fluorine on:
[Wikipedia]
[Google]
[Amazon]
Fluorine is a
 The
The
 At room temperature, fluorine is a gas of
At room temperature, fluorine is a gas of
, the Scientific Committee on Problems of the Environment (SCOPE), 1993. See table 1.9 in Section 1.4.5.2. Other radioisotopes have half-lives less than 70 seconds; most decay in less than half a second.. The isotopes and undergo β+ decay and
File:Fluorite-270246.jpg, Fluorite: Pink globular mass with crystal facets
File:Apatite Canada.jpg, Fluorapatite: Long, prismatic crystal, dull in lustre, protruding, at an angle, from
Other minerals such as
 In 1529,
In 1529,
 The
The
 Hydrogen and fluorine combine to yield hydrogen fluoride, in which discrete molecules form clusters by hydrogen bonding, resembling water more than
Hydrogen and fluorine combine to yield hydrogen fluoride, in which discrete molecules form clusters by hydrogen bonding, resembling water more than
 Binary fluorides of metalloids and p-block nonmetals are generally covalent and volatile, with varying reactivities. Period 3 and heavier nonmetals can form hypervalent fluorides..
Binary fluorides of metalloids and p-block nonmetals are generally covalent and volatile, with varying reactivities. Period 3 and heavier nonmetals can form hypervalent fluorides..

 The
The
 Moissan's method is used to produce industrial quantities of fluorine, via the electrolysis of a potassium bifluoride/
Moissan's method is used to produce industrial quantities of fluorine, via the electrolysis of a potassium bifluoride/
Image:The fluorine economy.svg, 675px, center, Clickable diagram of the fluorochemical industry according to mass flows
rect 9 6 81 34
 At least 17,000 metric tons of fluorine are produced each year. It costs only $5–8 per kilogram as uranium or sulfur hexafluoride, but many times more as an element because of handling challenges. Most processes using free fluorine in large amounts employ ''in situ'' generation under
At least 17,000 metric tons of fluorine are produced each year. It costs only $5–8 per kilogram as uranium or sulfur hexafluoride, but many times more as an element because of handling challenges. Most processes using free fluorine in large amounts employ ''in situ'' generation under
 As with other iron alloys, around metspar is added to each metric ton of steel; the fluoride ions lower its melting point and
As with other iron alloys, around metspar is added to each metric ton of steel; the fluoride ions lower its melting point and
 About 180,000 metric tons of fluoropolymers were produced in 2006 and 2007, generating over $3.5 billion revenue per year.. The global market was estimated at just under $6 billion in 2011. Fluoropolymers can only be formed by polymerizing free radicals.
Polytetrafluoroethylene (PTFE), sometimes called by its DuPont name Teflon, represents 60–80% by mass of the world's fluoropolymer production. The largest application is in
About 180,000 metric tons of fluoropolymers were produced in 2006 and 2007, generating over $3.5 billion revenue per year.. The global market was estimated at just under $6 billion in 2011. Fluoropolymers can only be formed by polymerizing free radicals.
Polytetrafluoroethylene (PTFE), sometimes called by its DuPont name Teflon, represents 60–80% by mass of the world's fluoropolymer production. The largest application is in
 Twenty percent of modern pharmaceuticals contain fluorine.. One of these, the cholesterol-reducer
Twenty percent of modern pharmaceuticals contain fluorine.. One of these, the cholesterol-reducer
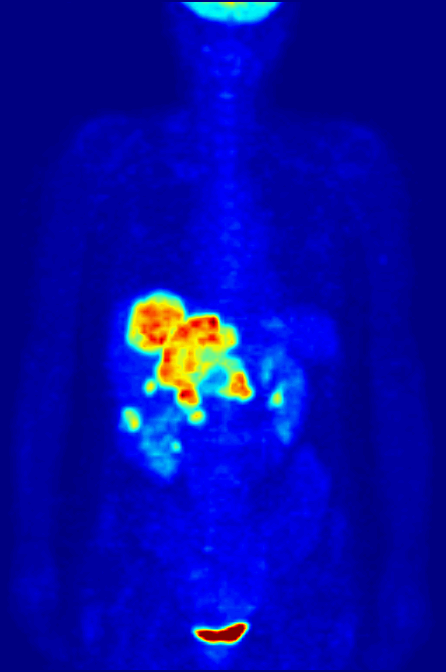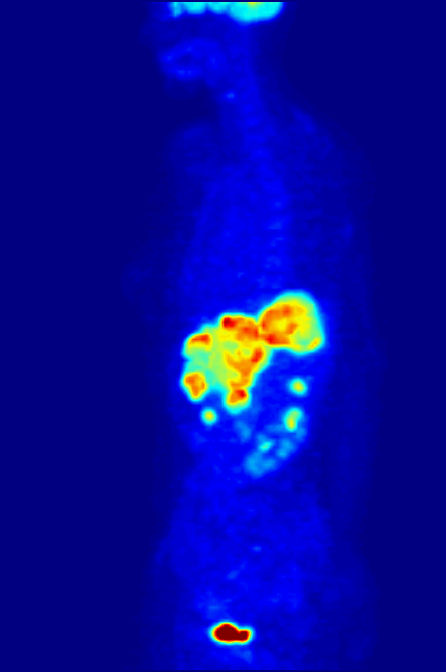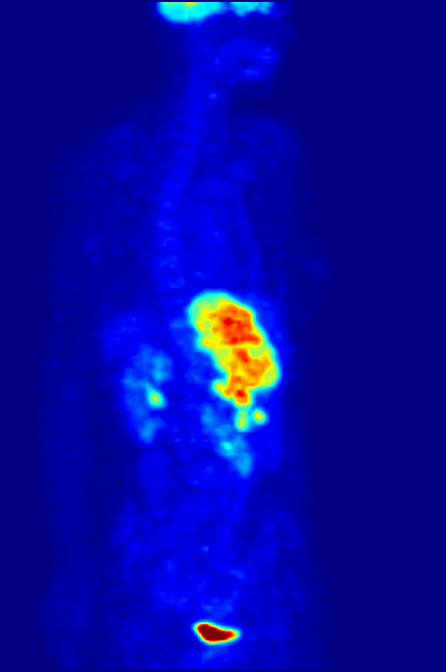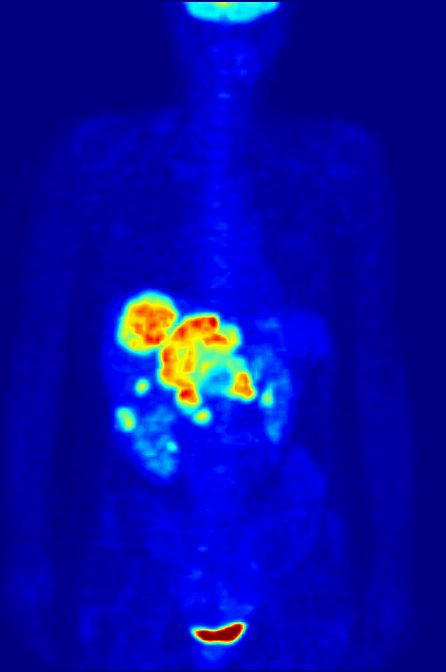 Fluorine-18 is often found in
Fluorine-18 is often found in
 Hydrofluoric acid is the weakest of the hydrohalic acids, having a pKa of 3.2 at 25 °C. Pure hydrogen fluoride is a volatile liquid due to the presence of hydrogen bonding, while the other hydrogen halides are gases. It is able to attack glass, concrete, metals, and organic matter.
Hydrofluoric acid is a contact poison with greater hazards than many strong acids like sulfuric acid even though it is weak: it remains neutral in aqueous solution and thus penetrates tissue faster, whether through inhalation, ingestion or the skin, and at least nine U.S. workers died in such accidents from 1984 to 1994. It reacts with calcium and magnesium in the blood leading to
Hydrofluoric acid is the weakest of the hydrohalic acids, having a pKa of 3.2 at 25 °C. Pure hydrogen fluoride is a volatile liquid due to the presence of hydrogen bonding, while the other hydrogen halides are gases. It is able to attack glass, concrete, metals, and organic matter.
Hydrofluoric acid is a contact poison with greater hazards than many strong acids like sulfuric acid even though it is weak: it remains neutral in aqueous solution and thus penetrates tissue faster, whether through inhalation, ingestion or the skin, and at least nine U.S. workers died in such accidents from 1984 to 1994. It reacts with calcium and magnesium in the blood leading to
 The
The
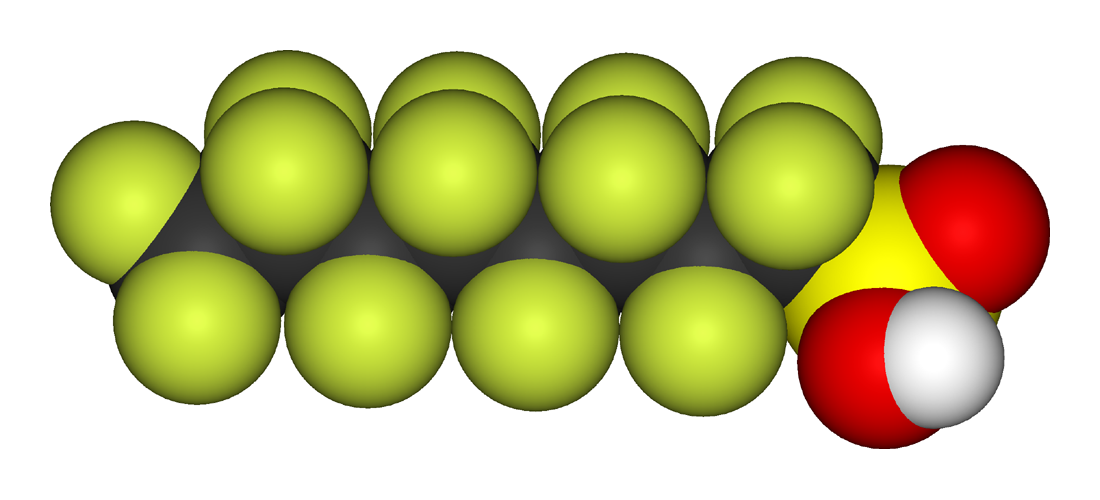 Organofluorines exhibit biopersistence due to the strength of the carbon–fluorine bond. Perfluoroalkyl acids (PFAAs), which are sparingly water-soluble owing to their acidic functional groups, are noted
Organofluorines exhibit biopersistence due to the strength of the carbon–fluorine bond. Perfluoroalkyl acids (PFAAs), which are sparingly water-soluble owing to their acidic functional groups, are noted
chemical element
A chemical element is a chemical substance whose atoms all have the same number of protons. The number of protons is called the atomic number of that element. For example, oxygen has an atomic number of 8: each oxygen atom has 8 protons in its ...
; it has symbol
A symbol is a mark, Sign (semiotics), sign, or word that indicates, signifies, or is understood as representing an idea, physical object, object, or wikt:relationship, relationship. Symbols allow people to go beyond what is known or seen by cr ...
F and atomic number
The atomic number or nuclear charge number (symbol ''Z'') of a chemical element is the charge number of its atomic nucleus. For ordinary nuclei composed of protons and neutrons, this is equal to the proton number (''n''p) or the number of pro ...
9. It is the lightest halogen
The halogens () are a group in the periodic table consisting of six chemically related elements: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and the radioactive elements astatine (At) and tennessine (Ts), though some authors would ...
and exists at standard conditions as pale yellow diatomic
Diatomic molecules () are molecules composed of only two atoms, of the same or different chemical elements. If a diatomic molecule consists of two atoms of the same element, such as hydrogen () or oxygen (), then it is said to be homonuclear mol ...
gas. Fluorine is extremely reactive as it reacts with all other elements except for the light noble gases
The noble gases (historically the inert gases, sometimes referred to as aerogens) are the members of group 18 of the periodic table: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn) and, in some cases, oganesson (Og) ...
. It is highly toxic
Toxicity is the degree to which a chemical substance or a particular mixture of substances can damage an organism. Toxicity can refer to the effect on a whole organism, such as an animal, bacterium, or plant, as well as the effect on a subst ...
.
Among the elements, fluorine ranks 24th in cosmic abundance and 13th in crustal abundance. Fluorite
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon.
The Mohs scal ...
, the primary mineral source of fluorine, which gave the element its name, was first described in 1529; as it was added to metal ore
Ore is natural rock or sediment that contains one or more valuable minerals, typically including metals, concentrated above background levels, and that is economically viable to mine and process. The grade of ore refers to the concentration ...
s to lower their melting points for smelting
Smelting is a process of applying heat and a chemical reducing agent to an ore to extract a desired base metal product. It is a form of extractive metallurgy that is used to obtain many metals such as iron-making, iron, copper extraction, copper ...
, the Latin verb meaning gave the mineral its name. Proposed as an element in 1810, fluorine proved difficult and dangerous to separate from its compounds, and several early experimenters died or sustained injuries from their attempts. Only in 1886 did French chemist Henri Moissan isolate elemental fluorine using low-temperature electrolysis
In chemistry and manufacturing, electrolysis is a technique that uses Direct current, direct electric current (DC) to drive an otherwise non-spontaneous chemical reaction. Electrolysis is commercially important as a stage in the separation of c ...
, a process still employed for modern production. Industrial production of fluorine gas for uranium enrichment
Enriched uranium is a type of uranium in which the percent composition of uranium-235 (written 235U) has been increased through the process of isotope separation. Naturally occurring uranium is composed of three major isotopes: uranium-238 (23 ...
, its largest application, began during the Manhattan Project
The Manhattan Project was a research and development program undertaken during World War II to produce the first nuclear weapons. It was led by the United States in collaboration with the United Kingdom and Canada.
From 1942 to 1946, the ...
in World War II
World War II or the Second World War (1 September 1939 – 2 September 1945) was a World war, global conflict between two coalitions: the Allies of World War II, Allies and the Axis powers. World War II by country, Nearly all of the wo ...
.
