational energy levels, which are particularly wide-spaced in because of its low mass. These widely spaced levels inhibit equal partition of heat energy into rotational motion in hydrogen at low temperatures. Diatomic gases composed of heavier atoms do not have such widely spaced levels and do not exhibit the same effect.
20th century
The existence of the
hydride anion was suggested by
Gilbert N. Lewis in 1916 for group 1 and 2 salt-like compounds. In 1920, Moers electrolyzed molten
lithium hydride (LiH), producing a
stoichiometric quantity of hydrogen at the anode.

Because of its simple atomic structure, consisting only of a proton and an electron, the
hydrogen atom, together with the spectrum of light produced from it or absorbed by it, has been central to the
development of the theory of atomic structure. The energy levels of hydrogen can be calculated fairly accurately using the
Bohr model
In atomic physics, the Bohr model or Rutherford–Bohr model was a model of the atom that incorporated some early quantum concepts. Developed from 1911 to 1918 by Niels Bohr and building on Ernest Rutherford's nuclear Rutherford model, model, i ...
of the atom, in which the electron "orbits" the proton, like how Earth orbits the Sun. However, the electron and proton are held together by electrostatic attraction, while planets and celestial objects are held by gravity. Due to the discretization of angular momentum postulated in early
quantum mechanics
Quantum mechanics is the fundamental physical Scientific theory, theory that describes the behavior of matter and of light; its unusual characteristics typically occur at and below the scale of atoms. Reprinted, Addison-Wesley, 1989, It is ...
by Bohr, the electron in the Bohr model can only occupy certain allowed distances from the proton, and therefore only certain allowed energies.
Hydrogen's unique position as the only neutral atom for which the Schrödinger equation can be directly solved, has significantly contributed to the understanding of quantum mechanics through the exploration of its energetics.
Furthermore, study of the corresponding simplicity of the hydrogen molecule and the corresponding cation H2+, brought understanding of the nature of the chemical bond, which followed shortly after the quantum mechanical treatment of the hydrogen atom had been developed in the mid-1920s.
Hydrogen-lifted airship
Because is only 7% the density of air, it was once widely used as a lifting gas in balloons and airships.
The first hydrogen-filled balloon was invented by Jacques Charles in 1783. Hydrogen provided the lift for the first reliable form of air-travel following the 1852 invention of the first hydrogen-lifted airship by Henri Giffard. German count Ferdinand von Zeppelin promoted the idea of rigid airships lifted by hydrogen that later were called Zeppelins; the first of which had its maiden flight in 1900.
Regularly scheduled flights started in 1910 and by the outbreak of World War I in August 1914, they had carried 35,000 passengers without a serious incident. Hydrogen-lifted airships in the form of blimps were used as observation platforms and bombers during the War II, especially on the US Eastern seaboard.
The first non-stop transatlantic crossing was made by the British airship ''R34 (airship), R34'' in 1919 and regular passenger service resumed in the 1920s. Hydrogen was used in the LZ 129 Hindenburg, ''Hindenburg'' airship, which caught fire over New Jersey on 6 May 1937.
The hydrogen that filled the airship was ignited, possibly by static electricity, and burst into flames. Following this Hindenburg disaster, commercial hydrogen airship travel Rigid airship#Demise, ceased. Hydrogen is still used, in preference to non-flammable but more expensive helium, as a lifting gas for Weather balloon#Materials and equipment, weather balloons.
Deuterium and tritium
Deuterium was discovered in December 1931 by Harold Urey, and
tritium
Tritium () or hydrogen-3 (symbol T or H) is a rare and radioactive isotope of hydrogen with a half-life of ~12.33 years. The tritium nucleus (t, sometimes called a ''triton'') contains one proton and two neutrons, whereas the nucleus of the ...
was prepared in 1934 by Ernest Rutherford, Mark Oliphant, and Paul Harteck.
Heavy water, which consists of deuterium in the place of regular hydrogen, was discovered by Urey's group in 1932.
