Carbenium ion on:
[Wikipedia]
[Google]
[Amazon]
 A carbenium ion is a positive ion with the structure RR′R″C+, that is, a
A carbenium ion is a positive ion with the structure RR′R″C+, that is, a
 The tropylium ion is an
The tropylium ion is an
 An arenium ion is a cyclohexadienyl cation that appears as a reactive intermediate in
An arenium ion is a cyclohexadienyl cation that appears as a reactive intermediate in 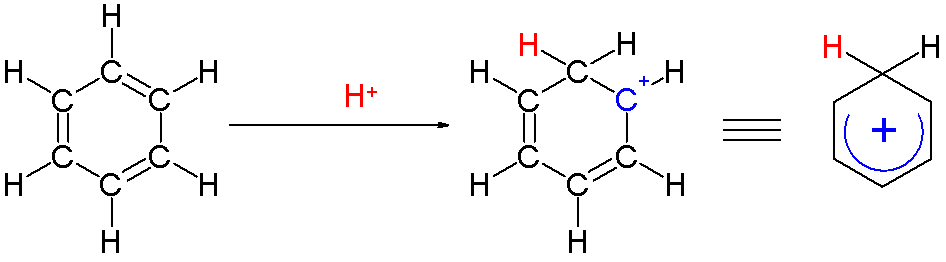 Two hydrogen atoms bonded to one carbon lie in a plane perpendicular to the benzene ring. The arenium ion is no longer an aromatic species; however it is relatively stable due to delocalization: the positive charge is delocalized over 5 carbon atoms via the π system, as depicted on the following
Two hydrogen atoms bonded to one carbon lie in a plane perpendicular to the benzene ring. The arenium ion is no longer an aromatic species; however it is relatively stable due to delocalization: the positive charge is delocalized over 5 carbon atoms via the π system, as depicted on the following  Another contribution to the stability of arenium ions is the energy gain resulting from the strong bond between the benzene and the complexed electrophile.
The smallest arenium ion is protonated
Another contribution to the stability of arenium ions is the energy gain resulting from the strong bond between the benzene and the complexed electrophile.
The smallest arenium ion is protonated
chemical species
A chemical species is a chemical substance or ensemble composed of chemically identical molecular entities that can explore the same set of molecular energy levels on a characteristic or delineated time scale. These energy levels determine the wa ...
with a trivalent
In chemistry, the valence (US spelling) or valency (British spelling) of an element is the measure of its combining capacity with other atoms when it forms chemical compounds or molecules.
Description
The combining capacity, or affinity of an ...
carbon that bears a +1 formal charge.
In older literature the name carbonium ion was used for this class, but now it refers exclusively to another family of carbocations, the carbonium ions, where the charged carbon is pentavalent. The current definitions were proposed by the chemist George Andrew Olah in 1972, and are now widely accepted.
Carbenium ions are generally highly reactive due to having an incomplete octet
Octet may refer to:
Music
* Octet (music), ensemble consisting of eight instruments or voices, or composition written for such an ensemble
** String octet, a piece of music written for eight string instruments
*** Octet (Mendelssohn), 1825 compos ...
of electrons; however, certain carbenium ions, such as the tropylium ion, are relatively stable due to the positive charge being delocalised between the carbon atoms.
Nomenclature
Carbenium ions are classified as primary, secondary, ortertiary
Tertiary ( ) is a widely used but obsolete term for the geologic period from 66 million to 2.6 million years ago.
The period began with the demise of the non-avian dinosaurs in the Cretaceous–Paleogene extinction event, at the start ...
depending on whether the number of carbon atoms bonded to the ionized carbon is 1, 2, or 3. (Ions with zero carbons attached to the ionized carbon, such as methenium
In organic chemistry, methenium (also called methylium, carbenium, methyl cation, or protonated methylene) is a cation with the formula . It can be viewed as a methylene radical (:) with an added proton (), or as a methyl radical (•) with on ...
, , are usually included in the primary class).
Reactivity
Stability typically increases with the number of alkyl groups bonded to the charge-bearing carbon. Tertiary carbocations are more stable (and form more readily) than secondary carbocations, because they are stabilized byhyperconjugation
In organic chemistry, hyperconjugation (σ-conjugation or no-bond resonance) refers to the delocalization of electrons with the participation of bonds of primarily σ-character. Usually, hyperconjugation involves the interaction of the electron ...
