Spin (physics) on:
[Wikipedia]
[Google]
[Amazon]
Spin is a
 Particles with spin can possess a magnetic dipole moment, just like a rotating electrically charged body in classical electrodynamics. These magnetic moments can be experimentally observed in several ways, e.g. by the deflection of particles by inhomogeneous
Particles with spin can possess a magnetic dipole moment, just like a rotating electrically charged body in classical electrodynamics. These magnetic moments can be experimentally observed in several ways, e.g. by the deflection of particles by inhomogeneous
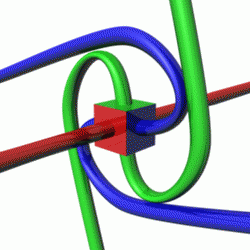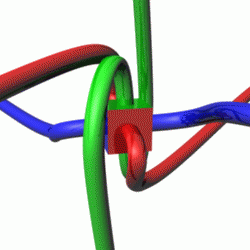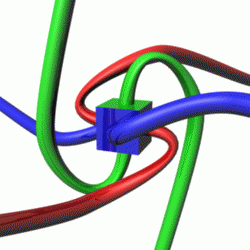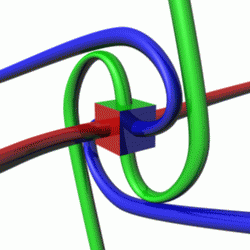 For a given quantum state, one could think of a spin vector whose components are the expectation values of the spin components along each axis, i.e.,
, For spin they are
For a given quantum state, one could think of a spin vector whose components are the expectation values of the spin components along each axis, i.e.,
, For spin they are
\begin
S_x = \frac\hbar2
\begin
0 &\sqrt &0 &0\\
\sqrt &0 &2 &0\\
0 &2 &0 &\sqrt\\
0 &0 &\sqrt &0
\end, \!\!\! &
\left, \frac, \frac\right\rangle_x =\!\!\! & \frac \begin 1 \\\\\\ 1 \end, \!\!\! &
\left, \frac, \frac\right\rangle_x =\!\!\! & \frac \begin\\ -1 \\ 1 \\\end, \!\!\! &
\left, \frac, \frac\right\rangle_x =\!\!\! & \frac \begin\\ -1 \\ -1 \\\end, \!\!\! &
\left, \frac, \frac\right\rangle_x =\!\!\! & \frac \begin -1 \\\\\\ 1 \end \\
S_y = \frac\hbar2
\begin
0 &-i\sqrt &0 &0\\
i\sqrt &0 &-2i &0\\
0 &2i &0 &-i\sqrt\\
0 &0 &i\sqrt &0
\end, \!\!\! &
\left, \frac, \frac\right\rangle_y =\!\!\! & \frac \begin\\\\\\ 1 \end, \!\!\! &
\left, \frac, \frac\right\rangle_y =\!\!\! & \frac \begin\\ 1 \\\\\end, \!\!\! &
\left, \frac, \frac\right\rangle_y =\!\!\! & \frac \begin\\ 1 \\\\\end, \!\!\! &
\left, \frac, \frac\right\rangle_y =\!\!\! & \frac \begin\\\\\\ 1 \end \\
S_z = \frac\hbar2
\begin
3 &0 &0 &0\\
0 &1 &0 &0\\
0 &0 &-1 &0\\
0 &0 &0 &-3
\end, \!\!\! &
\left, \frac, \frac\right\rangle_z =\!\!\! & \begin 1 \\ 0 \\ 0 \\ 0 \end, \!\!\! &
\left, \frac, \frac\right\rangle_z =\!\!\! & \begin 0 \\ 1 \\ 0 \\ 0 \end, \!\!\! &
\left, \frac, \frac\right\rangle_z =\!\!\! & \begin 0 \\ 0 \\ 1 \\ 0 \end, \!\!\! &
\left, \frac, \frac\right\rangle_z =\!\!\! & \begin 0 \\ 0 \\ 0 \\ 1 \end \\
\end
, For spin they are
\begin
\boldsymbol_x &= \frac
\begin
0 &\sqrt &0 &0 &0 &0 \\
\sqrt &0 &2\sqrt &0 &0 &0 \\
0 &2\sqrt &0 &3 &0 &0 \\
0 &0 &3 &0 &2\sqrt &0 \\
0 &0 &0 &2\sqrt &0 &\sqrt \\
0 &0 &0 &0 &\sqrt &0
\end, \\
\boldsymbol_y &= \frac
\begin
0 &-i\sqrt &0 &0 &0 &0 \\
i\sqrt &0 &-2i\sqrt &0 &0 &0 \\
0 &2i\sqrt &0 &-3i &0 &0 \\
0 &0 &3i &0 &-2i\sqrt &0 \\
0 &0 &0 &2i\sqrt &0 &-i\sqrt \\
0 &0 &0 &0 &i\sqrt &0
\end, \\
\boldsymbol_z &= \frac
\begin
5 &0 &0 &0 &0 &0 \\
0 &3 &0 &0 &0 &0 \\
0 &0 &1 &0 &0 &0 \\
0 &0 &0 &-1 &0 &0 \\
0 &0 &0 &0 &-3 &0 \\
0 &0 &0 &0 &0 &-5
\end.