Chemistry
Reactions of H2

is relatively unreactive. The thermodynamic basis of this low reactivity is the very strong H–H bond, with a bond dissociation energy of 435.7 kJ/mol. It does form coordination complexes called dihydrogen complexes. These species provide insights into the early steps in the interactions of hydrogen with metal catalysts. According to neutron diffraction, the metal and two H atoms form a triangle in these complexes. The H-H bond remains intact but is elongated. They are acidic.
Although exotic on Earth, the ion is common in the universe. It is a triangular species, like the aforementioned dihydrogen complexes. It is known as Trihydrogen cation, protonated molecular hydrogen or the trihydrogen cation.
Hydrogen reacts with chlorine to produce HCl and with bromine to produce Hydrogen bromide, HBr by a chain reaction. The reaction requires initiation. For example in the case of Br
2, the diatomic molecule is broken into atoms, . Propagating reactions consume hydrogen molecules and produce HBr, as well as Br and H atoms:
:
:
Finally the terminating reaction:
:
:.
consumes the remaining atoms.
The addition of H
2 to unsaturated organic compounds, such as alkenes and alkynes, is called hydrogenation. Even if the reaction is exothermic, energetically favorable, it does not take place even at higher temperatures. In the presence of a catalyst like finely divided platinum or nickel, the reaction proceeds at room temperature.
Hydrogen-containing compounds
Hydrogen can exist in both +1 and −1 oxidation states, forming compounds through ionic bonding, ionic and covalent bonding. It is a part of a wide range of substances, including water, hydrocarbons, and numerous other
organic compounds
Some chemical authorities define an organic compound as a chemical compound that contains a carbon–hydrogen or carbon–carbon bond; others consider an organic compound to be any chemical compound that contains carbon. For example, carbon-co ...
.
The H
+ ion—commonly referred to as a proton due to its single proton and absence of electrons—is central to Acid–base reaction, acid–base chemistry, although the proton does not move freely. In the Brønsted–Lowry acids, Brønsted–Lowry framework, acids are defined by their ability to donate H
+ ions to bases.
Hydrogen forms a vast variety of compounds with carbon known as hydrocarbons, and an even greater diversity with other elements (heteroatoms), giving rise to the broad class of organic compounds often associated with living organisms.

Hydrogen compounds with hydrogen in the oxidation state −1 are known as hydrides, which are usually formed between hydrogen and metals. The hydrides can be ionic (aka saline), covalent, nor metallic. With heating, H
2 reacts efficiently with the alkali and alkaline earth metals to give the Hydride#Ionic_hydrides, ionic hydrides of the formula MH and MH
2, respectively. These salt-like crystalline compounds have high melting points and all react with water to liberate hydrogen. Covalent hydrides are include boranes and polymeric aluminium hydride. Transition metals form metal hydrides via continuous dissolution of hydrogen into the metal.
[ A well known hydride is lithium aluminium hydride, the anion carries hydridic centers firmly attached to the Al(III). Perhaps the most extensive series of hydrides are the boranes, compounds consisting only of boron and hydrogen.]
Hydrogen bonding
When bonded to a more electronegative element, particularly fluorine, oxygen
Oxygen is a chemical element; it has chemical symbol, symbol O and atomic number 8. It is a member of the chalcogen group (periodic table), group in the periodic table, a highly reactivity (chemistry), reactive nonmetal (chemistry), non ...
, or nitrogen, hydrogen can participate in a form of medium-strength noncovalent bonding with another electronegative element with a lone pair like oxygen or nitrogen, a phenomenon called hydrogen bonding that is critical to the stability of many biological molecules. Hydrogen bonding alters molecule structures, viscosity, solubility, as well as melting and boiling points even protein folding dynamics.
Protons and acids
 In water, hydrogen bonding plays an important role in reaction thermodynamics. A hydrogen bond can shift over to proton transfer.
Under the Brønsted–Lowry acid–base theory, acids are proton donors, while bases are proton acceptors.