. Primary carbocations are highly unstable. Therefore, reactions such as the SN1 reaction and the E1 elimination reaction
An elimination reaction is a type of organic reaction in which two substituents are removed from a molecule in either a one- or two-step mechanism. The one-step mechanism is known as the E2 reaction, and the two-step mechanism is known as the E1 r ...
normally do not occur if a primary carbenium would be formed.
However, a carbon doubly bonded with the ionized carbon can stabilize the ion by resonance
Resonance describes the phenomenon of increased amplitude that occurs when the frequency of an applied Periodic function, periodic force (or a Fourier analysis, Fourier component of it) is equal or close to a natural frequency of the system ...
. Such cations as the '' allyl'' cation, , and the ''benzyl
In organic chemistry, benzyl is the substituent or molecular fragment possessing the structure . Benzyl features a benzene ring () attached to a methylene group () group.
Nomenclature
In IUPAC nomenclature, the prefix benzyl refers to a substi ...
'' cation, , are more stable than most other carbocations. Molecules which can form allyl or benzyl carbeniums are especially reactive. Carbenium ions can also be stabilized by heteroatom
In chemistry, a heteroatom () is, strictly, any atom that is not carbon or hydrogen.
Organic chemistry
In practice, the term is usually used more specifically to indicate that non-carbon atoms have replaced carbon in the backbone of the molecula ...
s.Hansjörg Grützmacher, Christina M. Marchand (1997), "Heteroatom stabilized carbenium ions", ''Coord. Chem. Rev.'', 163, 287–344.
Carbenium ions may undergo rearrangement reactions from less stable structures to equally stable or more stable ones with rate constants in excess of 109 s−1. This fact complicates synthetic pathways to many compounds. For example, when pentan-3-ol is heated with aqueous HCl, the initially formed 3-pentyl carbocation rearranges to a statistical mixture of the 3-pentyl and 2-pentyl. These cations react with chloride ion to produce 3-chloropentane and 2-chloropentane in a ratio of approximately 1:2.
Types of carbenium ions
Alkylium ions
Carbenium ions can be prepared directly from alkanes by removing ahydride
In chemistry, a hydride is formally the anion of hydrogen( H−). The term is applied loosely. At one extreme, all compounds containing covalently bound H atoms are called hydrides: water (H2O) is a hydride of oxygen, ammonia is a hydride ...
anion, , with a strong acid. For example, magic acid, a mixture of antimony pentafluoride () and fluorosulfuric acid (), turns isobutane into the trimethylcarbenium cation, .George A. Olah and Joachim Lukas (1967), "Stable Carbonium Ions. XLVII. Alkylcarbonium ion formation from alkanes via hydride (alkide) ion abstraction in fluorosulfonic acid-antimony pentafluoride-sulfuryl chlorofluoride solution". ''J. Am. Chem. Soc.'' 89 (18), 4739–4744
Aromatic carbenium ions
 The tropylium ion is an
The tropylium ion is an aromatic
In chemistry, aromaticity is a chemical property of cyclic ( ring-shaped), ''typically'' planar (flat) molecular structures with pi bonds in resonance (those containing delocalized electrons) that gives increased stability compared to satur ...
species with the formula . Its name derives from the molecule tropine
Tropine is a derivative of tropane containing a hydroxyl group at the third carbon. It is also called 3-tropanol.
Tropine is a central building block of many chemicals active in the nervous system, including tropane alkaloids. Some of these comp ...
(itself named for the molecule atropine). Salts of the tropylium cation can be stable, e.g. tropylium tetrafluoroborate. It can be made from cycloheptatriene
Cycloheptatriene (CHT) is an organic compound with the formula C7H8. It is a closed ring of seven carbon atoms joined by three double bonds (as the name implies) and four single bonds. This colourless liquid has been of recurring theoretical inter ...
(tropylidene) and bromine
Bromine is a chemical element with the symbol Br and atomic number 35. It is the third-lightest element in group 17 of the periodic table ( halogens) and is a volatile red-brown liquid at room temperature that evaporates readily to form a simi ...
or phosphorus pentachloride
Phosphorus pentachloride is the chemical compound with the formula PCl5. It is one of the most important phosphorus chlorides, others being PCl3 and POCl3. PCl5 finds use as a chlorinating reagent. It is a colourless, water-sensitive and moist ...