Owing to the expense of refining pure fluorine, most commercial applications use fluorine compounds, with about half of mined fluorite used in steelmaking
Steelmaking is the process of producing steel from iron ore and/or scrap. Steel has been made for millennia, and was commercialized on a massive scale in the 1850s and 1860s, using the Bessemer process, Bessemer and open hearth furnace, Siemens-M ...
. The rest of the fluorite is converted into hydrogen fluoride
Hydrogen fluoride (fluorane) is an Inorganic chemistry, inorganic compound with chemical formula . It is a very poisonous, colorless gas or liquid that dissolves in water to yield hydrofluoric acid. It is the principal industrial source of fluori ...
en route to various organic fluorides, or into cryolite
Cryolite ( Na3 Al F6, sodium hexafluoroaluminate) is a rare mineral identified with the once-large deposit at Ivittuut on the west coast of Greenland, mined commercially until 1987.
It is used in the reduction ("smelting") of aluminium, in pest ...
, which plays a key role in aluminium refining. The carbon–fluorine bond
The carbon–fluorine bond is a polar covalent bond between carbon and fluorine that is a component of all organofluorine compounds. It is one of the strongest single bonds in chemistry (after the B–F single bond, Si–F single bond, and H–F ...
is usually very stable. Organofluorine compounds are widely used as refrigerant
A refrigerant is a working fluid used in the cooling, heating, or reverse cooling/heating cycles of air conditioning systems and heat pumps, where they undergo a repeated phase transition from a liquid to a gas and back again. Refrigerants are ...
s, electrical insulation, and PTFE
Polytetrafluoroethylene (PTFE) is a synthetic fluoropolymer of tetrafluoroethylene, and has numerous applications because it is chemically inert. The commonly known brand name of PTFE-based composition is Teflon by Chemours, a spin-off fro ...
(Teflon). Pharmaceuticals such as atorvastatin
Atorvastatin, sold under the brand name Lipitor among others, is a statin medication used to prevent cardiovascular disease in those at high risk and to treat abnormal lipid levels. For the prevention of cardiovascular disease, statins are a ...
and fluoxetine
Fluoxetine, sold under the brand name Prozac, among others, is an Antidepressant, antidepressant medication of the selective serotonin reuptake inhibitor (SSRI) class used for the treatment of major depressive disorder, Anxiety disorder, anx ...
contain C−F bonds. The fluoride ion
Fluoride (). According to this source, is a possible pronunciation in British English. is an inorganic, monatomic anion of fluorine, with the chemical formula (also written ), whose salts are typically white or colorless. Fluoride salts typi ...
from dissolved fluoride salts inhibits dental cavities and so finds use in toothpaste
Toothpaste is a paste or gel dentifrice that is used with a toothbrush to clean and maintain the aesthetics of Human tooth, teeth. Toothpaste is used to promote oral hygiene: it is an abrasive that aids in removing dental plaque and food from th ...
and water fluoridation
Water fluoridation is the controlled addition of fluoride to Public water supply, public water supplies to reduce tooth decay. Fluoridated water maintains fluoride levels effective for cavity prevention, achieved naturally or through supplem ...
. Global fluorochemical sales amount to more than US$
The United States dollar (Currency symbol, symbol: Dollar sign, $; ISO 4217, currency code: USD) is the official currency of the United States and International use of the U.S. dollar, several other countries. The Coinage Act of 1792 introdu ...
15 billion a year.
Fluorocarbon
Fluorocarbons are chemical compounds with carbon-fluorine bonds. Compounds that contain many C-F bonds often have distinctive properties, e.g., enhanced stability, volatility, and hydrophobicity. Several fluorocarbons and their derivatives are ...
gases are generally greenhouse gas
Greenhouse gases (GHGs) are the gases in the atmosphere that raise the surface temperature of planets such as the Earth. Unlike other gases, greenhouse gases absorb the radiations that a planet emits, resulting in the greenhouse effect. T ...
es with global-warming potentials 100 to 23,500 times that of carbon dioxide
Carbon dioxide is a chemical compound with the chemical formula . It is made up of molecules that each have one carbon atom covalent bond, covalently double bonded to two oxygen atoms. It is found in a gas state at room temperature and at norma ...
, and SF6 has the highest global warming potential of any known substance. Organofluorine compounds often persist in the environment due to the strength of the carbon–fluorine bond. Fluorine has no known metabolic role in mammals; a few plant
Plants are the eukaryotes that form the Kingdom (biology), kingdom Plantae; they are predominantly Photosynthesis, photosynthetic. This means that they obtain their energy from sunlight, using chloroplasts derived from endosymbiosis with c ...
s and marine sponges synthesize organofluorine poisons (most often monofluoroacetates) that help deter predation.
Characteristics
Electron configuration
Fluorine atoms have nine electrons, one fewer thanneon
Neon is a chemical element; it has symbol Ne and atomic number 10. It is the second noble gas in the periodic table. Neon is a colorless, odorless, inert monatomic gas under standard conditions, with approximately two-thirds the density of ...
, and electron configuration
In atomic physics and quantum chemistry, the electron configuration is the distribution of electrons of an atom or molecule (or other physical structure) in atomic or molecular orbitals. For example, the electron configuration of the neon ato ...
1s22s22p5: two electrons in a filled inner shell and seven in an outer shell requiring one more to be filled. The outer electrons are ineffective at nuclear shielding, and experience a high effective nuclear charge
In atomic physics, the effective nuclear charge of an electron in a multi-electron atom or ion is the number of elementary charges (e) an electron experiences by the nucleus. It is denoted by ''Z''eff. The term "effective" is used because the shi ...
of 9 − 2 = 7; this affects the atom's physical properties.
Fluorine's first ionization energy is third-highest among all elements, behind helium and neon, which complicates the removal of electrons from neutral fluorine atoms. It also has a high electron affinity
The electron affinity (''E''ea) of an atom or molecule is defined as the amount of energy released when an electron attaches to a neutral atom or molecule in the gaseous state to form an anion.
::X(g) + e− → X−(g) + energy
This differs by si ...
, second only to chlorine
Chlorine is a chemical element; it has Symbol (chemistry), symbol Cl and atomic number 17. The second-lightest of the halogens, it appears between fluorine and bromine in the periodic table and its properties are mostly intermediate between ...
, and tends to capture an electron to become isoelectronic
Isoelectronicity is a phenomenon observed when two or more molecules have the same structure (positions and connectivities among atoms) and the same electronic configurations, but differ by what specific elements are at certain locations in th ...
with the noble gas neon; it has the highest electronegativity
Electronegativity, symbolized as , is the tendency for an atom of a given chemical element to attract shared electrons (or electron density) when forming a chemical bond. An atom's electronegativity is affected by both its atomic number and the ...
of any reactive element. Fluorine atoms have a small covalent radius
The covalent radius, ''r''cov, is a measure of the size of an atom that forms part of one covalent bond. It is usually measured either in picometres (pm) or angstroms (Å), with 1 Å = 100 pm.
In principle, the sum of the two cova ...
of around 60 picometer
The picometre (international spelling as used by the International Bureau of Weights and Measures; SI symbol: pm) or picometer ( American spelling) is a unit of length in the International System of Units (SI), equal to , or one trillionth o ...
s, similar to those of its period neighbors oxygen and neon..
Reactivity
bond energy
In chemistry, bond energy (''BE'') is one measure of the strength of a chemical bond. It is sometimes called the mean bond, bond enthalpy, average bond enthalpy, or bond strength. IUPAC defines bond energy as the average value of the gas-phase b ...
of difluorine is much lower than that of either or and similar to the easily cleaved peroxide
In chemistry, peroxides are a group of Chemical compound, compounds with the structure , where the R's represent a radical (a portion of a complete molecule; not necessarily a free radical) and O's are single oxygen atoms. Oxygen atoms are joined ...
bond; this, along with high electronegativity, accounts for fluorine's easy dissociation, high reactivity, and strong bonds to non-fluorine atoms. Conversely, bonds to other atoms are very strong because of fluorine's high electronegativity. Unreactive substances like powdered steel, glass fragments, and asbestos
Asbestos ( ) is a group of naturally occurring, Toxicity, toxic, carcinogenic and fibrous silicate minerals. There are six types, all of which are composed of long and thin fibrous Crystal habit, crystals, each fibre (particulate with length su ...
fibers react quickly with cold fluorine gas; wood and water spontaneously combust under a fluorine jet..
Reactions of elemental fluorine with metals require varying conditions. Alkali metal
The alkali metals consist of the chemical elements lithium (Li), sodium (Na), potassium (K),The symbols Na and K for sodium and potassium are derived from their Latin names, ''natrium'' and ''kalium''; these are still the origins of the names ...
s cause explosions and alkaline earth metal
The alkaline earth metals are six chemical elements in group (periodic table), group 2 of the periodic table. They are beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra).. The elements have very similar p ...
s display vigorous activity in bulk; to prevent passivation from the formation of metal fluoride layers, most other metals such as aluminium and iron must be powdered, and noble metal
A noble metal is ordinarily regarded as a metallic chemical element, element that is generally resistant to corrosion and is usually found in nature in its native element, raw form. Gold, platinum, and the other platinum group metals (ruthenium ...
s require pure fluorine gas at . Some solid nonmetals (sulfur, phosphorus) react vigorously in liquid fluorine. Hydrogen sulfide
Hydrogen sulfide is a chemical compound with the formula . It is a colorless chalcogen-hydride gas, and is toxic, corrosive, and flammable. Trace amounts in ambient atmosphere have a characteristic foul odor of rotten eggs. Swedish chemist ...
and sulfur dioxide
Sulfur dioxide (IUPAC-recommended spelling) or sulphur dioxide (traditional Commonwealth English) is the chemical compound with the formula . It is a colorless gas with a pungent smell that is responsible for the odor of burnt matches. It is r ...
combine readily with fluorine, the latter sometimes explosively; sulfuric acid
Sulfuric acid (American spelling and the preferred IUPAC name) or sulphuric acid (English in the Commonwealth of Nations, Commonwealth spelling), known in antiquity as oil of vitriol, is a mineral acid composed of the elements sulfur, oxygen, ...
exhibits much less activity, requiring elevated temperatures.
Hydrogen
Hydrogen is a chemical element; it has chemical symbol, symbol H and atomic number 1. It is the lightest and abundance of the chemical elements, most abundant chemical element in the universe, constituting about 75% of all baryon, normal matter ...
, like some of the alkali metals, reacts explosively with fluorine. Carbon
Carbon () is a chemical element; it has chemical symbol, symbol C and atomic number 6. It is nonmetallic and tetravalence, tetravalent—meaning that its atoms are able to form up to four covalent bonds due to its valence shell exhibiting 4 ...
, as lamp black
Carbon black (with subtypes acetylene black, channel black, furnace black, lamp black and thermal black) is a material produced by the incomplete combustion of coal tar, vegetable matter, or petroleum products, including fuel oil, fluid catalyti ...
, reacts at room temperature to yield tetrafluoromethane. Graphite combines with fluorine above to produce non-stoichiometric
Non-stoichiometric compounds are chemical compounds, almost always solid inorganic compounds, having chemical element, elemental composition whose proportions cannot be represented by a ratio of small natural numbers (i.e. an empirical formula); ...
carbon monofluoride; higher temperatures generate gaseous fluorocarbon
Fluorocarbons are chemical compounds with carbon-fluorine bonds. Compounds that contain many C-F bonds often have distinctive properties, e.g., enhanced stability, volatility, and hydrophobicity. Several fluorocarbons and their derivatives are ...
s, sometimes with explosions.. Carbon dioxide and carbon monoxide react at or just above room temperature,. whereas paraffins and other organic chemicals generate strong reactions: even completely substituted haloalkane
The haloalkanes (also known as halogenoalkanes or alkyl halides) are alkanes containing one or more halogen substituents of hydrogen atom. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalka ...
s such as carbon tetrachloride
Carbon tetrachloride, also known by many other names (such as carbon tet for short and tetrachloromethane, also IUPAC nomenclature of inorganic chemistry, recognised by the IUPAC), is a chemical compound with the chemical formula CCl4. It is a n ...
, normally incombustible, may explode.. Although nitrogen trifluoride
Nitrogen trifluoride is the inorganic compound with the formula (). It is a colorless, non-flammable, toxic gas with a slightly musty odor. In contrast with ammonia, it is nonbasic. It finds increasing use within the manufacturing of flat-panel ...
is stable, nitrogen requires an electric discharge
In electromagnetism, an electric discharge is the release and transmission of electricity in an applied electric field through a medium such as a gas (i.e., an outgoing flow of electric current through a non-metal medium).American Geophysical U ...
at elevated temperatures for reaction with fluorine to occur, due to the very strong triple bond
A triple bond in chemistry is a chemical bond between two atoms involving six Electron pair bond, bonding electrons instead of the usual two in a covalent bond, covalent single bond. Triple bonds are stronger than the equivalent covalent bond, sin ...
in elemental nitrogen; ammonia may react explosively... Oxygen
Oxygen is a chemical element; it has chemical symbol, symbol O and atomic number 8. It is a member of the chalcogen group (periodic table), group in the periodic table, a highly reactivity (chemistry), reactive nonmetal (chemistry), non ...
does not combine with fluorine under ambient conditions, but can be made to react using electric discharge at low temperatures and pressures; the products tend to disintegrate into their constituent elements when heated.. Heavier halogens react readily with fluorine as does the noble gas radon
Radon is a chemical element; it has symbol Rn and atomic number 86. It is a radioactive noble gas and is colorless and odorless. Of the three naturally occurring radon isotopes, only Rn has a sufficiently long half-life (3.825 days) for it to b ...
; of the other noble gases, only xenon
Xenon is a chemical element; it has symbol Xe and atomic number 54. It is a dense, colorless, odorless noble gas found in Earth's atmosphere in trace amounts. Although generally unreactive, it can undergo a few chemical reactions such as the ...
and krypton
Krypton (from 'the hidden one') is a chemical element; it has symbol (chemistry), symbol Kr and atomic number 36. It is a colorless, odorless noble gas that occurs in trace element, trace amounts in the Earth's atmosphere, atmosphere and is of ...
react, and only under special conditions.. Argon
Argon is a chemical element; it has symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third most abundant gas in Earth's atmosphere, at 0.934% (9340 ppmv). It is more than twice as abu ...
does not react with fluorine gas; however, it does form a compound with fluorine, argon fluorohydride.