A bare proton, essentially cannot exist in anything other than a vacuum. Otherwise it attaches to other atoms, ions, or molecules. Even species as inert as methane can be protonated. The term 'proton' is used loosely and metaphorically to refer to refer to solvated " without any implication that any single protons exist freely as a species. To avoid the implication of the naked proton in solution, acidic aqueous solutions are sometimes considered to contain the "hydronium ion" () or still more accurately, .
In water, hydrogen bonding plays an important role in reaction thermodynamics. A hydrogen bond can shift over to proton transfer.
Under the Brønsted–Lowry acid–base theory, acids are proton donors, while bases are proton acceptors.
A bare proton, essentially cannot exist in anything other than a vacuum. Otherwise it attaches to other atoms, ions, or molecules. Even species as inert as methane can be protonated. The term 'proton' is used loosely and metaphorically to refer to refer to solvated " without any implication that any single protons exist freely as a species. To avoid the implication of the naked proton in solution, acidic aqueous solutions are sometimes considered to contain the "hydronium ion" () or still more accurately, .
Occurrence
Cosmic
 Hydrogen, as atomic H, is the most Natural abundance, abundant
Hydrogen, as atomic H, is the most Natural abundance, abundant chemical element
A chemical element is a chemical substance whose atoms all have the same number of protons. The number of protons is called the atomic number of that element. For example, oxygen has an atomic number of 8: each oxygen atom has 8 protons in its ...
in the universe, making up 75% of Baryon, normal matter by mass and >90% by number of atoms. In the early universe, the protons formed in the first second after the Big Bang
The Big Bang is a physical theory that describes how the universe expanded from an initial state of high density and temperature. Various cosmological models based on the Big Bang concept explain a broad range of phenomena, including th ...
; neutral hydrogen atoms formed about 370,000 years later during the Recombination (cosmology), recombination epoch as the universe expanded and plasma had cooled enough for electrons to remain bound to protons.
In astrophysics, neutral hydrogen in the interstellar medium is called ''H I'' and ionized hydrogen is called ''H II''. Radiation from stars ionizes H I to H II, creating Strömgren sphere, spheres of ionized H II around stars. In the chronology of the universe neutral hydrogen dominated until the birth of stars during the era of reionization led to bubbles of ionized hydrogen that grew and merged over 500 million of years.
They are the source of the 21-cm hydrogen line at 1420 MHz that is detected in order to probe primordial hydrogen. The large amount of neutral hydrogen found in the damped Lyman-alpha systems is thought to dominate the Physical cosmology, cosmological baryonic density of the universe up to a redshift of ''z'' = 4.
Hydrogen is found in great abundance in stars and gas giant planets. Molecular clouds of are associated with star formation. Hydrogen plays a vital role in powering star
A star is a luminous spheroid of plasma (physics), plasma held together by Self-gravitation, self-gravity. The List of nearest stars and brown dwarfs, nearest star to Earth is the Sun. Many other stars are visible to the naked eye at night sk ...
s through the proton-proton reaction in lower-mass stars, and through the CNO cycle of nuclear fusion
Nuclear fusion is a nuclear reaction, reaction in which two or more atomic nuclei combine to form a larger nuclei, nuclei/neutrons, neutron by-products. The difference in mass between the reactants and products is manifested as either the rele ...
in case of stars more massive than the Sun.
A molecular form called protonated molecular hydrogen () is found in the interstellar medium, where it is generated by ionization of molecular hydrogen from cosmic rays. This ion has also been observed in the Primary atmosphere, upper atmosphere of Jupiter. The ion is long-lived in outer space due to the low temperature and density. is one of the most abundant ions in the universe, and it plays a notable role in the chemistry of the interstellar medium. Neutral triatomic hydrogen can exist only in an excited form and is unstable.
Terrestrial
Hydrogen is the third most abundant element on the Earth's surface,
Production and storage
Industrial routes
Nearly all of the world's current supply of hydrogen gas () is created from fossil fuels.[ Article in press.] Many methods exist for producing H2, but three dominate commercially: steam reforming often coupled to water-gas shift, partial oxidation of hydrocarbons, and water electrolysis.[
]
Steam reforming
Hydrogen is mainly produced by steam reforming, steam methane reforming (SMR), the reaction of water and methane.