\end
, The generalization of these matrices for arbitrary spin is
\begin
\left(S_x\right)_ & = \frac \left(\delta_ + \delta_\right) \sqrt, \\
\left(S_y\right)_ & = \frac \left(\delta_ - \delta_\right) \sqrt, \\
\left(S_z\right)_ & = \hbar (s + 1 - a) \delta_ = \hbar (s + 1 - b) \delta_,
\end
where indices a, b are integer numbers such that
1 \le a \le 2s + 1, \quad 1 \le b \le 2s + 1.
Also useful in the
\hat(\theta, \hat) = e^ =
I \cos \frac + i \left(\hat \cdot \boldsymbol\right) \sin \frac
for higher spins is tractable, but less simple.
 Spin was first discovered in the context of the emission spectrum of alkali metals. In 1924, Wolfgang Pauli introduced what he called a "two-valuedness not describable classically" associated with the electron in the outermost shell. This allowed him to formulate the Pauli exclusion principle, stating that no two electrons can have the same quantum state in the same quantum system.
The physical interpretation of Pauli's "degree of freedom" was initially unknown. Ralph Kronig, one of Landé's assistants, suggested in early 1925 that it was produced by the self-rotation of the electron. When Pauli heard about the idea, he criticized it severely, noting that the electron's hypothetical surface would have to be moving faster than the speed of light in order for it to rotate quickly enough to produce the necessary angular momentum. This would violate the theory of relativity. Largely due to Pauli's criticism, Kronig decided not to publish his idea.
In the autumn of 1925, the same thought came to Dutch physicists George Uhlenbeck and Samuel Goudsmit at Leiden University. Under the advice of Paul Ehrenfest, they published their results. It met a favorable response, especially after Llewellyn Thomas managed to resolve a factor-of-two discrepancy between experimental results and Uhlenbeck and Goudsmit's calculations (and Kronig's unpublished results). This discrepancy was due to the orientation of the electron's tangent frame, in addition to its position.
Mathematically speaking, a fiber bundle description is needed. The tangent bundle effect is additive and relativistic; that is, it vanishes if goes to infinity. It is one half of the value obtained without regard for the tangent-space orientation, but with opposite sign. Thus the combined effect differs from the latter by a factor two ( Thomas precession, known to Ludwik Silberstein in 1914).
Despite his initial objections, Pauli formalized the theory of spin in 1927, using the modern theory of
Spin was first discovered in the context of the emission spectrum of alkali metals. In 1924, Wolfgang Pauli introduced what he called a "two-valuedness not describable classically" associated with the electron in the outermost shell. This allowed him to formulate the Pauli exclusion principle, stating that no two electrons can have the same quantum state in the same quantum system.
The physical interpretation of Pauli's "degree of freedom" was initially unknown. Ralph Kronig, one of Landé's assistants, suggested in early 1925 that it was produced by the self-rotation of the electron. When Pauli heard about the idea, he criticized it severely, noting that the electron's hypothetical surface would have to be moving faster than the speed of light in order for it to rotate quickly enough to produce the necessary angular momentum. This would violate the theory of relativity. Largely due to Pauli's criticism, Kronig decided not to publish his idea.
In the autumn of 1925, the same thought came to Dutch physicists George Uhlenbeck and Samuel Goudsmit at Leiden University. Under the advice of Paul Ehrenfest, they published their results. It met a favorable response, especially after Llewellyn Thomas managed to resolve a factor-of-two discrepancy between experimental results and Uhlenbeck and Goudsmit's calculations (and Kronig's unpublished results). This discrepancy was due to the orientation of the electron's tangent frame, in addition to its position.
Mathematically speaking, a fiber bundle description is needed. The tangent bundle effect is additive and relativistic; that is, it vanishes if goes to infinity. It is one half of the value obtained without regard for the tangent-space orientation, but with opposite sign. Thus the combined effect differs from the latter by a factor two ( Thomas precession, known to Ludwik Silberstein in 1914).