Phases
diatomic molecule
Diatomic molecules () are molecules composed of only two atoms, of the same or different chemical elements. If a diatomic molecule consists of two atoms of the same element, such as hydrogen () or oxygen (), then it is said to be homonuclear mol ...
s, pale yellow when pure (sometimes described as yellow-green). It has a characteristic halogen-like pungent and biting odor detectable at 20 ppb. Fluorine condenses into a bright yellow liquid at , a transition temperature similar to those of oxygen and nitrogen.
Fluorine has two solid forms, α- and β-fluorine. The latter crystallizes at and is transparent and soft, with the same disordered cubic
Cubic may refer to:
Science and mathematics
* Cube (algebra), "cubic" measurement
* Cube, a three-dimensional solid object bounded by six square faces, facets or sides, with three meeting at each vertex
** Cubic crystal system, a crystal system w ...
structure of freshly crystallized solid oxygen, unlike the orthorhombic
In crystallography, the orthorhombic crystal system is one of the 7 crystal systems. Orthorhombic Lattice (group), lattices result from stretching a cubic crystal system, cubic lattice along two of its orthogonal pairs by two different factors, res ...
systems of other solid halogens. Further cooling to induces a phase transition
In physics, chemistry, and other related fields like biology, a phase transition (or phase change) is the physical process of transition between one state of a medium and another. Commonly the term is used to refer to changes among the basic Sta ...
into opaque and hard α-fluorine, which has a monoclinic
In crystallography, the monoclinic crystal system is one of the seven crystal systems. A crystal system is described by three Vector (geometric), vectors. In the monoclinic system, the crystal is described by vectors of unequal lengths, as in t ...
structure with dense, angled layers of molecules. The transition from β- to α-fluorine is more exothermic
In thermodynamics, an exothermic process () is a thermodynamic process or reaction that releases energy from the system to its surroundings, usually in the form of heat, but also in a form of light (e.g. a spark, flame, or flash), electricity (e ...
than the condensation of fluorine, and can be violent..
Isotopes
Only oneisotope
Isotopes are distinct nuclear species (or ''nuclides'') of the same chemical element. They have the same atomic number (number of protons in their Atomic nucleus, nuclei) and position in the periodic table (and hence belong to the same chemica ...
of fluorine occurs naturally in abundance, the stable isotope . It has a high magnetogyric ratio and exceptional sensitivity to magnetic fields; because it is also the only stable isotope, it is used in magnetic resonance imaging
Magnetic resonance imaging (MRI) is a medical imaging technique used in radiology to generate pictures of the anatomy and the physiological processes inside the body. MRI scanners use strong magnetic fields, magnetic field gradients, and ...
. Eighteen radioisotope
A radionuclide (radioactive nuclide, radioisotope or radioactive isotope) is a nuclide that has excess numbers of either neutrons or protons, giving it excess nuclear energy, and making it unstable. This excess energy can be used in one of three ...
s with mass number
The mass number (symbol ''A'', from the German word: ''Atomgewicht'', "atomic weight"), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. It is appro ...
s 13–31 have been synthesized, of which is the most stable with a half-life Half-life is a mathematical and scientific description of exponential or gradual decay.
Half-life, half life or halflife may also refer to:
Film
* Half-Life (film), ''Half-Life'' (film), a 2008 independent film by Jennifer Phang
* ''Half Life: ...
of 109.734 minutes. is a natural trace radioisotope
A trace radioisotope is a radioisotope that occurs naturally in trace amounts (i.e. extremely small). Generally speaking, trace radioisotopes have half-lives that are short in comparison with the age of the Earth, since primordial nuclides tend to ...
produced by cosmic ray spallation of atmospheric argon
Argon is a chemical element; it has symbol Ar and atomic number 18. It is in group 18 of the periodic table and is a noble gas. Argon is the third most abundant gas in Earth's atmosphere, at 0.934% (9340 ppmv). It is more than twice as abu ...
as well as by reaction of protons with natural oxygen: 18O + p → 18F + n.SCOPE 50 - Radioecology after Chernobyl, the Scientific Committee on Problems of the Environment (SCOPE), 1993. See table 1.9 in Section 1.4.5.2. Other radioisotopes have half-lives less than 70 seconds; most decay in less than half a second.. The isotopes and undergo β+ decay and
electron capture
Electron capture (K-electron capture, also K-capture, or L-electron capture, L-capture) is a process in which the proton-rich nucleus of an electrically neutral atom absorbs an inner atomic electron, usually from the K or L electron shells. Th ...
, lighter isotopes decay by proton emission
Proton emission (also known as proton radioactivity) is a rare type of radioactive decay in which a proton is ejected from a atomic nucleus, nucleus. Proton emission can occur from high-lying excited states in a nucleus following a beta decay ...
, and those heavier than undergo β− decay (the heaviest ones with delayed neutron emission
Neutron emission is a mode of radioactive decay in which one or more neutrons are ejected from a Atomic nucleus, nucleus. It occurs in the most neutron-rich/proton-deficient nuclides, and also from excited states of other nuclides as in photodisin ...
). Two metastable isomers of fluorine are known, , with a half-life of 162(7) nanoseconds, and , with a half-life of 2.2(1) milliseconds.
Occurrence
Universe
Among the lighter elements, fluorine's abundance value of 400 ppb (parts per billion) – 24th among elements in the universe – is exceptionally low: other elements from carbon to magnesium are twenty or more times as common. This is becausestellar nucleosynthesis
In astrophysics, stellar nucleosynthesis is the creation of chemical elements by nuclear fusion reactions within stars. Stellar nucleosynthesis has occurred since the original creation of hydrogen, helium and lithium during the Big Bang. As a ...
processes bypass fluorine, and any fluorine atoms otherwise created have high nuclear cross section
The nuclear cross section of a nucleus is used to describe the probability that a nuclear reaction will occur. The concept of a nuclear cross section can be quantified physically in terms of "characteristic area" where a larger area means a larg ...
s, allowing collisions with hydrogen or helium to generate oxygen or neon respectively.
Beyond this transient existence, three explanations have been proposed for the presence of fluorine:..
* during type II supernova
A Type II supernova or SNII (plural: ''supernovae'') results from the rapid collapse and violent explosion of a massive star. A star must have at least eight times, but no more than 40 to 50 times, the mass of the Sun () to undergo this type ...
e, bombardment of neon atoms by neutrino
A neutrino ( ; denoted by the Greek letter ) is an elementary particle that interacts via the weak interaction and gravity. The neutrino is so named because it is electrically neutral and because its rest mass is so small ('' -ino'') that i ...
s could transmute them to fluorine;
* the solar wind of Wolf–Rayet star
Wolf–Rayet stars, often abbreviated as WR stars, are a rare heterogeneous set of stars with unusual spectroscopy, spectra showing prominent broad emission lines of ionised helium and highly ionised nitrogen or carbon. The spectra indicate very ...
s could blow fluorine away from any hydrogen or helium atoms; or
* fluorine is borne out on convection currents arising from fusion in asymptotic giant branch
The asymptotic giant branch (AGB) is a region of the Hertzsprung–Russell diagram populated by evolved cool luminous stars. This is a period of stellar evolution undertaken by all low- to intermediate-mass stars (about 0.5 to 8 solar masses) lat ...
stars.
Earth
Fluorine is the 13th most abundant element in Earth's crust at 600–700 ppm (parts per million) by mass. Though believed not to occur naturally, elemental fluorine has been shown to be present as an occlusion in antozonite, a variant of fluorite. Most fluorine exists as fluoride-containing minerals.Fluorite
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon.
The Mohs scal ...
, fluorapatite
Fluorapatite, often with the alternate spelling of fluoroapatite, is a phosphate mineral with the formula Ca5(PO4)3F (calcium fluorophosphate). Fluorapatite is a hard crystalline solid. Although samples can have various color (green, brown, blu ...
and cryolite
Cryolite ( Na3 Al F6, sodium hexafluoroaluminate) is a rare mineral identified with the once-large deposit at Ivittuut on the west coast of Greenland, mined commercially until 1987.
It is used in the reduction ("smelting") of aluminium, in pest ...
are the most industrially significant. Fluorite (), also known as fluorspar, abundant worldwide, is the main source of fluoride, and hence fluorine. China and Mexico are the major suppliers.... Fluorapatite (Ca5(PO4)3F), which contains most of the world's fluoride, is an inadvertent source of fluoride as a byproduct of fertilizer production. Cryolite (), used in the production of aluminium, is the most fluorine-rich mineral. Economically viable natural sources of cryolite have been exhausted, and most is now synthesised commercially.
matrix
Matrix (: matrices or matrixes) or MATRIX may refer to:
Science and mathematics
* Matrix (mathematics), a rectangular array of numbers, symbols or expressions
* Matrix (logic), part of a formula in prenex normal form
* Matrix (biology), the m ...
of aggregate-like rock
File:Ivigtut cryolite edit.jpg, Cryolite: A parallelogram-shaped outline with diatomic molecules arranged in two layers
topaz
Topaz is a silicate mineral made of aluminium, aluminum and fluorine with the chemical formula aluminium, Alsilicon, Sioxygen, O(fluorine, F, hydroxide, OH). It is used as a gemstone in jewelry and other adornments. Common topaz in its natural ...
contain fluorine. Fluorides, unlike other halides, are insoluble and do not occur in commercially favorable concentrations in saline waters. Trace quantities of organofluorines of uncertain origin have been detected in volcanic eruptions and geothermal springs. The existence of gaseous fluorine in crystals, suggested by the smell of crushed antozonite, is contentious; a 2012 study reported the presence of 0.04% by weight in antozonite, attributing these inclusions to radiation from the presence of tiny amounts of uranium
Uranium is a chemical element; it has chemical symbol, symbol U and atomic number 92. It is a silvery-grey metal in the actinide series of the periodic table. A uranium atom has 92 protons and 92 electrons, of which 6 are valence electrons. Ura ...
.
History
Early discoveries
 In 1529,
In 1529, Georgius Agricola
Georgius Agricola (; born Georg Bauer; 24 March 1494 – 21 November 1555) was a German Humanist scholar, mineralogist and metallurgist. Born in the small town of Glauchau, in the Electorate of Saxony of the Holy Roman Empire, he was b ...
described fluorite as an additive used to lower the melting point of metals during smelting
Smelting is a process of applying heat and a chemical reducing agent to an ore to extract a desired base metal product. It is a form of extractive metallurgy that is used to obtain many metals such as iron-making, iron, copper extraction, copper ...
.. He penned the Latin word ''fluorēs'' (''fluor,'' flow) for fluorite rocks. The name later evolved into ''fluorspar'' (still commonly used) and then ''fluorite''.. The composition of fluorite was later determined to be calcium difluoride..
Hydrofluoric acid
Hydrofluoric acid is a solution of hydrogen fluoride (HF) in water. Solutions of HF are colorless, acidic and highly corrosive. A common concentration is 49% (48–52%) but there are also stronger solutions (e.g. 70%) and pure HF has a boiling p ...
was used in glass etching from 1720 onward. Andreas Sigismund Marggraf first characterized it in 1764 when he heated fluorite with sulfuric acid, and the resulting solution corroded its glass container. Swedish chemist Carl Wilhelm Scheele
Carl Wilhelm Scheele (, ; 9 December 1742 – 21 May 1786) was a Swedish Pomerania, German-Swedish pharmaceutical chemist.
Scheele discovered oxygen (although Joseph Priestley published his findings first), and identified the elements molybd ...
repeated the experiment in 1771, and named the acidic product ''fluss-spats-syran'' (fluorspar acid). In 1810, the French physicist André-Marie Ampère
André-Marie Ampère (, ; ; 20 January 177510 June 1836) was a French physicist and mathematician who was one of the founders of the science of classical electromagnetism, which he referred to as ''electrodynamics''. He is also the inventor of ...
suggested that hydrogen and an element analogous to chlorine constituted hydrofluoric acid. He also proposed in a letter to Sir Humphry Davy
Sir Humphry Davy, 1st Baronet (17 December 177829 May 1829) was a British chemist and inventor who invented the Davy lamp and a very early form of arc lamp. He is also remembered for isolating, by using electricity, several Chemical element, e ...
dated August 26, 1812 that this then-unknown substance may be named ''fluorine'' from fluoric compounds and the ''-ine'' suffix of other halogens. This word, often with modifications, is used in most European languages; however, Greek, Russian, and some others, following Ampère's later suggestion, use the name ''ftor'' or derivatives, from the Greek φθόριος (''phthorios'', destructive). The New Latin name ''fluorum'' gave the element its current symbol F; Fl was used in early papers.
Isolation
upleft, 1887 drawing of Moissan's apparatus Initial studies on fluorine were so dangerous that several 19th-century experimenters were deemed "fluorine martyrs" after misfortunes with hydrofluoric acid. Isolation of elemental fluorine was hindered by the extreme corrosiveness of both elemental fluorine itself and hydrogen fluoride, as well as the lack of a simple and suitableelectrolyte
An electrolyte is a substance that conducts electricity through the movement of ions, but not through the movement of electrons. This includes most soluble Salt (chemistry), salts, acids, and Base (chemistry), bases, dissolved in a polar solven ...
. Edmond Frémy postulated that electrolysis
In chemistry and manufacturing, electrolysis is a technique that uses Direct current, direct electric current (DC) to drive an otherwise non-spontaneous chemical reaction. Electrolysis is commercially important as a stage in the separation of c ...
of pure hydrogen fluoride to generate fluorine was feasible and devised a method to produce anhydrous samples from acidified potassium bifluoride; instead, he discovered that the resulting (dry) hydrogen fluoride did not conduct electricity.. Frémy's former student Henri Moissan persevered, and after much trial and error found that a mixture of potassium bifluoride and dry hydrogen fluoride was a conductor, enabling electrolysis. To prevent rapid corrosion of the platinum in his electrochemical cell
An electrochemical cell is a device that either generates electrical energy from chemical reactions in a so called galvanic cell, galvanic or voltaic cell, or induces chemical reactions (electrolysis) by applying external electrical energy in an ...
s, he cooled the reaction to extremely low temperatures in a special bath and forged cells from a more resistant mixture of platinum and iridium
Iridium is a chemical element; it has the symbol Ir and atomic number 77. This very hard, brittle, silvery-white transition metal of the platinum group, is considered the second-densest naturally occurring metal (after osmium) with a density ...