Partial oxidation of hydrocarbons
Other methods for CO and production include partial oxidation of hydrocarbons:
Water electrolysis
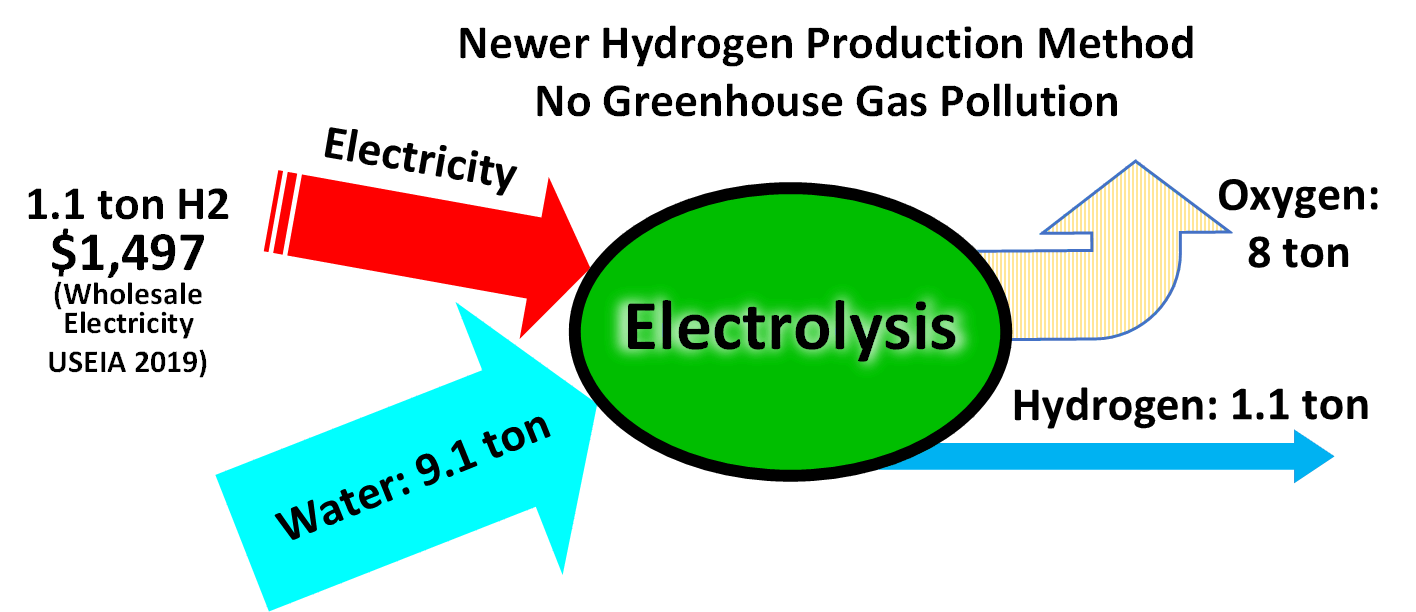 Electrolysis of water is a conceptually simple method of producing hydrogen.
:
Commercial electrolyzers use nickel-based catalysts in strongly alkaline solution. Platinum is a better catalyst but is expensive. The hydrogen created through electrolysis using renewable energy is commonly referred to as "green hydrogen".
Electrolysis of water is a conceptually simple method of producing hydrogen.
:
Commercial electrolyzers use nickel-based catalysts in strongly alkaline solution. Platinum is a better catalyst but is expensive. The hydrogen created through electrolysis using renewable energy is commonly referred to as "green hydrogen".electrolysis
In chemistry and manufacturing, electrolysis is a technique that uses Direct current, direct electric current (DC) to drive an otherwise non-spontaneous chemical reaction. Electrolysis is commercially important as a stage in the separation of c ...
process is more expensive than producing hydrogen from methane without carbon capture and storage.
Methane pyrolysis
Hydrogen can be produced by pyrolysis of natural gas (methane), producing hydrogen gas and solid carbon with the aid a catalyst and 74 kJ/mol input heat:
: (ΔH° = 74 kJ/mol)
The carbon may be sold as a manufacturing feedstock or fuel, or landfilled.