Despite his initial objections, Pauli formalized the theory of spin in 1927, using the modern theory of
Goudsmit on the discovery of electron spin.
*''
Milestones in 'spin' since 1896.
ECE 495N Lecture 36: Spin
Online lecture by S. Datta {{Authority control Rotational symmetry Quantum field theory Physical quantities
conserved quantity
In mathematics, a conserved quantity of a dynamical system is a function of the dependent variables, the value of which remains constant along each trajectory of the system.
Not all systems have conserved quantities, and conserved quantities are ...
carried by elementary particles, and thus by composite particles ( hadrons) and atomic nuclei
The atomic nucleus is the small, dense region consisting of protons and neutrons at the center of an atom, discovered in 1911 by Ernest Rutherford based on the 1909 Geiger–Marsden gold foil experiment. After the discovery of the neutron in ...
.
Spin is one of two types of angular momentum in quantum mechanics, the other being ''orbital angular momentum''. The orbital angular momentum operator is the quantum-mechanical counterpart to the classical angular momentum of orbital revolution and appears when there is periodic structure to its wavefunction as the angle varies. For photons, spin is the quantum-mechanical counterpart of the polarization
Polarization or polarisation may refer to:
Mathematics
*Polarization of an Abelian variety, in the mathematics of complex manifolds
*Polarization of an algebraic form, a technique for expressing a homogeneous polynomial in a simpler fashion by ...
of light; for electrons, the spin has no classical counterpart.
The existence of electron spin angular momentum is inferred from experiments, such as the Stern–Gerlach experiment, in which silver atoms were observed to possess two possible discrete angular momenta despite having no orbital angular momentum. The existence of the electron spin can also be inferred theoretically from the spin–statistics theorem and from the Pauli exclusion principle—and vice versa, given the particular spin of the electron, one may derive the Pauli exclusion principle.
Spin is described mathematically as a vector for some particles such as photons, and as spinors and bispinors for other particles such as electrons. Spinors and bispinors behave similarly to vectors: they have definite magnitudes and change under rotations; however, they use an unconventional "direction". All elementary particles of a given kind have the same magnitude of spin angular momentum, though its direction may change. These are indicated by assigning the particle a spin quantum number.
The SI unit
The International System of Units, known by the international abbreviation SI in all languages and sometimes Pleonasm#Acronyms and initialisms, pleonastically as the SI system, is the modern form of the metric system and the world's most wid ...
of spin is the same as classical angular momentum (i.e., N· m· s, J·s, or kg·m2·s−1). In practice, spin is given as a dimensionless spin quantum number by dividing the spin angular momentum by the reduced Planck constant , which has the same dimensions as angular momentum, although this is not the full computation of this value. Very often, the "spin quantum number" is simply called "spin". The fact that it is a quantum number is implicit.
History
Wolfgang Pauli in 1924 was the first to propose a doubling of the number of available electron states due to a two-valued non-classical "hidden rotation". In 1925, George Uhlenbeck and Samuel Goudsmit at Leiden University suggested the simple physical interpretation of a particle spinning around its own axis, in the spirit of the old quantum theory ofBohr
Niels Henrik David Bohr (; 7 October 1885 – 18 November 1962) was a Danish physicist who made foundational contributions to understanding atomic structure and quantum theory, for which he received the Nobel Prize in Physics in 1922. B ...
and Sommerfeld. Ralph Kronig anticipated the Uhlenbeck–Goudsmit model in discussion with Hendrik Kramers several months earlier in Copenhagen, but did not publish. The mathematical theory was worked out in depth by Pauli in 1927. When Paul Dirac derived his relativistic quantum mechanics in 1928, electron spin was an essential part of it.