, and used fluorite stoppers. In 1886, after 74 years of effort by many chemists, Moissan isolated elemental fluorine..
In 1906, two months before his death, Moissan received the Nobel Prize in Chemistry
The Nobel Prize in Chemistry () is awarded annually by the Royal Swedish Academy of Sciences to scientists in the various fields of chemistry. It is one of the five Nobel Prizes established by the will of Alfred Nobel in 1895, awarded for outst ...
,. with the following citation:.
Later uses
 The
The Frigidaire
Frigidaire Appliance Company is the American consumer and Commercial area, commercial home appliances brand subsidiary of multinational company Electrolux, a Swedish multinational home appliance manufacturer, headquartered in Stockholm.
History ...
division of General Motors
General Motors Company (GM) is an American Multinational corporation, multinational Automotive industry, automotive manufacturing company headquartered in Detroit, Michigan, United States. The company is most known for owning and manufacturing f ...
(GM) experimented with chlorofluorocarbon refrigerants in the late 1920s, and Kinetic Chemicals was formed as a joint venture between GM and DuPont
Dupont, DuPont, Du Pont, duPont, or du Pont may refer to:
People
* Dupont (surname) Dupont, also spelled as DuPont, duPont, Du Pont, or du Pont is a French surname meaning "of the bridge", historically indicating that the holder of the surname re ...
in 1930 hoping to market Freon-12 () as one such refrigerant
A refrigerant is a working fluid used in the cooling, heating, or reverse cooling/heating cycles of air conditioning systems and heat pumps, where they undergo a repeated phase transition from a liquid to a gas and back again. Refrigerants are ...
. It replaced earlier and more toxic compounds, increased demand for kitchen refrigerators, and became profitable; by 1949 DuPont had bought out Kinetic and marketed several other Freon
Freon ( ) is a registered trademark of the Chemours Company and generic descriptor for a number of halocarbon products. They are stable, nonflammable, low toxicity gases or liquids which have generally been used as refrigerants and as aerosol p ...
compounds... Polytetrafluoroethylene
Polytetrafluoroethylene (PTFE) is a synthetic fluoropolymer of tetrafluoroethylene, and has numerous applications because it is chemically inert. The commonly known brand name of PTFE-based composition is Teflon by Chemours, a corporate spin-of ...
(Teflon) was serendipitously discovered in 1938 by Roy J. Plunkett while working on refrigerants at Kinetic, and its superlative chemical and thermal resistance lent it to accelerated commercialization and mass production by 1941.
Large-scale production of elemental fluorine began during World War II. Germany used high-temperature electrolysis to make tons of the planned incendiary chlorine trifluoride and the Manhattan Project
The Manhattan Project was a research and development program undertaken during World War II to produce the first nuclear weapons. It was led by the United States in collaboration with the United Kingdom and Canada.
From 1942 to 1946, the ...
used huge quantities to produce uranium hexafluoride for uranium enrichment. Since is as corrosive as fluorine, gaseous diffusion
Gaseous diffusion is a technology that was used to produce enriched uranium by forcing gaseous uranium hexafluoride (UF6) through microporous membranes. This produces a slight separation (enrichment factor 1.0043) between the molecules containi ...
plants required special materials: nickel for membranes, fluoropolymers for seals, and liquid fluorocarbons as coolants and lubricants. This burgeoning nuclear industry later drove post-war fluorochemical development.
Compounds
Fluorine has a rich chemistry, encompassing organic and inorganic domains. It combines with metals, nonmetals,metalloid
A metalloid is a chemical element which has a preponderance of material property, properties in between, or that are a mixture of, those of metals and Nonmetal (chemistry), nonmetals. The word metalloid comes from the Latin language, Latin ''meta ...
s, and most noble gases.. Fluorine's high electron affinity results in a preference for ionic bonding
Ionic bonding is a type of chemical bonding that involves the Coulomb's law, electrostatic attraction between oppositely charged ions, or between two atoms with sharply different electronegativities, and is the primary interaction occurring in io ...
; when it forms covalent bond
A covalent bond is a chemical bond that involves the sharing of electrons to form electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs. The stable balance of attractive and repulsive forces between atom ...
s, these are polar, and almost always single..The metastable boron
Boron is a chemical element; it has symbol B and atomic number 5. In its crystalline form it is a brittle, dark, lustrous metalloid; in its amorphous form it is a brown powder. As the lightest element of the boron group it has three ...
and nitrogen monofluoride have higher-order fluorine bonds, and some metal complex
A coordination complex is a chemical compound consisting of a central atom or ion, which is usually metallic and is called the ''coordination centre'', and a surrounding array of bound molecules or ions, that are in turn known as ''ligands'' or ...
es use it as a bridging ligand
In coordination chemistry, a bridging ligand is a ligand that connects two or more atoms, usually metal ions. The ligand may be atomic or polyatomic. Virtually all complex organic compounds can serve as bridging ligands, so the term is usually r ...
. Hydrogen bonding
In chemistry, a hydrogen bond (H-bond) is a specific type of molecular interaction that exhibits partial covalent character and cannot be described as a purely electrostatic force. It occurs when a hydrogen (H) atom, Covalent bond, covalently b ...
is another possibility.
Oxidation states
In compounds, fluorine almost exclusively assumes anoxidation state
In chemistry, the oxidation state, or oxidation number, is the hypothetical Electrical charge, charge of an atom if all of its Chemical bond, bonds to other atoms are fully Ionic bond, ionic. It describes the degree of oxidation (loss of electrons ...
of −1. Fluorine in is defined to have oxidation state 0. The unstable species and , which decompose at around 40 K, have intermediate oxidation states; and a few related species are predicted to be stable.
Metals
Alkali metals form ionic and highly soluble monofluorides; these have the cubic arrangement of sodium chloride and analogous chlorides. Alkaline earth difluorides possess strong ionic bonds but are insoluble in water,. with the exception of beryllium difluoride, which also exhibits some covalent character and has aquartz
Quartz is a hard, crystalline mineral composed of silica (silicon dioxide). The Atom, atoms are linked in a continuous framework of SiO4 silicon–oxygen Tetrahedral molecular geometry, tetrahedra, with each oxygen being shared between two tet ...
-like structure.. Rare earth element
The rare-earth elements (REE), also called the rare-earth metals or rare earths, and sometimes the lanthanides or lanthanoids (although scandium and yttrium, which do not belong to this series, are usually included as rare earths), are a set o ...
s and many other metals form mostly ionic trifluorides..
Covalent bonding first comes to prominence in the tetrafluorides: those of zirconium
Zirconium is a chemical element; it has Symbol (chemistry), symbol Zr and atomic number 40. First identified in 1789, isolated in impure form in 1824, and manufactured at scale by 1925, pure zirconium is a lustrous transition metal with a greyis ...
, hafnium
Hafnium is a chemical element; it has symbol Hf and atomic number 72. A lustrous, silvery gray, tetravalent transition metal, hafnium chemically resembles zirconium and is found in many zirconium minerals. Its existence was predicted by Dm ...
. and several actinides
The actinide () or actinoid () series encompasses at least the 14 metallic chemical elements in the 5f series, with atomic numbers from 89 to 102, actinium through nobelium. Number 103, lawrencium, is also generally included despite being part ...
are ionic with high melting points, while those of titanium
Titanium is a chemical element; it has symbol Ti and atomic number 22. Found in nature only as an oxide, it can be reduced to produce a lustrous transition metal with a silver color, low density, and high strength, resistant to corrosion in ...
, vanadium
Vanadium is a chemical element; it has Symbol (chemistry), symbol V and atomic number 23. It is a hard, silvery-grey, malleable transition metal. The elemental metal is rarely found in nature, but once isolated artificially, the formation of an ...
, and niobium
Niobium is a chemical element; it has chemical symbol, symbol Nb (formerly columbium, Cb) and atomic number 41. It is a light grey, crystalline, and Ductility, ductile transition metal. Pure niobium has a Mohs scale of mineral hardness, Mohs h ...
are polymeric, melting or decomposing at no more than . Pentafluorides continue this trend with their linear polymers and oligomer
In chemistry and biochemistry, an oligomer () is a molecule that consists of a few repeating units which could be derived, actually or conceptually, from smaller molecules, monomers.Quote: ''Oligomer molecule: A molecule of intermediate relativ ...
ic complexes. Thirteen metal hexafluorides are known, all octahedral, and are mostly volatile solids but for liquid and , and gaseous .. Rhenium heptafluoride, the only characterized metal heptafluoride, is a low-melting molecular solid with pentagonal bipyramidal molecular geometry. Gold heptafluoride is a low-temperature complex of molecular F2 with AuF5, with NPA calculations indicating that the fluorine in the F2 ligand is nearly neutral while those in the AuF5 portion of the molecule have strong negative partial charges. This is consistent with the F2 ligand representing fluorine in the zero oxidation state. Metal fluorides with more fluorine atoms are particularly reactive.
Hydrogen
hydrogen chloride
The Chemical compound, compound hydrogen chloride has the chemical formula and as such is a hydrogen halide. At room temperature, it is a colorless gas, which forms white fumes of hydrochloric acid upon contact with atmospheric water vapor. Hyd ...
.... It boils at a much higher temperature than heavier hydrogen halides and unlike them is miscible
Miscibility () is the property of two substances to mix in all proportions (that is, to fully dissolve in each other at any concentration), forming a homogeneous mixture (a solution). Such substances are said to be miscible (etymologically ...
with water. Hydrogen fluoride readily hydrates on contact with water to form aqueous hydrogen fluoride, also known as hydrofluoric acid. Unlike the other hydrohalic acids, which are strong
Strong may refer to:
Education
* The Strong, an educational institution in Rochester, New York, United States
* Strong Hall (Lawrence, Kansas), an administrative hall of the University of Kansas
* Strong School, New Haven, Connecticut, United ...
, hydrofluoric acid is a weak acid
Acid strength is the tendency of an acid, symbolised by the chemical formula , to dissociate into a proton, , and an anion, . The dissociation or ionization of a strong acid in solution is effectively complete, except in its most concentrated s ...
at low concentrations. However, it can attack glass, something the other acids cannot do..
Other reactive nonmetals
 Binary fluorides of metalloids and p-block nonmetals are generally covalent and volatile, with varying reactivities. Period 3 and heavier nonmetals can form hypervalent fluorides..
Binary fluorides of metalloids and p-block nonmetals are generally covalent and volatile, with varying reactivities. Period 3 and heavier nonmetals can form hypervalent fluorides..
Boron trifluoride
Boron trifluoride is the inorganic compound with the formula . This pungent, colourless, and toxic gas forms white fumes in moist air. It is a useful Lewis acid and a versatile building block for other boron compounds.
Structure and bonding
The g ...
is planar and possesses an incomplete octet. It functions as a Lewis acid
A Lewis acid (named for the American physical chemist Gilbert N. Lewis) is a chemical species that contains an empty orbital which is capable of accepting an electron pair from a Lewis base to form a Lewis adduct. A Lewis base, then, is any ...
and combines with Lewis bases like ammonia to form adduct
In chemistry, an adduct (; alternatively, a contraction of "addition product") is a product of a direct addition of two or more distinct molecules, resulting in a single reaction product containing all atoms of all components. The resultant is ...
s. Carbon tetrafluoride
Tetrafluoromethane, also known as carbon tetrafluoride or R-14, is the simplest perfluorocarbon (Carbon, CFluorine, F4). As its IUPAC name indicates, tetrafluoromethane is the perfluorinated counterpart to the hydrocarbon methane. It can also be c ...
is tetrahedral and inert; its group analogues, silicon and germanium tetrafluoride, are also tetrahedral but behave as Lewis acids. The pnictogen
, -
! colspan=2 style="text-align:left;" , ↓ Period
, -
! 2
,
, -
! 3
,
, -
! 4
,
, -
! 5
,
, -
! 6
,
, -
! 7
,
, -
, colspan="2",
----
''Legend''
A pnictogen ( or ; from "to choke" and -gen, "generator") is any ...
s form trifluorides that increase in reactivity and basicity with higher molecular weight, although nitrogen trifluoride
Nitrogen trifluoride is the inorganic compound with the formula (). It is a colorless, non-flammable, toxic gas with a slightly musty odor. In contrast with ammonia, it is nonbasic. It finds increasing use within the manufacturing of flat-panel ...
resists hydrolysis and is not basic.. The pentafluorides of phosphorus, arsenic, and antimony are more reactive than their respective trifluorides, with antimony pentafluoride
Antimony pentafluoride is the inorganic compound with the formula Sb F5. This colorless, viscous liquid is a strong Lewis acid and a component of the superacid fluoroantimonic acid, formed upon mixing liquid HF with liquid SbF5 in 1:1 ratio. It ...
the strongest neutral Lewis acid known, only behind gold pentafluoride..
Chalcogen
The chalcogens (ore forming) ( ) are the chemical elements in group 16 of the periodic table. This group is also known as the oxygen family. Group 16 consists of the elements oxygen (O), sulfur (S), selenium (Se), tellurium (Te), and the rad ...
s have diverse fluorides: unstable difluorides have been reported for oxygen (the only known compound with oxygen in an oxidation state of +2), sulfur, and selenium; tetrafluorides and hexafluorides exist for sulfur, selenium, and tellurium. The latter are stabilized by more fluorine atoms and lighter central atoms, so sulfur hexafluoride
Sulfur hexafluoride or sulphur hexafluoride ( British spelling) is an inorganic compound with the formula SF6. It is a colorless, odorless, non-flammable, and non-toxic gas. has an octahedral geometry, consisting of six fluorine atoms attache ...
is especially inert.. Chlorine, bromine, and iodine can each form mono-, tri-, and pentafluorides, but only iodine heptafluoride has been characterized among possible interhalogen heptafluorides. Many of them are powerful sources of fluorine atoms, and industrial applications using chlorine trifluoride require precautions similar to those using fluorine..