This route could have a lower carbon footprint than existing hydrogen production processes, but mechanisms for removing the carbon and preventing it from reacting with the catalyst remain obstacles for industrial scale use.
Thermochemical
Water splitting is the process by which water is decomposed into its components. Relevant to the biological scenario is this simple equation:
:
The reaction occurs in the Light-dependent reactions, light reactions in all photosynthetic organisms. A few organisms, including the alga ''Chlamydomonas reinhardtii'' and cyanobacteria, have evolved a second step in the dark reactions in which protons and electrons are reduced to form gas by specialized hydrogenases in the chloroplast.
Efforts have been undertaken to genetically modify cyanobacterial hydrogenases to more efficiently generate gas even in the presence of oxygen. Efforts have also been undertaken with genetically modified Biological hydrogen production (Algae), alga in a bioreactor.
Relevant to the thermal water-splitting scenario is this simple equation:
:
More than 200 thermochemical cycles can be used for water splitting. Many of these cycles such as the iron oxide cycle, cerium(IV) oxide–cerium(III) oxide cycle, zinc zinc-oxide cycle, sulfur-iodine cycle, copper-chlorine cycle and hybrid sulfur cycle have been evaluated for their commercial potential to produce hydrogen and oxygen from water and heat without using electricity. A number of labs (including in France, Germany, Greece, Japan, and the United States) are developing thermochemical methods to produce hydrogen from solar energy and water.
Natural routes
Biohydrogen
is produced by enzymes called hydrogenases. This process allows the host organism to use fermentation as a source of energy. These same enzymes also can oxidize H2, such that the host organisms can subsist by reducing oxidized substrates using electrons extracted from H2.
The hydrogenase enzyme feature iron
Iron is a chemical element; it has symbol Fe () and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, forming much of Earth's o ...
or nickel-iron centers at their active sites. The natural cycle of hydrogen production and consumption by organisms is called the hydrogen cycle.[
Confirming the existence of hydrogenases in the human gut, occurs in human breath. The concentration in the breath of fasting people at rest is typically less than 5 parts per million (ppm) but can be 50 ppm when people with intestinal disorders consume molecules they cannot absorb during diagnostic hydrogen breath tests.
]
Serpentinization
Serpentinization is a geological mechanism that produce highly Reduction (chemistry), reducing conditions.iron
Iron is a chemical element; it has symbol Fe () and atomic number 26. It is a metal that belongs to the first transition series and group 8 of the periodic table. It is, by mass, the most common element on Earth, forming much of Earth's o ...
and steel in Anoxic waters, oxygen-free groundwater and in reducing soils below the water table.
Laboratory syntheses
is produced in laboratory settings, such as in the small-scale electrolysis of water using metal electrodes and water containing an electrolyte, which liberates hydrogen gas at the cathode:
Storage
If H2 is to be used as an energy source, its storage is important. It dissolves only poorly in solvents. For example, at room temperature and 0.1 MPascal (unit), pascal, ca. 0.05 moles dissolves in one kilogram of diethyl ether.
Applications
Petrochemical industry
Large quantities of are used in the "upgrading" of fossil fuels. Key consumers of include hydrodesulfurization, and hydrocracking. Many of these reactions can be classified as hydrogenolysis, i.e., the cleavage of bonds by hydrogen. Illustrative is the separation of sulfur from liquid fossil fuels:
Hydrogenation
Hydrogenation, the addition of to various substrates, is done on a large scale. Hydrogenation of produces ammonia by the Haber process:
Fuel
The potential for using hydrogen (H2) as a fuel has been widely discussed. Hydrogen can be used in fuel cells to produce electricity, or burned to generate heat.
Liquid hydrogen
Liquid hydrogen () is the liquid state of the element hydrogen. Hydrogen is found naturally in the molecule, molecular H2 form.