Quantum number
As the name suggests, spin was originally conceived as the rotation of a particle around some axis. While the question of whether elementary particles actually rotate is ambiguous (as they appear point-like), this picture is correct insofar as spin obeys the same mathematical laws as quantized angular momenta do; in particular, spin implies that the particle's phase changes with angle. On the other hand, spin has some peculiar properties that distinguish it from orbital angular momenta: * Spin quantum numbers may take half-integer values. * Although the direction of its spin can be changed, an elementary particle cannot be made to spin faster or slower. * The spin of a charged particle is associated with a magnetic dipole moment with a -factor differing from 1. This could occur classically only if the internal charge of the particle were distributed differently from its mass. The conventional definition of the spin quantum number is , where can be any non-negative integer. Hence the allowed values of are 0, , 1, , 2, etc. The value of for an elementary particle depends only on the type of particle and cannot be altered in any known way (in contrast to the ''spin direction'' described below). The spin angular momentum of any physical system is quantized. The allowed values of are where is the Planck constant, and is the reduced Planck constant. In contrast, orbital angular momentum can only take on integer values of ; i.e., even-numbered values of .Fermions and bosons
Those particles with half-integer spins, such as , , , are known as fermions, while those particles with integer spins, such as 0, 1, 2, are known as bosons. The two families of particles obey different rules and ''broadly'' have different roles in the world around us. A key distinction between the two families is that fermions obey the Pauli exclusion principle: that is, there cannot be two identical fermions simultaneously having the same quantum numbers (meaning, roughly, having the same position, velocity and spin direction). Fermions obey the rules of Fermi–Dirac statistics. In contrast, bosons obey the rules of Bose–Einstein statistics and have no such restriction, so they may "bunch together" in identical states. Also, composite particles can have spins different from their component particles. For example, a helium-4 atom in the ground state has spin 0 and behaves like a boson, even though the quarks and electrons which make it up are all fermions. This has some profound consequences: * Quarks and leptons (including electrons and neutrinos), which make up what is classically known asmatter
In classical physics and general chemistry, matter is any substance that has mass and takes up space by having volume. All everyday objects that can be touched are ultimately composed of atoms, which are made up of interacting subatomic par ...
, are all fermions with spin . The common idea that "matter takes up space" actually comes from the Pauli exclusion principle acting on these particles to prevent the fermions from being in the same quantum state. Further compaction would require electrons to occupy the same energy states, and therefore a kind of pressure
Pressure (symbol: ''p'' or ''P'') is the force applied perpendicular to the surface of an object per unit area over which that force is distributed. Gauge pressure (also spelled ''gage'' pressure)The preferred spelling varies by country a ...
(sometimes known as degeneracy pressure of electrons) acts to resist the fermions being overly close. Elementary fermions with other spins (, , etc.) are not known to exist.
* Elementary particles which are thought of as carrying forces are all bosons with spin 1. They include the photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are Massless particle, massless ...
, which carries the electromagnetic force, the gluon ( strong force), and the W and Z bosons ( weak force). The ability of bosons to occupy the same quantum state is used in the laser
A laser is a device that emits light through a process of optical amplification based on the stimulated emission of electromagnetic radiation. The word "laser" is an acronym for "light amplification by stimulated emission of radiation". The firs ...
, which aligns many photons having the same quantum number (the same direction and frequency), superfluid liquid helium
Liquid helium is a physical state of helium at very low temperatures at standard atmospheric pressures. Liquid helium may show superfluidity.
At standard pressure, the chemical element helium exists in a liquid form only at the extremely low t ...
resulting from helium-4 atoms being bosons, and superconductivity, where pairs of electrons (which individually are fermions) act as single composite bosons. Elementary bosons with other spins (0, 2, 3, etc.) were not historically known to exist, although they have received considerable theoretical treatment and are well established within their respective mainstream theories. In particular, theoreticians have proposed the graviton (predicted to exist by some quantum gravity theories) with spin 2, and the Higgs boson (explaining electroweak symmetry breaking) with spin 0. Since 2013, the Higgs boson with spin 0 has been considered proven to exist. It is the first scalar elementary particle (spin 0) known to exist in nature.
* Atomic nuclei have nuclear spin which may be either half-integer or integer, so that the nuclei may be either fermions or bosons.
Spin–statistics theorem
The spin–statistics theorem splits particles into two groups: bosons and fermions, where bosons obey Bose–Einstein statistics, and fermions obey Fermi–Dirac statistics (and therefore the Pauli exclusion principle). Specifically, the theory states that particles with an integer spin are bosons, while all other particles have half-integer spins and are fermions. As an example,electron
The electron (, or in nuclear reactions) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary partic ...
s have half-integer spin and are fermions that obey the Pauli exclusion principle, while photons have integer spin and do not. The theorem relies on both quantum mechanics and the theory of special relativity
In physics, the special theory of relativity, or special relativity for short, is a scientific theory regarding the relationship between space and time. In Albert Einstein's original treatment, the theory is based on two postulates:
# The law ...
, and this connection between spin and statistics has been called "one of the most important applications of the special relativity theory".