Noble gases

Noble gas
The noble gases (historically the inert gases, sometimes referred to as aerogens) are the members of Group (periodic table), group 18 of the periodic table: helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), radon (Rn) and, in some ...
es, having complete electron shells, defied reaction with other elements until 1962 when Neil Bartlett reported synthesis of xenon hexafluoroplatinate
Xenon hexafluoroplatinate is the product of the reaction of platinum hexafluoride with xenon, in an experiment that proved the chemical reactivity of the noble gases. This experiment was performed by Neil Bartlett at the University of British Co ...
; xenon difluoride
Xenon is a chemical element; it has symbol Xe and atomic number 54. It is a dense, colorless, odorless noble gas found in Earth's atmosphere in trace amounts. Although generally unreactive, it can undergo a few chemical reactions such as th ...
, tetrafluoride, hexafluoride, and multiple oxyfluorides have been isolated since then. Among other noble gases, krypton forms a difluoride, and radon and fluorine generate a solid suspected to be radon difluoride.. Binary fluorides of lighter noble gases are exceptionally unstable: argon and hydrogen fluoride combine under extreme conditions to give argon fluorohydride. Helium has no long-lived fluorides,. and no neon fluoride has ever been observed; helium fluorohydride has been detected for milliseconds at high pressures and low temperatures.
Organic compounds
carbon–fluorine bond
The carbon–fluorine bond is a polar covalent bond between carbon and fluorine that is a component of all organofluorine compounds. It is one of the strongest single bonds in chemistry (after the B–F single bond, Si–F single bond, and H–F ...
is organic chemistry
Organic chemistry is a subdiscipline within chemistry involving the science, scientific study of the structure, properties, and reactions of organic compounds and organic matter, organic materials, i.e., matter in its various forms that contain ...
's strongest,. and gives stability to organofluorines. It is almost non-existent in nature, but is used in artificial compounds. Research in this area is usually driven by commercial applications; the compounds involved are diverse and reflect the complexity inherent in organic chemistry.
Discrete molecules
The substitution of hydrogen atoms in analkane
In organic chemistry, an alkane, or paraffin (a historical trivial name that also has other meanings), is an acyclic saturated hydrocarbon. In other words, an alkane consists of hydrogen and carbon atoms arranged in a tree structure in whi ...
by progressively more fluorine atoms gradually alters several properties: melting and boiling points are lowered, density increases, solubility in hydrocarbons decreases and overall stability increases. Perfluorocarbon
Fluorocarbons are chemical compounds with carbon-fluorine bonds. Compounds that contain many C-F bonds often have distinctive properties, e.g., enhanced stability, volatility, and hydrophobicity. Several fluorocarbons and their derivatives are ...
s, in which all hydrogen atoms are substituted, are insoluble in most organic solvents, reacting at ambient conditions only with sodium in liquid ammonia.
The term ''perfluorinated compound
A perfluorinated compound (PFC) or perfluoro compound is an Organofluorine chemistry, organofluorine compound that lacks C-H bonds. Many perfluorinated compounds have properties that are quite different from their C-H containing analogues. Common ...
'' is used for what would otherwise be a perfluorocarbon if not for the presence of a functional group
In organic chemistry, a functional group is any substituent or moiety (chemistry), moiety in a molecule that causes the molecule's characteristic chemical reactions. The same functional group will undergo the same or similar chemical reactions r ...
, often a carboxylic acid
In organic chemistry, a carboxylic acid is an organic acid that contains a carboxyl group () attached to an Substituent, R-group. The general formula of a carboxylic acid is often written as or , sometimes as with R referring to an organyl ...
. These compounds share many properties with perfluorocarbons such as stability and hydrophobicity
In chemistry, hydrophobicity is the chemical property of a molecule (called a hydrophobe) that is seemingly intermolecular force, repelled from a mass of water. In contrast, hydrophiles are attracted to water.
Hydrophobic molecules tend to b ...
, while the functional group augments their reactivity, enabling them to adhere to surfaces or act as surfactant
Surfactants are chemical compounds that decrease the surface tension or interfacial tension between two liquids, a liquid and a gas, or a liquid and a solid. The word ''surfactant'' is a Blend word, blend of "surface-active agent",
coined in ...
s.. Fluorosurfactant
Per- and polyfluoroalkyl substances (also PFAS, PFASs, and informally referred to as "forever chemicals") are a group of synthetic Organofluorine chemistry, organofluorine chemical compounds that have multiple fluorine atoms attached to an alkyl ...
s, in particular, can lower the surface tension
Surface tension is the tendency of liquid surfaces at rest to shrink into the minimum surface area possible. Surface tension (physics), tension is what allows objects with a higher density than water such as razor blades and insects (e.g. Ge ...
of water more than their hydrocarbon-based analogues. Fluorotelomers, which have some unfluorinated carbon atoms near the functional group, are also regarded as perfluorinated..
Polymers
Polymers exhibit the same stability increases afforded by fluorine substitution (for hydrogen) in discrete molecules; their melting points generally increase too.Polytetrafluoroethylene
Polytetrafluoroethylene (PTFE) is a synthetic fluoropolymer of tetrafluoroethylene, and has numerous applications because it is chemically inert. The commonly known brand name of PTFE-based composition is Teflon by Chemours, a corporate spin-of ...
(PTFE), the simplest fluoropolymer and perfluoro analogue of polyethylene
Polyethylene or polythene (abbreviated PE; IUPAC name polyethene or poly(methylene)) is the most commonly produced plastic. It is a polymer, primarily used for packaging (plastic bags, plastic films, geomembranes and containers including bott ...
with structural unit ––, demonstrates this change as expected, but its very high melting point makes it difficult to mold. Various PTFE derivatives are less temperature-tolerant but easier to mold: fluorinated ethylene propylene
Fluorinated ethylene propylene (FEP) is a copolymer of hexafluoropropylene and tetrafluoroethylene. It differs from the polytetrafluoroethylene (PTFE) resins in that it is melt-processable using conventional injection molding and Plastic extrusio ...
replaces some fluorine atoms with trifluoromethyl
The trifluoromethyl group is a functional group that has the formula . The naming of is group is derived from the methyl group (which has the formula ), by replacing each hydrogen atom by a fluorine atom. Some common examples are trifluoromethane ...
groups, perfluoroalkoxy alkane
Perfluoroalkoxy alkanes (PFA) are fluoropolymers. They are copolymers of tetrafluoroethylene (C2F4) and perfluoroethers (C2F3ORf, where Rf is a perfluorinated group such as trifluoromethyl (CF3)). The properties of these polymers are similar ...
s do the same with trifluoromethoxy groups, and Nafion
Nafion is a brand name for a sulfonated tetrafluoroethylene based fluoropolymer-copolymer synthesized in 1962 by Dr. Donald J. Connolly at the DuPont Experimental Station in Wilmington Delaware (U.S. Patent 3,282,875). Additional work on the polym ...
contains perfluoroether side chains capped with sulfonic acid
In organic chemistry, sulfonic acid (or sulphonic acid) refers to a member of the class of organosulfur compounds with the general formula , where R is an organic alkyl or aryl group and the group a sulfonyl hydroxide. As a substituent, it is kn ...
groups. Other fluoropolymers retain some hydrogen atoms; polyvinylidene fluoride
Polyvinylidene fluoride or polyvinylidene difluoride (PVDF) is a highly non-reactive thermoplastic fluoropolymer produced by the polymerization of vinylidene difluoride. Its chemical formula is (C2H2F2)''n''.
PVDF is a specialty plastic use ...
has half the fluorine atoms of PTFE and polyvinyl fluoride has a quarter, but both behave much like perfluorinated polymers.
Production
Elemental fluorine and virtually all fluorine compounds are produced fromhydrogen fluoride
Hydrogen fluoride (fluorane) is an Inorganic chemistry, inorganic compound with chemical formula . It is a very poisonous, colorless gas or liquid that dissolves in water to yield hydrofluoric acid. It is the principal industrial source of fluori ...
or its aqueous solution, hydrofluoric acid
Hydrofluoric acid is a solution of hydrogen fluoride (HF) in water. Solutions of HF are colorless, acidic and highly corrosive. A common concentration is 49% (48–52%) but there are also stronger solutions (e.g. 70%) and pure HF has a boiling p ...
. Hydrogen fluoride is produced in kiln
A kiln is a thermally insulated chamber, a type of oven, that produces temperatures sufficient to complete some process, such as hardening, drying, or Chemical Changes, chemical changes. Kilns have been used for millennia to turn objects m ...
s by the endothermic reaction
An endothermic process is a chemical or physical process that absorbs heat from its surroundings. In terms of thermodynamics, it is a thermodynamic process with an increase in the enthalpy (or internal energy ) of the system.Oxtoby, D. W; Gillis, ...
of fluorite
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon.
The Mohs scal ...
(CaF2) with sulfuric acid:
:CaF2 + H2SO4 → 2 HF(g) + CaSO4
The gaseous HF can then be absorbed in water or liquefied.
About 20% of manufactured HF is a byproduct of fertilizer production, which produces hexafluorosilicic acid (H2SiF6), which can be degraded to release HF thermally and by hydrolysis:
:H2SiF6 → 2 HF + SiF4
:SiF4 + 2 H2O → 4 HF + SiO2
Industrial routes to F2
 Moissan's method is used to produce industrial quantities of fluorine, via the electrolysis of a potassium bifluoride/
Moissan's method is used to produce industrial quantities of fluorine, via the electrolysis of a potassium bifluoride/hydrogen fluoride
Hydrogen fluoride (fluorane) is an Inorganic chemistry, inorganic compound with chemical formula . It is a very poisonous, colorless gas or liquid that dissolves in water to yield hydrofluoric acid. It is the principal industrial source of fluori ...
mixture: hydrogen ions are reduced at a steel container cathode
A cathode is the electrode from which a conventional current leaves a polarized electrical device such as a lead-acid battery. This definition can be recalled by using the mnemonic ''CCD'' for ''Cathode Current Departs''. Conventional curren ...
and fluoride ions are oxidized at a carbon block anode
An anode usually is an electrode of a polarized electrical device through which conventional current enters the device. This contrasts with a cathode, which is usually an electrode of the device through which conventional current leaves the devic ...
, under 8–12 volts, to generate hydrogen and fluorine gas respectively. Temperatures are elevated, KF•2HF melting at and being electrolyzed at . KF, which acts to provide electrical conductivity, is essential since pure HF cannot be electrolyzed because it is virtually non-conductive. Fluorine can be stored in steel cylinders that have passivated interiors, at temperatures below ; otherwise nickel can be used. Regulator valves and pipework are made of nickel, the latter possibly using Monel instead. Frequent passivation, along with the strict exclusion of water and greases, must be undertaken. In the laboratory, glassware may carry fluorine gas under low pressure and anhydrous conditions; some sources instead recommend nickel-Monel-PTFE systems..
Laboratory routes
While preparing for a 1986 conference to celebrate the centennial of Moissan's achievement, Karl O. Christe reasoned that chemical fluorine generation should be feasible since some metal fluoride anions have no stable neutral counterparts; their acidification potentially triggers oxidation instead. He devised a method which evolves fluorine at high yield and atmospheric pressure:. :2 KMnO4 + 2 KF + 10 HF + 3 H2O2 → 2 K2MnF6 + 8 H2O + 3 O2↑ :2 K2MnF6 + 4 SbF5 → 4 KSbF6 + 2 MnF3 + F2↑ Christe later commented that the reactants "had been known for more than 100 years and even Moissan could have come up with this scheme." As late as 2008, some references still asserted that fluorine was too reactive for any chemical isolation.Industrial applications
Fluorite mining, which supplies most global fluorine, peaked in 1989 when 5.6 millionmetric ton
The tonne ( or ; symbol: t) is a unit of mass equal to 1,000 kilograms. It is a non-SI unit accepted for use with SI. It is also referred to as a metric ton in the United States to distinguish it from the non-metric units of the sh ...
s of ore were extracted. Chlorofluorocarbon restrictions lowered this to 3.6 million tons in 1994; production has since been increasing. Around 4.5 million tons of ore and revenue of US$
The United States dollar (Currency symbol, symbol: Dollar sign, $; ISO 4217, currency code: USD) is the official currency of the United States and International use of the U.S. dollar, several other countries. The Coinage Act of 1792 introdu ...
550 million were generated in 2003; later reports estimated 2011 global fluorochemical sales at $15 billion and predicted 2016–18 production figures of 3.5 to 5.9 million tons, and revenue of at least $20 billion.. Froth flotation separates mined fluorite into two main metallurgical grades of equal proportion: 60–85% pure metspar is almost all used in iron smelting whereas 97%+ pure acidspar is mainly converted to the key industrial intermediate hydrogen fluoride..
Fluorite
Fluorite (also called fluorspar) is the mineral form of calcium fluoride, CaF2. It belongs to the halide minerals. It crystallizes in isometric cubic habit, although octahedral and more complex isometric forms are not uncommon.