To exist as a liquid, H2 must be cooled below its critical point (thermodynamics), critical point of 33 Kelvins, ...
and liquid oxygen together serve as cryogenic propellants in liquid-propellant rockets, as in the RS-25, Space Shuttle main engines. NASA has investigated the use of rocket propellant made from atomic hydrogen, boron or carbon that is frozen into solid molecular hydrogen particles suspended in liquid helium. Upon warming, the mixture vaporizes to allow the atomic species to recombine, heating the mixture to high temperature.
Hydrogen produced when there is a surplus of Variable renewable energy, variable renewable electricity could in principle be stored and later used to generate heat or to re-generate electricity. It can be further transformed into synthetic fuels such as ammonia and methanol. Disadvantages of hydrogen fuel include high costs of storage and distribution due to hydrogen's explosivity, its large volume compared to other fuels, and its tendency to make pipes brittle.
Nickel–hydrogen battery
The very long-lived, rechargeable nickel–hydrogen battery developed for satellite power systems uses pressurized gaseous H2. The International Space Station, 2001 Mars Odyssey, Mars Odyssey and the Mars Global Surveyor are equipped with nickel-hydrogen batteries. In the dark part of its orbit, the Hubble Space Telescope is also powered by nickel-hydrogen batteries, which were finally replaced in May 2009, more than 19 years after launch and 13 years beyond their design life.
Semiconductor industry
Hydrogen is employed to saturate broken ("dangling") bonds of amorphous silicon and amorphous carbon that helps stabilizing material properties. Hydrogen, introduced as a unintended side-effect of production, acts as a shallow electron donor leading to N-type semiconductor, n-type conductivity in ZnO, with important uses in transducers and phosphors. Detailed analysis of ZnO and of MgO show evidence of four and six-fold hydrogen multicentre bonds.
The doping behavior of hydrogen varies with the material.
Niche and evolving uses
Other than the uses mentioned above, hydrogen is also used in smaller scales in the following applications:
*Shielding gas: Hydrogen is used as a shielding gas in welding methods such as atomic hydrogen welding.
*Coolant: Hydrogen is used as a coolant in large power stations generators due to its high thermal conductivity and low density. The first hydrogen-cooled turbogenerator went into service using gaseous hydrogen as a coolant in the rotor and the stator in 1937 at Dayton, Ohio, Dayton, Ohio.
*Cryogenic research: Liquid is used in cryogenic research, including superconductivity studies.
*Leak detection: Pure or mixed with nitrogen (sometimes called forming gas), hydrogen is a tracer gas for Leak detection, detection of minute leaks. Applications can be found in the automotive, chemical, power generation, aerospace, and telecommunications industries. Hydrogen is an authorized food additive (E 949) that allows food package leak testing, as well as having anti-oxidizing properties.
*Neutron moderation: Deuterium (hydrogen-2) is used in CANDU reactor, nuclear fission applications as a neutron moderator, moderator to slow neutron
The neutron is a subatomic particle, symbol or , that has no electric charge, and a mass slightly greater than that of a proton. The Discovery of the neutron, neutron was discovered by James Chadwick in 1932, leading to the discovery of nucle ...
s.
*Nuclear fusion fuel: Deuterium is used in nuclear fusion
Nuclear fusion is a nuclear reaction, reaction in which two or more atomic nuclei combine to form a larger nuclei, nuclei/neutrons, neutron by-products. The difference in mass between the reactants and products is manifested as either the rele ...
reactions.
Safety and precautions
In hydrogen pipelines and steel storage vessels, hydrogen molecules are prone to react with metals, causing hydrogen embrittlement and leaks in the pipeline or storage vessel.[Text was copied from this source, which is available under a creativecommons:by/4.0/, Creative Commons Attribution 4.0 International License] Since it is lighter than air, hydrogen does not easily accumulate to form a combustible gas mixture.