Relation to classical rotation
Since elementary particles are point-like, self-rotation is not well-defined for them. However, spin implies that the phase of the particle depends on the angle as , for rotation of angle ''θ'' around the axis parallel to the spin ''S''. This is equivalent to the quantum-mechanical interpretation of momentum as phase dependence in the position, and of orbital angular momentum as phase dependence in the angular position. Photon spin is the quantum-mechanical description of lightpolarization
Polarization or polarisation may refer to:
Mathematics
*Polarization of an Abelian variety, in the mathematics of complex manifolds
*Polarization of an algebraic form, a technique for expressing a homogeneous polynomial in a simpler fashion by ...
, where spin +1 and spin −1 represent two opposite directions of circular polarization. Thus, light of a defined circular polarization consists of photons with the same spin, either all +1 or all −1. Spin represents polarization for other vector bosons as well.
For fermions, the picture is less clear. Angular velocity is equal by Ehrenfest theorem to the derivative of the Hamiltonian to its conjugate momentum, which is the total angular momentum operator . Therefore, if the Hamiltonian ''H'' is dependent upon the spin ''S'', ''dH''/''dS'' is non-zero, and the spin causes angular velocity, and hence actual rotation, i.e. a change in the phase-angle relation over time. However, whether this holds for free electron is ambiguous, since for an electron, ''S''2 is constant, and therefore it is a matter of interpretation whether the Hamiltonian includes such a term. Nevertheless, spin appears in the Dirac equation, and thus the relativistic Hamiltonian of the electron, treated as a Dirac field
In quantum field theory, a fermionic field is a quantum field whose quanta are fermions; that is, they obey Fermi–Dirac statistics. Fermionic fields obey canonical anticommutation relations rather than the canonical commutation relations of boso ...
, can be interpreted as including a dependence in the spin ''S''. Under this interpretation, free electrons also self-rotate, with the Zitterbewegung effect understood as this rotation.
Magnetic moments
 Particles with spin can possess a magnetic dipole moment, just like a rotating electrically charged body in classical electrodynamics. These magnetic moments can be experimentally observed in several ways, e.g. by the deflection of particles by inhomogeneous
Particles with spin can possess a magnetic dipole moment, just like a rotating electrically charged body in classical electrodynamics. These magnetic moments can be experimentally observed in several ways, e.g. by the deflection of particles by inhomogeneous magnetic field
A magnetic field is a vector field that describes the magnetic influence on moving electric charges, electric currents, and magnetic materials. A moving charge in a magnetic field experiences a force perpendicular to its own velocity and t ...
s in a Stern–Gerlach experiment, or by measuring the magnetic fields generated by the particles themselves.
The intrinsic magnetic moment of a spin- particle with charge , mass , and spin angular momentum , is
:
where the dimensionless quantity is called the spin -factor. For exclusively orbital rotations it would be 1 (assuming that the mass and the charge occupy spheres of equal radius).
The electron, being a charged elementary particle, possesses a nonzero magnetic moment. One of the triumphs of the theory of quantum electrodynamics is its accurate prediction of the electron -factor, which has been experimentally determined to have the value , with the digits in parentheses denoting measurement uncertainty in the last two digits at one standard deviation. The value of 2 arises from the Dirac equation, a fundamental equation connecting the electron's spin with its electromagnetic properties, and the correction of ... arises from the electron's interaction with the surrounding electromagnetic field
An electromagnetic field (also EM field or EMF) is a classical (i.e. non-quantum) field produced by (stationary or moving) electric charges. It is the field described by classical electrodynamics (a classical field theory) and is the classica ...
, including its own field.
Composite particles also possess magnetic moments associated with their spin. In particular, the neutron
The neutron is a subatomic particle, symbol or , which has a neutral (not positive or negative) charge, and a mass slightly greater than that of a proton. Protons and neutrons constitute the nuclei of atoms. Since protons and neutrons behav ...
possesses a non-zero magnetic moment despite being electrically neutral. This fact was an early indication that the neutron is not an elementary particle. In fact, it is made up of quarks, which are electrically charged particles. The magnetic moment of the neutron comes from the spins of the individual quarks and their orbital motions.