The Mohs scal ...
rect 9 172 81 199 Fluorapatite
Fluorapatite, often with the alternate spelling of fluoroapatite, is a phosphate mineral with the formula Ca5(PO4)3F (calcium fluorophosphate). Fluorapatite is a hard crystalline solid. Although samples can have various color (green, brown, blu ...
rect 142 5 244 34 Hydrogen fluoride
Hydrogen fluoride (fluorane) is an Inorganic chemistry, inorganic compound with chemical formula . It is a very poisonous, colorless gas or liquid that dissolves in water to yield hydrofluoric acid. It is the principal industrial source of fluori ...
rect 142 65 245 97 Metal smelting
rect 142 121 244 154 Glass production
rect 309 5 411 33 Fluorocarbons
Fluorocarbons are chemical compounds with carbon-fluorine bonds. Compounds that contain many C-F bonds often have distinctive properties, e.g., enhanced stability, volatility, and hydrophobicity. Several fluorocarbons and their derivatives are ...
rect 310 63 413 92 Sodium hexafluoroaluminate
rect 311 121 414 154 Pickling (metal)
Pickling is a metal surface treatment used to remove impurities, such as stains, inorganic contaminants, and rust or scale from ferrous metals, copper, precious metals and aluminium alloys. A solution called ''pickle liquor'', which usually conta ...
rect 310 171 412 200 Fluorosilicic acid
rect 309 211 412 243 Alkane cracking
rect 483 6 585 34 Hydrofluorocarbon
Hydrofluorocarbons (HFCs) are synthetic organic compounds that contain fluorine and hydrogen atoms, and are the most common type of organofluorine compounds. Most are gases at room temperature and pressure. They are frequently used in air condit ...
s
rect 484 47 585 76 Hydrochlorofluorocarbons
rect 483 88 586 116 Chlorofluorocarbon
Chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs) are fully or partly Halogenation, halogenated hydrocarbons that contain carbon (C), hydrogen (H), chlorine (Cl), and fluorine (F). They are produced as volatility (chemistry), volat ...
rect 483 128 585 160 Teflon
Polytetrafluoroethylene (PTFE) is a synthetic fluoropolymer of tetrafluoroethylene, and has numerous applications because it is chemically inert. The commonly known brand name of PTFE-based composition is Teflon by Chemours, a spin-off from ...
rect 484 170 586 200 Water fluoridation
Water fluoridation is the controlled addition of fluoride to Public water supply, public water supplies to reduce tooth decay. Fluoridated water maintains fluoride levels effective for cavity prevention, achieved naturally or through supplem ...
rect 483 210 586 238 Uranium enrichment
Enriched uranium is a type of uranium in which the percent composition of uranium-235 (written 235U) has been increased through the process of isotope separation. Naturally occurring uranium is composed of three major isotopes: uranium-238 (23 ...
rect 484 258 586 287 Sulfur hexafluoride
Sulfur hexafluoride or sulphur hexafluoride ( British spelling) is an inorganic compound with the formula SF6. It is a colorless, odorless, non-flammable, and non-toxic gas. has an octahedral geometry, consisting of six fluorine atoms attache ...
rect 484 297 585 357 Tungsten hexafluoride
Tungsten(VI) fluoride, also known as tungsten hexafluoride, is an inorganic compound with the formula . It is a toxic, corrosive, colorless gas, with a density of about (roughly 11 times heavier than air). It is the densest known gas under stand ...
rect 28 246 177 293 Phosphogypsum
Phosphogypsum (PG) is the calcium sulfate hydrate formed as a by-product of the production of fertilizer, particularly phosphoric acid, from phosphate rock. It is mainly composed of gypsum (). Although gypsum is a widely used material in the cons ...
desc bottom-left
 At least 17,000 metric tons of fluorine are produced each year. It costs only $5–8 per kilogram as uranium or sulfur hexafluoride, but many times more as an element because of handling challenges. Most processes using free fluorine in large amounts employ ''in situ'' generation under
At least 17,000 metric tons of fluorine are produced each year. It costs only $5–8 per kilogram as uranium or sulfur hexafluoride, but many times more as an element because of handling challenges. Most processes using free fluorine in large amounts employ ''in situ'' generation under vertical integration
In microeconomics, management and international political economy, vertical integration, also referred to as vertical consolidation, is an arrangement in which the supply chain of a company is integrated and owned by that company. Usually each ...
.
The largest application of fluorine gas, consuming up to 7,000 metric tons annually, is in the preparation of for the nuclear fuel cycle
The nuclear fuel cycle, also known as the nuclear fuel chain, describes the series of stages that nuclear fuel undergoes during its production, use, and recycling or disposal. It consists of steps in the ''front end'', which are the preparation o ...
. Fluorine is used to fluorinate uranium tetrafluoride
Uranium tetrafluoride is the inorganic compound with the formula UF4. It is a green solid with an insignificant vapor pressure and low solubility in water. Uranium in its tetravalent ( uranous) state is important in various technological process ...
, itself formed from uranium dioxide and hydrofluoric acid. Fluorine is monoisotopic, so any mass differences between molecules are due to the presence of or , enabling uranium enrichment via gaseous diffusion or gas centrifuge
A gas centrifuge is a device that performs isotope separation of gases. A centrifuge relies on the principles of centrifugal force accelerating molecules so that particles of different masses are physically separated in a gradient along the radiu ...
. About 6,000 metric tons per year go into producing the inert dielectric
In electromagnetism, a dielectric (or dielectric medium) is an Insulator (electricity), electrical insulator that can be Polarisability, polarised by an applied electric field. When a dielectric material is placed in an electric field, electric ...
for high-voltage transformers and circuit breakers, eliminating the need for hazardous polychlorinated biphenyl
Polychlorinated biphenyls (PCBs) are organochlorine compounds with the formula Carbon, C12Hydrogen, H10−''x''Chloride, Cl''x''; they were once widely used in the manufacture of carbonless copy paper, as heat transfer fluids, and as dielectri ...
s associated with devices. Several fluorine compounds are used in electronics: rhenium and tungsten hexafluoride in chemical vapor deposition
Chemical vapor deposition (CVD) is a vacuum deposition method used to produce high-quality, and high-performance, solid materials. The process is often used in the semiconductor industry to produce thin films.
In typical CVD, the wafer (electro ...
, tetrafluoromethane in plasma etching
Plasma etching is a form of plasma processing used to fabricate integrated circuits. It involves a high-speed stream of glow discharge (Plasma (physics), plasma) of an appropriate gas mixture being shot (in pulses) at a sample. The plasma source, ...
and nitrogen trifluoride
Nitrogen trifluoride is the inorganic compound with the formula (). It is a colorless, non-flammable, toxic gas with a slightly musty odor. In contrast with ammonia, it is nonbasic. It finds increasing use within the manufacturing of flat-panel ...
in cleaning equipment. Fluorine is also used in the synthesis of organic fluorides, but its reactivity often necessitates conversion first to the gentler , , or , which together allow calibrated fluorination. Fluorinated pharmaceuticals use sulfur tetrafluoride instead.
Inorganic fluorides
viscosity
Viscosity is a measure of a fluid's rate-dependent drag (physics), resistance to a change in shape or to movement of its neighboring portions relative to one another. For liquids, it corresponds to the informal concept of ''thickness''; for e ...
.. Alongside its role as an additive in materials like enamels and welding rod coats, most acidspar is reacted with sulfuric acid to form hydrofluoric acid, which is used in steel pickling
Pickling is the process of food preservation, preserving or extending the shelf life of food by either Anaerobic organism, anaerobic fermentation (food), fermentation in brine or immersion in vinegar. The pickling procedure typically affects t ...
, glass etching and alkane cracking. One-third of HF goes into synthesizing cryolite
Cryolite ( Na3 Al F6, sodium hexafluoroaluminate) is a rare mineral identified with the once-large deposit at Ivittuut on the west coast of Greenland, mined commercially until 1987.
It is used in the reduction ("smelting") of aluminium, in pest ...
and aluminium trifluoride
Aluminium fluoride is an inorganic compound with the formula . It forms hydrates . Anhydrous and its hydrates are all colorless solids. Anhydrous is used in the production of aluminium. Several occur as minerals.
Occurrence and production
Aside ...
, both fluxes in the Hall–Héroult process
The Hall–Héroult process is the major industrial process for smelting aluminium. It involves dissolving aluminium oxide (alumina) (obtained most often from bauxite, aluminium's chief ore, through the Bayer process) in molten cryolite and e ...
for aluminium extraction; replenishment is necessitated by their occasional reactions with the smelting apparatus. Each metric ton of aluminium requires about of flux. Fluorosilicates consume the second largest portion, with sodium fluorosilicate used in water fluoridation and laundry effluent treatment, and as an intermediate en route to cryolite and silicon tetrafluoride. Other important inorganic fluorides include those of cobalt
Cobalt is a chemical element; it has Symbol (chemistry), symbol Co and atomic number 27. As with nickel, cobalt is found in the Earth's crust only in a chemically combined form, save for small deposits found in alloys of natural meteoric iron. ...
, nickel
Nickel is a chemical element; it has symbol Ni and atomic number 28. It is a silvery-white lustrous metal with a slight golden tinge. Nickel is a hard and ductile transition metal. Pure nickel is chemically reactive, but large pieces are slo ...
, and ammonium
Ammonium is a modified form of ammonia that has an extra hydrogen atom. It is a positively charged (cationic) polyatomic ion, molecular ion with the chemical formula or . It is formed by the protonation, addition of a proton (a hydrogen nucleu ...
.
Organic fluorides
Organofluorides consume over 20% of mined fluorite and over 40% of hydrofluoric acid, with refrigerant gases dominating andfluoropolymers
A fluoropolymer is a fluorocarbon-based polymer with multiple carbon–fluorine bonds. It is characterized by a high resistance to solvents, acids, and Base (chemistry), bases. The best known fluoropolymer is polytetrafluoroethylene under the bra ...
increasing their market share.. Surfactants
Surfactants are chemical compounds that decrease the surface tension or interfacial tension between two liquids, a liquid and a gas, or a liquid and a solid. The word ''surfactant'' is a blend of "surface-active agent",
coined in 1950. As t ...
are a minor application but generate over $1 billion in annual revenue.. Due to the danger from direct hydrocarbon–fluorine reactions above , industrial fluorocarbon production is indirect, mostly through halogen exchange reactions such as Swarts fluorination, in which chlorocarbon chlorines are substituted for fluorines by hydrogen fluoride under catalysts. Electrochemical fluorination subjects hydrocarbons to electrolysis in hydrogen fluoride, and the Fowler process treats them with solid fluorine carriers like cobalt trifluoride.
Refrigerant gases
Halogenated refrigerants, termed Freons in informal contexts, are identified by R-numbers that denote the amount of fluorine, chlorine, carbon, and hydrogen present.Chlorofluorocarbons
Chlorofluorocarbons (CFCs) and hydrochlorofluorocarbons (HCFCs) are fully or partly halogenated hydrocarbons that contain carbon (C), hydrogen (H), chlorine (Cl), and fluorine (F). They are produced as volatile derivatives of methane, ethane, ...
(CFCs) like R-11, R-12, and R-114 once dominated organofluorines, peaking in production in the 1980s. Used for air conditioning systems, propellants and solvents, their production was below one-tenth of this peak by the early 2000s, after widespread international prohibition. Hydrochlorofluorocarbons (HCFCs) and hydrofluorocarbons (HFCs) were designed as replacements; their synthesis consumes more than 90% of the fluorine in the organic industry. Important HCFCs include R-22, chlorodifluoromethane
Chlorodifluoromethane or difluoromonochloromethane is a hydrochlorofluorocarbon (HCFC). This colorless gas is better known as HCFC-22, or R-22, or . It was commonly used as a propellant and refrigerant. These applications were phased out under ...
, and R-141b. The main HFC is R-134a
1,1,1,2-Tetrafluoroethane (also known as norflurane ( INN), R-134a, Klea 134a, Freon 134a, Forane 134a, Genetron 134a, Green Gas, Florasol 134a, Suva 134a, HFA-134a, or HFC-134a) is a hydrofluorocarbon (HFC) and haloalkane refrigerant with therm ...
with a new type of molecule HFO-1234yf, a Hydrofluoroolefin
Hydrofluoroolefins (HFOs) are Saturated and unsaturated compounds, unsaturated organic compounds composed of hydrogen, fluorine and carbon. These organofluorine compounds are of interest as refrigerants. Unlike traditional hydrofluorocarbons (HFCs ...
(HFO) coming to prominence owing to its global warming potential
Global warming potential (GWP) is a measure of how much heat a greenhouse gas traps in the atmosphere over a specific time period, relative to carbon dioxide (). It is expressed as a multiple of warming caused by the same mass of carbon dioxide ( ...
of less than 1% that of HFC-134a..
Polymers
 About 180,000 metric tons of fluoropolymers were produced in 2006 and 2007, generating over $3.5 billion revenue per year.. The global market was estimated at just under $6 billion in 2011. Fluoropolymers can only be formed by polymerizing free radicals.
Polytetrafluoroethylene (PTFE), sometimes called by its DuPont name Teflon, represents 60–80% by mass of the world's fluoropolymer production. The largest application is in
About 180,000 metric tons of fluoropolymers were produced in 2006 and 2007, generating over $3.5 billion revenue per year.. The global market was estimated at just under $6 billion in 2011. Fluoropolymers can only be formed by polymerizing free radicals.
Polytetrafluoroethylene (PTFE), sometimes called by its DuPont name Teflon, represents 60–80% by mass of the world's fluoropolymer production. The largest application is in electrical insulation
Electricity is the set of physical phenomena associated with the presence and motion of matter possessing an electric charge. Electricity is related to magnetism, both being part of the phenomenon of electromagnetism, as described by ...
since PTFE is an excellent dielectric
In electromagnetism, a dielectric (or dielectric medium) is an Insulator (electricity), electrical insulator that can be Polarisability, polarised by an applied electric field. When a dielectric material is placed in an electric field, electric ...
. It is also used in the chemical industry where corrosion resistance is needed, in coating pipes, tubing, and gaskets. Another major use is in PFTE-coated fiberglass cloth for stadium roofs. The major consumer application is for non-stick cookware.. Jerked PTFE film becomes expanded PTFE (ePTFE), a fine-pored membrane
A membrane is a selective barrier; it allows some things to pass through but stops others. Such things may be molecules, ions, or other small particles. Membranes can be generally classified into synthetic membranes and biological membranes. Bi ...
sometimes referred to by the brand name Gore-Tex and used for rainwear, protective apparel, and filters
Filtration is a physical process that separates solid matter and fluid from a mixture.
Filter, filtering, filters or filtration may also refer to:
Science and technology
Computing
* Filter (higher-order function), in functional programming
* Fil ...