See also
* Combined cycle hydrogen power plant
*
*
*
*
*
* (for hydrogen)
*
*
References
Further reading
*
*
*
*
*
External links
Basic Hydrogen Calculations of Quantum Mechanics
at ''The Periodic Table of Videos'' (University of Nottingham)
High temperature hydrogen phase diagram
{{Authority control
Hydrogen,
Chemical elements
Reactive nonmetals
Diatomic nonmetals
Nuclear fusion fuels
Airship technology
Reducing agents
Refrigerants
Gaseous signaling molecules
E-number additives
 Hydrogen has three naturally occurring isotopes, denoted , and . Other, highly unstable nuclei ( to ) have been synthesized in the laboratory but not observed in nature.
is the most common hydrogen isotope, with an abundance of >99.98%. Because the nucleus of this isotope consists of only a single proton, it is given the descriptive but rarely used formal name ''protium''. It is the only stable isotope with no neutrons; see diproton for a discussion of why others do not exist.
, the other stable hydrogen isotope, is known as
Hydrogen has three naturally occurring isotopes, denoted , and . Other, highly unstable nuclei ( to ) have been synthesized in the laboratory but not observed in nature.
is the most common hydrogen isotope, with an abundance of >99.98%. Because the nucleus of this isotope consists of only a single proton, it is given the descriptive but rarely used formal name ''protium''. It is the only stable isotope with no neutrons; see diproton for a discussion of why others do not exist.
, the other stable hydrogen isotope, is known as 
 Hydrogen compounds with hydrogen in the oxidation state −1 are known as hydrides, which are usually formed between hydrogen and metals. The hydrides can be ionic (aka saline), covalent, nor metallic. With heating, H2 reacts efficiently with the alkali and alkaline earth metals to give the Hydride#Ionic_hydrides, ionic hydrides of the formula MH and MH2, respectively. These salt-like crystalline compounds have high melting points and all react with water to liberate hydrogen. Covalent hydrides are include boranes and polymeric aluminium hydride. Transition metals form metal hydrides via continuous dissolution of hydrogen into the metal. A well known hydride is lithium aluminium hydride, the anion carries hydridic centers firmly attached to the Al(III). Perhaps the most extensive series of hydrides are the boranes, compounds consisting only of boron and hydrogen.
Hydrides can bond to these electropositive elements not only as a terminal ligand but also as bridging ligands. In diborane (), four H's are terminal and two bridge between the two B atoms.
Hydrogen compounds with hydrogen in the oxidation state −1 are known as hydrides, which are usually formed between hydrogen and metals. The hydrides can be ionic (aka saline), covalent, nor metallic. With heating, H2 reacts efficiently with the alkali and alkaline earth metals to give the Hydride#Ionic_hydrides, ionic hydrides of the formula MH and MH2, respectively. These salt-like crystalline compounds have high melting points and all react with water to liberate hydrogen. Covalent hydrides are include boranes and polymeric aluminium hydride. Transition metals form metal hydrides via continuous dissolution of hydrogen into the metal. A well known hydride is lithium aluminium hydride, the anion carries hydridic centers firmly attached to the Al(III). Perhaps the most extensive series of hydrides are the boranes, compounds consisting only of boron and hydrogen.
Hydrides can bond to these electropositive elements not only as a terminal ligand but also as bridging ligands. In diborane (), four H's are terminal and two bridge between the two B atoms.
 Hydrogen, as atomic H, is the most Natural abundance, abundant
Hydrogen, as atomic H, is the most Natural abundance, abundant  Electrolysis of water is a conceptually simple method of producing hydrogen.
:
Commercial electrolyzers use nickel-based catalysts in strongly alkaline solution. Platinum is a better catalyst but is expensive. The hydrogen created through electrolysis using renewable energy is commonly referred to as "green hydrogen".
Electrolysis of brine to yield chlorine also produces high purity hydrogen as a co-product, which is used for a variety of transformations such as hydrogenations.
The
Electrolysis of water is a conceptually simple method of producing hydrogen.
:
Commercial electrolyzers use nickel-based catalysts in strongly alkaline solution. Platinum is a better catalyst but is expensive. The hydrogen created through electrolysis using renewable energy is commonly referred to as "green hydrogen".
Electrolysis of brine to yield chlorine also produces high purity hydrogen as a co-product, which is used for a variety of transformations such as hydrogenations.
The 