Neutrino
A neutrino ( ; denoted by the Greek letter ) is a fermion (an elementary particle with spin of ) that interacts only via the weak interaction and gravity. The neutrino is so named because it is electrically neutral and because its rest mass ...
s are both elementary and electrically neutral. The minimally extended Standard Model
The Standard Model of particle physics is the theory describing three of the four known fundamental forces ( electromagnetic, weak and strong interactions - excluding gravity) in the universe and classifying all known elementary particles. I ...
that takes into account non-zero neutrino masses predicts neutrino magnetic moments of:
:
where the are the neutrino magnetic moments, are the neutrino masses, and is the Bohr magneton. New physics above the electroweak scale could, however, lead to significantly higher neutrino magnetic moments. It can be shown in a model-independent way that neutrino magnetic moments larger than about 10−14 are "unnatural" because they would also lead to large radiative contributions to the neutrino mass. Since the neutrino masses are known to be at most about 1 eV, the large radiative corrections would then have to be "fine-tuned" to cancel each other, to a large degree, and leave the neutrino mass small. The measurement of neutrino magnetic moments is an active area of research. Experimental results have put the neutrino magnetic moment at less than times the electron's magnetic moment.
On the other hand elementary particles with spin but without electric charge, such as a photon
A photon () is an elementary particle that is a quantum of the electromagnetic field, including electromagnetic radiation such as light and radio waves, and the force carrier for the electromagnetic force. Photons are Massless particle, massless ...
or a Z boson, do not have a magnetic moment.
Curie temperature and loss of alignment
In ordinary materials, the magnetic dipole moments of individual atoms produce magnetic fields that cancel one another, because each dipole points in a random direction, with the overall average being very near zero.Ferromagnet
Ferromagnetism is a property of certain materials (such as iron) which results in a large observed magnetic permeability, and in many cases a large magnetic coercivity allowing the material to form a permanent magnet. Ferromagnetic material ...
ic materials below their Curie temperature
In physics and materials science, the Curie temperature (''T''C), or Curie point, is the temperature above which certain materials lose their permanent magnetic properties, which can (in most cases) be replaced by induced magnetism. The Cu ...
, however, exhibit magnetic domains in which the atomic dipole moments spontaneously align locally, producing a macroscopic, non-zero magnetic field from the domain. These are the ordinary "magnets" with which we are all familiar.
In paramagnetic materials, the magnetic dipole moments of individual atoms will partially align with an externally applied magnetic field. In diamagnetic materials, on the other hand, the magnetic dipole moments of individual atoms align oppositely to any externally applied magnetic field, even if it requires energy to do so.
The study of the behavior of such " spin models" is a thriving area of research in condensed matter physics. For instance, the Ising model describes spins (dipoles) that have only two possible states, up and down, whereas in the Heisenberg model the spin vector is allowed to point in any direction. These models have many interesting properties, which have led to interesting results in the theory of phase transitions.
Direction
Spin projection quantum number and multiplicity
In classical mechanics, the angular momentum of a particle possesses not only a magnitude (how fast the body is rotating), but also a direction (either up or down on the axis of rotation of the particle). Quantum-mechanical spin also contains information about direction, but in a more subtle form. Quantum mechanics states that the component of angular momentum for a spin-''s'' particle measured along any direction can only take on the values : where is the spin component along the -th axis (either , , or ), is the spin projection quantum number along the -th axis, and is the principal spin quantum number (discussed in the previous section). Conventionally the direction chosen is the axis: : where is the spin component along the axis, is the spin projection quantum number along the axis. One can see that there are possible values of . The number "" is the multiplicity of the spin system. For example, there are only two possible values for a spin- particle: and . These correspond to quantum states in which the spin component is pointing in the +''z'' or −''z'' directions respectively, and are often referred to as "spin up" and "spin down". For a spin- particle, like a delta baryon, the possible values are +, +, −, −.Vector
 For a given quantum state, one could think of a spin vector whose components are the expectation values of the spin components along each axis, i.e.,
, For spin they are
For a given quantum state, one could think of a spin vector whose components are the expectation values of the spin components along each axis, i.e.,
, For spin they are
quantum mechanics
Quantum mechanics is a fundamental theory in physics that provides a description of the physical properties of nature at the scale of atoms and subatomic particles. It is the foundation of all quantum physics including quantum chemistry, q ...
of multiparticle systems, the general Pauli group
In physics and mathematics, the Pauli group G_1 on 1 qubit is the 16-element matrix group consisting of the 2 × 2 identity matrix I and all of the Pauli matrices
:X = \sigma_1 =
\begin
0&1\\
1&0
\end,\quad
Y = \sigma_2 =
\beg ...
is defined to consist of all -fold tensor
In mathematics, a tensor is an algebraic object that describes a multilinear relationship between sets of algebraic objects related to a vector space. Tensors may map between different objects such as vectors, scalars, and even other tens ...
products of Pauli matrices.