; ePTFE fibers may be made into seals and dust filters. Other fluoropolymers, including fluorinated ethylene propylene
Fluorinated ethylene propylene (FEP) is a copolymer of hexafluoropropylene and tetrafluoroethylene. It differs from the polytetrafluoroethylene (PTFE) resins in that it is melt-processable using conventional injection molding and Plastic extrusio ...
, mimic PTFE's properties and can substitute for it; they are more moldable, but also more costly and have lower thermal stability. Films from two different fluoropolymers replace glass in solar cells..
The chemically resistant (but expensive) fluorinated ionomers are used as electrochemical cell membranes, of which the first and most prominent example is Nafion
Nafion is a brand name for a sulfonated tetrafluoroethylene based fluoropolymer-copolymer synthesized in 1962 by Dr. Donald J. Connolly at the DuPont Experimental Station in Wilmington Delaware (U.S. Patent 3,282,875). Additional work on the polym ...
. Developed in the 1960s, it was initially deployed as fuel cell material in spacecraft and then replaced mercury-based chloralkali process
The chloralkali process (also chlor-alkali and chlor alkali) is an industrial process for the electrolysis of sodium chloride (NaCl) solutions. It is the technology used to produce chlorine and sodium hydroxide (caustic soda), which are commodi ...
cells. Recently, the fuel cell application has reemerged with efforts to install proton exchange membrane fuel cells into automobiles.... Fluoroelastomers such as Viton are crosslink
In chemistry and biology, a cross-link is a bond or a short sequence of bonds that links one polymer chain to another. These links may take the form of covalent bonds or ionic bonds and the polymers can be either synthetic polymers or natural ...
ed fluoropolymer mixtures mainly used in O-ring
An O-ring, also known as a packing or a toric joint, is a mechanical gasket in the shape of a torus; it is a loop of elastomer with a round cross section (geometry), cross-section, designed to be seated in a groove and compressed during assembl ...
s; perfluorobutane (C4F10) is used as a fire-extinguishing agent.
Surfactants
Fluorosurfactants are small organofluorine molecules used for repelling water and stains. Although expensive (comparable to pharmaceuticals at $200–2000 per kilogram), they yielded over $1 billion in annual revenues by 2006;Scotchgard
Scotchgard is a 3M brand of stain repellent and durable water repellent applied to fabrics, upholstery, and carpets to protect them from stains. Scotchgard products typically are based on organofluorine chemicals dissolved in a petroleum d ...
alone generated over $300 million in 2000... Fluorosurfactants are a minority in the overall surfactant market, most of which is taken up by much cheaper hydrocarbon-based products. Applications in paints
Paint is a material or mixture that, when applied to a solid material and allowed to dry, adds a film-like layer. As art, this is used to create an image or images known as a painting. Paint can be made in many colors and types. Most paints are ...
are burdened by compounding
In the field of pharmacy, compounding (performed in compounding pharmacies) is preparation of custom medications to fit unique needs of patients that cannot be met with mass-produced formulations. This may be done, for example, to provide medic ...
costs; this use was valued at only $100 million in 2006.
Agrichemicals
About 30% of agrichemicals contain fluorine, most of themherbicide
Herbicides (, ), also commonly known as weed killers, are substances used to control undesired plants, also known as weeds.EPA. February 201Pesticides Industry. Sales and Usage 2006 and 2007: Market Estimates. Summary in press releasMain page f ...
s and fungicide
Fungicides are pesticides used to kill parasitic fungi or their spores. Fungi can cause serious damage in agriculture, resulting in losses of yield and quality. Fungicides are used both in agriculture and to fight fungal infections in animals, ...
s with a few crop regulators. Fluorine substitution, usually of a single atom or at most a trifluoromethyl
The trifluoromethyl group is a functional group that has the formula . The naming of is group is derived from the methyl group (which has the formula ), by replacing each hydrogen atom by a fluorine atom. Some common examples are trifluoromethane ...
group, is a robust modification with effects analogous to fluorinated pharmaceuticals: increased biological stay time, membrane crossing, and altering of molecular recognition. Trifluralin is a prominent example, with large-scale use in the U.S. as a weedkiller,.. but it is a suspected carcinogen and has been banned in many European countries. Sodium monofluoroacetate (1080) is a mammalian poison in which one sodium acetate
Sodium acetate, CH3COONa, also abbreviated Sodium, NaOxygen, OAcetyl, Ac, is the sodium Salt (chemistry), salt of acetic acid. This salt is colorless, deliquescent, and hygroscopy, hygroscopic.
Applications
Biotechnological
Sodium acetate is u ...
hydrogen is replaced with fluorine; it disrupts cell metabolism by replacing acetate in the citric acid cycle
The citric acid cycle—also known as the Krebs cycle, Szent–Györgyi–Krebs cycle, or TCA cycle (tricarboxylic acid cycle)—is a series of chemical reaction, biochemical reactions that release the energy stored in nutrients through acetyl-Co ...
. First synthesized in the late 19th century, it was recognized as an insecticide
Insecticides are pesticides used to kill insects. They include ovicides and larvicides used against insect eggs and larvae, respectively. The major use of insecticides is in agriculture, but they are also used in home and garden settings, i ...
in the early 20th century, and was later deployed in its current use. New Zealand, the largest consumer of 1080, uses it to protect kiwis from the invasive Australian common brushtail possum
The common brushtail possum (''Trichosurus vulpecula'', from the Ancient Greek, Greek for "furry tailed" and the Latin for "little fox", previously in the genus ''Phalangista'') is a nocturnal, semiarboreal marsupial of the family Phalangeridae ...
.. Europe and the U.S. have banned 1080.
Medicinal applications
Dental care
Population studies from the mid-20th century onwards showtopical
A topical medication is a medication that is applied to a particular place on or in the body. Most often topical medication means application to body surfaces such as the skin or mucous membranes to treat ailments via a large range of classes ...
fluoride reduces dental caries
Tooth decay, also known as caries,The word 'caries' is a mass noun, and is not a plural of 'carie'.'' is the breakdown of teeth due to acids produced by bacteria. The resulting cavities may be a number of different colors, from yellow to black ...
. This was first attributed to the conversion of tooth enamel hydroxyapatite
Hydroxyapatite (International Mineralogical Association, IMA name: hydroxylapatite) (Hap, HAp, or HA) is a naturally occurring mineral form of calcium apatite with the Chemical formula, formula , often written to denote that the Crystal struc ...
into the more durable fluorapatite, but studies on pre-fluoridated teeth refuted this hypothesis, and current theories involve fluoride aiding enamel growth in small caries.. After studies of children in areas where fluoride was naturally present in drinking water, controlled public water supply fluoridation to fight tooth decay began in the 1940s and is now applied to water supplying 6 percent of the global population, including two-thirds of Americans... Reviews of the scholarly literature in 2000 and 2007 associated water fluoridation with a significant reduction of tooth decay in children.; see for a summary. Despite such endorsements and evidence of no adverse effects other than mostly benign dental fluorosis
Dental fluorosis is a common disorder, characterized by Enamel hypocalcification, hypocalcification of tooth enamel caused by ingestion of excessive fluoride during enamel formation.
Dental fluorosis appears as a range of visual changes in ename ...
, opposition still exists on ethical and safety grounds. The benefits of fluoridation have lessened, possibly due to other fluoride sources, but are still measurable in low-income groups. Sodium monofluorophosphate and sometimes sodium or tin(II) fluoride are often found in fluoride toothpaste
Toothpaste is a paste or gel dentifrice that is used with a toothbrush to clean and maintain the aesthetics of Human tooth, teeth. Toothpaste is used to promote oral hygiene: it is an abrasive that aids in removing dental plaque and food from th ...
s, first introduced in the U.S. in 1955 and now ubiquitous in developed countries, alongside fluoridated mouthwashes, gels, foams, and varnishes...
Pharmaceuticals
 Twenty percent of modern pharmaceuticals contain fluorine.. One of these, the cholesterol-reducer
Twenty percent of modern pharmaceuticals contain fluorine.. One of these, the cholesterol-reducer atorvastatin
Atorvastatin, sold under the brand name Lipitor among others, is a statin medication used to prevent cardiovascular disease in those at high risk and to treat abnormal lipid levels. For the prevention of cardiovascular disease, statins are a ...
(Lipitor), made more revenue than any other drug until it became generic in 2011.. The combination asthma prescription Seretide, a top-ten revenue drug in the mid-2000s, contains two active ingredients, one of which – fluticasone – is fluorinated. Many drugs are fluorinated to delay inactivation and lengthen dosage periods because the carbon–fluorine bond is very stable.. Fluorination also increases lipophilicity
Lipophilicity (from Greek λίπος "fat" and φίλος "friendly") is the ability of a chemical compound to dissolve in fats, oils, lipids, and non-polar solvents such as hexane or toluene. Such compounds are called lipophilic (translated ...
because the bond is more hydrophobic than the carbon–hydrogen bond
In chemistry, the carbon–hydrogen bond ( bond) is a chemical bond between carbon and hydrogen atoms that can be found in many organic compounds. This bond is a covalent bond, covalent, single bond, meaning that carbon shares its outer valence el ...
, and this often helps in cell membrane penetration and hence bioavailability
In pharmacology, bioavailability is a subcategory of absorption and is the fraction (%) of an administered drug that reaches the systemic circulation.
By definition, when a medication is administered intravenously, its bioavailability is 100%. H ...
..
Tricyclics and other pre-1980s antidepressant
Antidepressants are a class of medications used to treat major depressive disorder, anxiety disorders, chronic pain, and addiction.
Common side effects of antidepressants include Xerostomia, dry mouth, weight gain, dizziness, headaches, akathi ...
s had several side effects due to their non-selective interference with neurotransmitter
A neurotransmitter is a signaling molecule secreted by a neuron to affect another cell across a Chemical synapse, synapse. The cell receiving the signal, or target cell, may be another neuron, but could also be a gland or muscle cell.
Neurotra ...
s other than the serotonin
Serotonin (), also known as 5-hydroxytryptamine (5-HT), is a monoamine neurotransmitter with a wide range of functions in both the central nervous system (CNS) and also peripheral tissues. It is involved in mood, cognition, reward, learning, ...
target; the fluorinated fluoxetine
Fluoxetine, sold under the brand name Prozac, among others, is an Antidepressant, antidepressant medication of the selective serotonin reuptake inhibitor (SSRI) class used for the treatment of major depressive disorder, Anxiety disorder, anx ...
was selective and one of the first to avoid this problem. Many current antidepressants receive this same treatment, including the selective serotonin reuptake inhibitor
Selective serotonin reuptake inhibitors (SSRIs) are a class of drugs that are typically used as antidepressants in the treatment of major depressive disorder, anxiety disorders, and other psychological conditions.
SSRIs primarily work by blo ...
s: citalopram
Citalopram, sold under the brand name Celexa among others, is an antidepressant of the selective serotonin reuptake inhibitor (SSRI) class. It is used to treat major depressive disorder, obsessive compulsive disorder, panic disorder, and soci ...
, its enantiomer
In chemistry, an enantiomer (Help:IPA/English, /ɪˈnænti.əmər, ɛ-, -oʊ-/ Help:Pronunciation respelling key, ''ih-NAN-tee-ə-mər''), also known as an optical isomer, antipode, or optical antipode, is one of a pair of molecular entities whi ...
escitalopram
Escitalopram ( ), sold under the brand names Lexapro and Cipralex, among others, is an antidepressant medication of the selective serotonin reuptake inhibitor (SSRI) class. It is mainly used to treat major depressive disorder, generalized ...
, and fluvoxamine
Fluvoxamine, sold under the brand name Luvox among others, is an antidepressant of the selective serotonin reuptake inhibitor (SSRI) class. It is primarily used to treat major depressive disorder and, perhaps more-especially, obsessive–compu ...
and paroxetine
Paroxetine ( ), sold under the brand name Paxil among others, is an Antidepressant, antidepressant medication of the selective serotonin reuptake inhibitor (SSRI) class used to treat major depressive disorder, obsessive-compulsive disorder, o ...
. Quinolones are artificial broad-spectrum antibiotic
A broad-antimicrobial spectrum, spectrum antibiotic is an antibiotic that acts on the two major bacterial groups, Gram-positive and Gram-negative, or any antibiotic that acts against a wide range of disease-causing bacteria. These medications are ...
s that are often fluorinated to enhance their effects. These include ciprofloxacin
Ciprofloxacin is a fluoroquinolone antibiotic used to treat a number of bacterial infections. This includes bone and joint infections, intra-abdominal infections, certain types of infectious diarrhea, respiratory tract infections, skin ...
and levofloxacin
Levofloxacin, sold under the brand name Levaquin among others, is a broad-spectrum antibiotic of the fluoroquinolone drug class. It is the left-handed isomer of the medication ofloxacin. It is used to treat a number of bacterial infections ...
.. Fluorine also finds use in steroids: fludrocortisone is a blood pressure-raising mineralocorticoid
Mineralocorticoids are a class of corticosteroids, which in turn are a class of steroid hormones. Mineralocorticoids are produced in the adrenal cortex and influence salt and water balances (electrolyte balance and fluid balance). The primary ...
, and triamcinolone and dexamethasone
Dexamethasone is a fluorinated glucocorticoid medication used to treat rheumatic problems, a number of skin diseases, severe allergies, asthma, chronic obstructive pulmonary disease (COPD), croup, brain swelling, eye pain following eye su ...
are strong glucocorticoid
Glucocorticoids (or, less commonly, glucocorticosteroids) are a class of corticosteroids, which are a class of steroid hormones. Glucocorticoids are corticosteroids that bind to the glucocorticoid receptor that is present in almost every vertebra ...
s.. The majority of inhaled anesthetic
An anesthetic (American English) or anaesthetic (British English; see spelling differences) is a drug used to induce anesthesia — in other words, to result in a temporary loss of sensation or awareness. They may be divided into t ...
s are heavily fluorinated; the prototype halothane
Halothane, sold under the brand name Fluothane among others, is a general anaesthetic. It can be used to induce or maintain anaesthesia. One of its benefits is that it does not increase the production of saliva, which can be particularly useful ...
is much more inert and potent than its contemporaries. Later compounds such as the fluorinated ether
In organic chemistry, ethers are a class of compounds that contain an ether group, a single oxygen atom bonded to two separate carbon atoms, each part of an organyl group (e.g., alkyl or aryl). They have the general formula , where R and R� ...
s sevoflurane
Sevoflurane, sold under the brand name Sevorane, among others, is a sweet-smelling, nonflammable, highly fluorinated methyl isopropyl ether used as an inhalational anaesthetic for induction and maintenance of general anesthesia. After desflu ...
and desflurane
Desflurane (1,2,2,2-tetrafluoroethyl difluoromethyl ether) is a highly fluorinated methyl ethyl ether used for maintenance of general anesthesia. Like halothane, enflurane, and isoflurane, it is a racemic mixture of (''R'') and (''S'') optic ...
are better than halothane and are almost insoluble in blood, allowing faster waking times...