The analog formula of Euler's formula in terms of the Pauli matrices
: Parity
In tables of the spin quantum number for nuclei or particles, the spin is often followed by a "+" or "−". This refers to theparity
Parity may refer to:
* Parity (computing)
** Parity bit in computing, sets the parity of data for the purpose of error detection
** Parity flag in computing, indicates if the number of set bits is odd or even in the binary representation of the r ...
with "+" for even parity (wave function unchanged by spatial inversion) and "−" for odd parity (wave function negated by spatial inversion). For example, see the isotopes of bismuth, in which the list of isotopes includes the column nuclear spin and parity. For Bi-209, the only stable isotope, the entry 9/2– means that the nuclear spin is 9/2 and the parity is odd.
Applications
Spin has important theoretical implications and practical applications. Well-established ''direct'' applications of spin include: * Nuclear magnetic resonance (NMR) spectroscopy in chemistry; * Electron spin resonance (ESR or EPR) spectroscopy in chemistry and physics; * Magnetic resonance imaging (MRI) in medicine, a type of applied NMR, which relies on proton spin density; * Giant magnetoresistive (GMR) drive-head technology in modern hard disks. Electron spin plays an important role inmagnetism
Magnetism is the class of physical attributes that are mediated by a magnetic field, which refers to the capacity to induce attractive and repulsive phenomena in other entities. Electric currents and the magnetic moments of elementary particle ...
, with applications for instance in computer memories. The manipulation of ''nuclear spin'' by radio-frequency waves ( nuclear magnetic resonance) is important in chemical spectroscopy and medical imaging.
Spin–orbit coupling leads to the fine structure of atomic spectra, which is used in atomic clock
An atomic clock is a clock that measures time by monitoring the resonant frequency of atoms. It is based on atoms having different energy levels. Electron states in an atom are associated with different energy levels, and in transitions betw ...
s and in the modern definition of the second. Precise measurements of the -factor of the electron have played an important role in the development and verification of quantum electrodynamics. ''Photon spin'' is associated with the polarization
Polarization or polarisation may refer to:
Mathematics
*Polarization of an Abelian variety, in the mathematics of complex manifolds
*Polarization of an algebraic form, a technique for expressing a homogeneous polynomial in a simpler fashion by ...
of light ( photon polarization).
An emerging application of spin is as a binary information carrier in spin transistors. The original concept, proposed in 1990, is known as Datta–Das spin transistor. Electronics based on spin transistors are referred to as spintronics. The manipulation of spin in dilute magnetic semiconductor materials, such as metal-doped ZnO or TiO2 imparts a further degree of freedom and has the potential to facilitate the fabrication of more efficient electronics.
There are many ''indirect'' applications and manifestations of spin and the associated Pauli exclusion principle, starting with the periodic table
The periodic table, also known as the periodic table of the (chemical) elements, is a rows and columns arrangement of the chemical elements. It is widely used in chemistry, physics, and other sciences, and is generally seen as an icon of ...
of chemistry.
History
 Spin was first discovered in the context of the emission spectrum of alkali metals. In 1924, Wolfgang Pauli introduced what he called a "two-valuedness not describable classically" associated with the electron in the outermost shell. This allowed him to formulate the Pauli exclusion principle, stating that no two electrons can have the same quantum state in the same quantum system.
The physical interpretation of Pauli's "degree of freedom" was initially unknown. Ralph Kronig, one of Landé's assistants, suggested in early 1925 that it was produced by the self-rotation of the electron. When Pauli heard about the idea, he criticized it severely, noting that the electron's hypothetical surface would have to be moving faster than the speed of light in order for it to rotate quickly enough to produce the necessary angular momentum. This would violate the theory of relativity. Largely due to Pauli's criticism, Kronig decided not to publish his idea.
In the autumn of 1925, the same thought came to Dutch physicists George Uhlenbeck and Samuel Goudsmit at Leiden University. Under the advice of Paul Ehrenfest, they published their results. It met a favorable response, especially after Llewellyn Thomas managed to resolve a factor-of-two discrepancy between experimental results and Uhlenbeck and Goudsmit's calculations (and Kronig's unpublished results). This discrepancy was due to the orientation of the electron's tangent frame, in addition to its position.
Mathematically speaking, a fiber bundle description is needed. The tangent bundle effect is additive and relativistic; that is, it vanishes if goes to infinity. It is one half of the value obtained without regard for the tangent-space orientation, but with opposite sign. Thus the combined effect differs from the latter by a factor two ( Thomas precession, known to Ludwik Silberstein in 1914).