PET scanning
 Fluorine-18 is often found in
Fluorine-18 is often found in radioactive tracer
A radioactive tracer, radiotracer, or radioactive label is a synthetic derivative of a natural compound in which one or more atoms have been replaced by a radionuclide (a radioactive atom). By virtue of its radioactive decay, it can be used to ...
s for positron emission tomography, as its half-life of almost two hours is long enough to allow for its transport from production facilities to imaging centers.. The most common tracer is fluorodeoxyglucose which, after intravenous injection, is taken up by glucose-requiring tissues such as the brain and most malignant tumors; computer-assisted tomography can then be used for detailed imaging.
Oxygen carriers
Liquid fluorocarbons can hold large volumes of oxygen or carbon dioxide, more so than blood, and have attracted attention for their possible uses in artificial blood and in liquid breathing.. Because fluorocarbons do not normally mix with water, they must be mixed into emulsions (small droplets of perfluorocarbon suspended in water) to be used as blood.. One such product, Oxycyte, has been through initial clinical trials. These substances can aid endurance athletes and are banned from sports; one cyclist's near death in 1998 prompted an investigation into their abuse. Applications of pure perfluorocarbon liquid breathing (which uses pure perfluorocarbon liquid, not a water emulsion) include assisting burn victims and premature babies with deficient lungs. Partial and complete lung filling have been considered, though only the former has had any significant tests in humans.. An Alliance Pharmaceuticals effort reached clinical trials but was abandoned because the results were not better than normal therapies..Biological role
Fluorine is not essential for humans and other mammals, but small amounts are known to be beneficial for the strengthening of dental enamel (where the formation of fluorapatite makes the enamel more resistant to attack, from acids produced by bacterial fermentation of sugars). Small amounts of fluorine may be beneficial for bone strength, but the latter has not been definitively established. Both the WHO and the Institute of Medicine of the US National Academies publish recommended daily allowance (RDA) and upper tolerated intake of fluorine, which varies with age and gender. Natural organofluorines have been found in microorganisms, plants and, recently, animals. The most common is fluoroacetate, which is used as a defense against herbivores by at least 40 plants in Africa, Australia and Brazil. Other examples include terminally fluorinatedfatty acid
In chemistry, in particular in biochemistry, a fatty acid is a carboxylic acid with an aliphatic chain, which is either saturated and unsaturated compounds#Organic chemistry, saturated or unsaturated. Most naturally occurring fatty acids have an ...
s, fluoroacetone, and 2-fluorocitrate. An enzyme that binds fluorine to carbon – adenosyl-fluoride synthase – was discovered in bacteria in 2002.
Toxicity
Elemental fluorine is highly toxic to living organisms. Its effects in humans start at concentrations lower thanhydrogen cyanide
Hydrogen cyanide (formerly known as prussic acid) is a chemical compound with the chemical formula, formula HCN and structural formula . It is a highly toxic and flammable liquid that boiling, boils slightly above room temperature, at . HCN is ...
's 50 ppm and are similar to those of chlorine: significant irritation of the eyes and respiratory system as well as liver and kidney damage occur above 25 ppm, which is the immediately dangerous to life and health value for fluorine.. The eyes and nose are seriously damaged at 100 ppm, and inhalation of 1,000 ppm fluorine will cause death in minutes, compared to 270 ppm for hydrogen cyanide.
Hydrofluoric acid
 Hydrofluoric acid is the weakest of the hydrohalic acids, having a pKa of 3.2 at 25 °C. Pure hydrogen fluoride is a volatile liquid due to the presence of hydrogen bonding, while the other hydrogen halides are gases. It is able to attack glass, concrete, metals, and organic matter.
Hydrofluoric acid is a contact poison with greater hazards than many strong acids like sulfuric acid even though it is weak: it remains neutral in aqueous solution and thus penetrates tissue faster, whether through inhalation, ingestion or the skin, and at least nine U.S. workers died in such accidents from 1984 to 1994. It reacts with calcium and magnesium in the blood leading to
Hydrofluoric acid is the weakest of the hydrohalic acids, having a pKa of 3.2 at 25 °C. Pure hydrogen fluoride is a volatile liquid due to the presence of hydrogen bonding, while the other hydrogen halides are gases. It is able to attack glass, concrete, metals, and organic matter.
Hydrofluoric acid is a contact poison with greater hazards than many strong acids like sulfuric acid even though it is weak: it remains neutral in aqueous solution and thus penetrates tissue faster, whether through inhalation, ingestion or the skin, and at least nine U.S. workers died in such accidents from 1984 to 1994. It reacts with calcium and magnesium in the blood leading to hypocalcemia
Hypocalcemia is a medical condition characterized by low calcium levels in the blood serum. The normal range of blood calcium is typically between 2.1–2.6 mmol/L (8.8–10.7 mg/dL, 4.3–5.2 mEq/L), while levels less than 2.1 ...
and possible death through cardiac arrhythmia
Arrhythmias, also known as cardiac arrhythmias, are irregularities in the heartbeat, including when it is too fast or too slow. Essentially, this is anything but normal sinus rhythm. A resting heart rate that is too fast – above 100 beat ...
.. Insoluble calcium fluoride formation triggers strong pain. and burns larger than 160 cm2 (25 in2) can cause serious systemic toxicity..
Exposure may not be evident for eight hours for 50% HF, rising to 24 hours for lower concentrations, and a burn may initially be painless as hydrogen fluoride affects nerve function. If skin has been exposed to HF, damage can be reduced by rinsing it under a jet of water for 10–15 minutes and removing contaminated clothing. Calcium gluconate is often applied next, providing calcium ions to bind with fluoride; skin burns can be treated with 2.5% calcium gluconate gel or special rinsing solutions.... Hydrofluoric acid absorption requires further medical treatment; calcium gluconate may be injected or administered intravenously. Using calcium chloride – a common laboratory reagent – in lieu of calcium gluconate is contraindicated, and may lead to severe complications. Excision or amputation of affected parts may be required.
Fluoride ion
Soluble fluorides are moderately toxic: 5–10 g sodium fluoride, or 32–64 mg fluoride ions per kilogram of body mass, represents a lethal dose for adults. One-fifth of the lethal dose can cause adverse health effects, and chronic excess consumption may lead to skeletal fluorosis, which affects millions in Asia and Africa, and, in children, to reduced intelligence.. Ingested fluoride forms hydrofluoric acid in the stomach which is easily absorbed by the intestines, where it crosses cell membranes, binds with calcium and interferes with various enzymes, before urinaryexcretion
Excretion is elimination of metabolic waste, which is an essential process in all organisms. In vertebrates, this is primarily carried out by the lungs, Kidney (vertebrates), kidneys, and skin. This is in contrast with secretion, where the substa ...
. Exposure limits are determined by urine testing of the body's ability to clear fluoride ions..
Historically, most cases of fluoride poisoning have been caused by accidental ingestion of insecticides containing inorganic fluorides.. Most current calls to poison control centers for possible fluoride poisoning come from the ingestion of fluoride-containing toothpaste. Malfunctioning water fluoridation equipment is another cause: one incident in Alaska affected almost 300 people and killed one person.. Dangers from toothpaste are aggravated for small children, and the Centers for Disease Control and Prevention
The Centers for Disease Control and Prevention (CDC) is the National public health institutes, national public health agency of the United States. It is a Federal agencies of the United States, United States federal agency under the United S ...
recommends supervising children below six brushing their teeth so that they do not swallow toothpaste. One regional study examined a year of pre-teen fluoride poisoning reports totaling 87 cases, including one death from ingesting insecticide. Most had no symptoms, but about 30% had stomach pains. A larger study across the U.S. had similar findings: 80% of cases involved children under six, and there were few serious cases.
Environmental concerns
Atmosphere
 The
The Montreal Protocol
The Montreal Protocol on Substances That Deplete the Ozone Layer is an international treaty designed to protect the ozone layer by phasing out the production of numerous substances that are responsible for ozone depletion. It was agreed on 16 ...
, signed in 1987, set strict regulations on chlorofluorocarbons (CFCs) and bromofluorocarbon
Bromofluorocarbons (BFCs) are molecules based on carbon, bromine, and fluorine. The most common use has traditionally been in Wildfire suppression, fire suppression systems. The brand name "Halon" is frequently used interchangeably for BFCs. ow ...
s due to their ozone damaging potential (ODP). The high stability which suited them to their original applications also meant that they were not decomposing until they reached higher altitudes, where liberated chlorine and bromine atoms attacked ozone molecules.. Even with the ban, and early indications of its efficacy, predictions warned that several generations would pass before full recovery. With one-tenth the ODP of CFCs, hydrochlorofluorocarbons (HCFCs) are the current replacements,. and are themselves scheduled for substitution by 2030–2040 by hydrofluorocarbons (HFCs) with no chlorine and zero ODP.. In 2007 this date was brought forward to 2020 for developed countries;. the Environmental Protection Agency had already prohibited one HCFC's production and capped those of two others in 2003. Fluorocarbon gases are generally greenhouse gas
Greenhouse gases (GHGs) are the gases in the atmosphere that raise the surface temperature of planets such as the Earth. Unlike other gases, greenhouse gases absorb the radiations that a planet emits, resulting in the greenhouse effect. T ...
es with global-warming potential
Global warming potential (GWP) is a measure of how much heat a greenhouse gas traps in the atmosphere over a specific time period, relative to carbon dioxide (). It is expressed as a multiple of warming caused by the same mass of carbon dioxide ( ...
s (GWPs) of about 100 to 10,000; sulfur hexafluoride has a value of around 20,000. An outlier is HFO-1234yf which is a new type of refrigerant called a Hydrofluoroolefin
Hydrofluoroolefins (HFOs) are Saturated and unsaturated compounds, unsaturated organic compounds composed of hydrogen, fluorine and carbon. These organofluorine compounds are of interest as refrigerants. Unlike traditional hydrofluorocarbons (HFCs ...
(HFO) and has attracted global demand due to its GWP of less than 1 compared to 1,430 for the current refrigerant standard HFC-134a.
Biopersistence
 Organofluorines exhibit biopersistence due to the strength of the carbon–fluorine bond. Perfluoroalkyl acids (PFAAs), which are sparingly water-soluble owing to their acidic functional groups, are noted
Organofluorines exhibit biopersistence due to the strength of the carbon–fluorine bond. Perfluoroalkyl acids (PFAAs), which are sparingly water-soluble owing to their acidic functional groups, are noted persistent organic pollutant
Persistent organic pollutants (POPs) are organic compounds that are resistant to degradation through chemical, biological, and photolytic processes. They are toxic and adversely affect human health and the environment around the world. Because ...
s; perfluorooctanesulfonic acid
Perfluorooctanesulfonic acid (PFOS) (conjugate acid, conjugate base perfluorooctanesulfonate) is a chemical compound having an eight-carbon fluorocarbon chain and a sulfonic acid functional group, and thus it is a perfluorosulfonic acid and a Per ...
(PFOS) and perfluorooctanoic acid
Perfluorooctanoic acid (PFOA; conjugate acid, conjugate base perfluorooctanoate; also known colloquially as C8, from its chemical formula C8HF15O2) is a perfluorinated carboxylic acid produced and used worldwide as an industrial surfactant in ch ...
(PFOA) are most often researched. PFAAs have been found in trace quantities worldwide from polar bears to humans, with PFOS and PFOA known to reside in breast milk and the blood of newborn babies. A 2013 review showed a slight correlation between groundwater and soil PFAA levels and human activity; there was no clear pattern of one chemical dominating, and higher amounts of PFOS were correlated to higher amounts of PFOA.. In the body, PFAAs bind to proteins such as serum albumin
Serum albumin, often referred to simply as blood albumin, is an albumin (a type of globular protein) found in vertebrate blood. Human serum albumin is encoded by the ''ALB'' gene. Other mammalian forms, such as bovine serum albumin, are chem ...
; they tend to concentrate within humans in the liver and blood before excretion through the kidneys. Dwell time in the body varies greatly by species, with half-lives of days in rodents, and years in humans.... High doses of PFOS and PFOA cause cancer and death in newborn rodents but human studies have not established an effect at current exposure levels.
See also
* Argon fluoride laser *Electrophilic fluorination Electrophilic fluorination is the combination of a carbon-centered nucleophile with an electrophilic source of fluorine to afford organofluorine compounds. Although elemental fluorine and reagents incorporating an oxygen-fluorine bond can be used fo ...
* Fluoride selective electrode, which measures fluoride concentration
* Fluorine absorption dating
Fluorine absorption dating is a method used to determine the amount of time an object has been underground.
Fluorine absorption dating is based on the fact that groundwater contains fluoride ions. Items such as bone that are buried in soil will ...
* Fluorous chemistry, a process used to separate reagents from organic solvents
* Krypton fluoride laser
A krypton fluoride laser (KrF laser) is a particular type of excimer laser, which is sometimes (more correctly) called an exciplex laser. With its 248 nanometer wavelength, it is a deep ultraviolet laser which is commonly used in the production of ...
* Radical fluorination
Notes
Sources
Citations
Indexed references
* * * * * * * * * * *. * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * *< * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * * *External links
* {{authority control Chemical elements Halogens Reactive nonmetals Diatomic nonmetals Fluorinating agents Oxidizing agents Industrial gases Gases with color