Despite his initial objections, Pauli formalized the theory of spin in 1927, using the modern theory of
Spin was first discovered in the context of the emission spectrum of alkali metals. In 1924, Wolfgang Pauli introduced what he called a "two-valuedness not describable classically" associated with the electron in the outermost shell. This allowed him to formulate the Pauli exclusion principle, stating that no two electrons can have the same quantum state in the same quantum system.
The physical interpretation of Pauli's "degree of freedom" was initially unknown. Ralph Kronig, one of Landé's assistants, suggested in early 1925 that it was produced by the self-rotation of the electron. When Pauli heard about the idea, he criticized it severely, noting that the electron's hypothetical surface would have to be moving faster than the speed of light in order for it to rotate quickly enough to produce the necessary angular momentum. This would violate the theory of relativity. Largely due to Pauli's criticism, Kronig decided not to publish his idea.
In the autumn of 1925, the same thought came to Dutch physicists George Uhlenbeck and Samuel Goudsmit at Leiden University. Under the advice of Paul Ehrenfest, they published their results. It met a favorable response, especially after Llewellyn Thomas managed to resolve a factor-of-two discrepancy between experimental results and Uhlenbeck and Goudsmit's calculations (and Kronig's unpublished results). This discrepancy was due to the orientation of the electron's tangent frame, in addition to its position.
Mathematically speaking, a fiber bundle description is needed. The tangent bundle effect is additive and relativistic; that is, it vanishes if goes to infinity. It is one half of the value obtained without regard for the tangent-space orientation, but with opposite sign. Thus the combined effect differs from the latter by a factor two ( Thomas precession, known to Ludwik Silberstein in 1914).
Despite his initial objections, Pauli formalized the theory of spin in 1927, using the modern theory of quantum mechanics
Quantum mechanics is a fundamental theory in physics that provides a description of the physical properties of nature at the scale of atoms and subatomic particles. It is the foundation of all quantum physics including quantum chemistry, q ...
invented by Schrödinger and Heisenberg. He pioneered the use of Pauli matrices as a representation
Representation may refer to:
Law and politics
*Representation (politics), political activities undertaken by elected representatives, as well as other theories
** Representative democracy, type of democracy in which elected officials represent a ...
of the spin operators and introduced a two-component spinor wave-function. Uhlenbeck and Goudsmit treated spin as arising from classical rotation, while Pauli emphasized, that spin is non-classical and intrinsic property.
Pauli's theory of spin was non-relativistic. However, in 1928, Paul Dirac published the Dirac equation, which described the relativistic electron
The electron (, or in nuclear reactions) is a subatomic particle with a negative one elementary electric charge. Electrons belong to the first generation of the lepton particle family,
and are generally thought to be elementary partic ...
. In the Dirac equation, a four-component spinor (known as a " Dirac spinor") was used for the electron wave-function. Relativistic spin explained gyromagnetic anomaly, which was (in retrospect) first observed by Samuel Jackson Barnett in 1914 (see Einstein–de Haas effect). In 1940, Pauli proved the '' spin–statistics theorem'', which states that fermions have half-integer spin, and bosons have integer spin.
In retrospect, the first direct experimental evidence of the electron spin was the Stern–Gerlach experiment of 1922. However, the correct explanation of this experiment was only given in 1927.
See also
* Chirality (physics) * Dynamic nuclear polarisation * Helicity (particle physics) * Holstein–Primakoff transformation * Kramers theorem * Pauli equation * Pauli–Lubanski pseudovector * Rarita–Schwinger equation * Representation theory of SU(2) * Spin angular momentum of light *Spin engineering
Spin engineering describes the control and manipulation of quantum spin systems to develop devices and materials. This includes the use of the spin degrees of freedom as a probe for spin based phenomena.
Because of the basic importance of quantum ...
* Spin-flip
* Spin isomers of hydrogen
* Spin–orbit interaction
* Spin tensor
* Spin wave
* Yrast
''Yrast'' ( , ) is a technical term in nuclear physics that refers to a state of a nucleus with a minimum of energy (when it is least excited) for a given angular momentum. ''Yr'' is a Swedish adjective sharing the same root as the English ''whirl' ...
References
Further reading
* * * * * * * * *Sin-Itiro Tomonaga, The Story of Spin, 1997External links
*Goudsmit on the discovery of electron spin.
*''
Nature
Nature, in the broadest sense, is the physical world or universe. "Nature" can refer to the phenomena of the physical world, and also to life in general. The study of nature is a large, if not the only, part of science. Although humans ar ...
'':Milestones in 'spin' since 1896.
ECE 495N Lecture 36: Spin
Online lecture by S. Datta {{Authority control Rotational symmetry Quantum field theory Physical quantities