It is a planar, cyclic, heptagon
In geometry, a heptagon or septagon is a seven-sided polygon or 7-gon.
The heptagon is sometimes referred to as the septagon, using "sept-" (an elision of ''septua-'', a Latin-derived numerical prefix, rather than '' hepta-'', a Greek-derived nu ...
al ion; it also has 6 π-electrons (4''n'' + 2, where ''n'' = 1), which fulfills Hückel's rule
In organic chemistry, Hückel's rule predicts that a planar ring molecule will have aromatic properties if it has 4''n'' + 2 π electrons, where ''n'' is a non-negative integer. The quantum mechanical basis for its formulation was ...
of aromaticity. It can coordinate as a ligand
In coordination chemistry, a ligand is an ion or molecule ( functional group) that binds to a central metal atom to form a coordination complex. The bonding with the metal generally involves formal donation of one or more of the ligand's elec ...
to metal
A metal (from Greek μέταλλον ''métallon'', "mine, quarry, metal") is a material that, when freshly prepared, polished, or fractured, shows a lustrous appearance, and conducts electricity and heat relatively well. Metals are typicall ...
atoms.
The structure shown is a composite of seven resonance contributors in which each carbon carries part of the positive charge.
In 1891 G. Merling obtained a water-soluble salt from a reaction of cycloheptatriene and bromine. The structure was elucidated by Eggers Doering and Knox in 1954.
Another aromatic carbenium ion is the cyclopropenyl or cyclopropenium ion, , obtained by Ronald Breslow and John T. Groves in 1970. Though less stable than the tropylium cation, this carbenium ion can also form salts at room temperature. Solutions of such salts were found by Breslow and Groves to have spectroscopic and chemical properties matching expectations for an aromatic carbenium ion.
Triphenylmethyl (trityl) cation
The triphenylcarbenium or triphenylmethyl cation, , is especially stable because the positive charge can be distributed among 10 of the carbon atoms (the 3 carbon atoms in the ''ortho'' and ''para'' positions of each of the three phenyl groups, plus the central carbon atom). It exists in the compounds triphenylmethyl hexafluorophosphate , triphenylmethyl tetrafluoroborate , and triphenylmethyl perchlorate .N. C. Deno, J. J. Jaruzelski, and Alan Schriesheim (1955) "Carbonium ions. I. An acidity function (''C''0) derived from arylcarbonium ion equilibria." ''J. Am. Chem. Soc.'', 77 (11), 3044–3051. Its derivatives include thetriarylmethane dye
Triarylmethane dyes are synthetic organic compounds containing triphenylmethane backbones. As dyes, these compounds are intensely colored. They are produced industrially as dyes.
Families
Triarylmethane dyes can be grouped into families accordin ...
s.
Arenium ions
 An arenium ion is a cyclohexadienyl cation that appears as a reactive intermediate in
An arenium ion is a cyclohexadienyl cation that appears as a reactive intermediate in electrophilic aromatic substitution
Electrophilic aromatic substitution is an organic reaction in which an atom that is attached to an aromatic system (usually hydrogen) is replaced by an electrophile. Some of the most important electrophilic aromatic substitutions are aromatic n ...
. For historic reasons this complex is also called a ''Wheland intermediate'', or a ''σ-complex''.
: Two hydrogen atoms bonded to one carbon lie in a plane perpendicular to the benzene ring. The arenium ion is no longer an aromatic species; however it is relatively stable due to delocalization: the positive charge is delocalized over 5 carbon atoms via the π system, as depicted on the following
Two hydrogen atoms bonded to one carbon lie in a plane perpendicular to the benzene ring. The arenium ion is no longer an aromatic species; however it is relatively stable due to delocalization: the positive charge is delocalized over 5 carbon atoms via the π system, as depicted on the following resonance structures
In chemistry, resonance, also called mesomerism, is a way of describing bonding in certain molecules or polyatomic ions by the combination of several contributing structures (or ''forms'', also variously known as ''resonance structures'' or '' ...
:
: Another contribution to the stability of arenium ions is the energy gain resulting from the strong bond between the benzene and the complexed electrophile.
The smallest arenium ion is protonated
Another contribution to the stability of arenium ions is the energy gain resulting from the strong bond between the benzene and the complexed electrophile.
The smallest arenium ion is protonated benzene
Benzene is an organic chemical compound with the molecular formula C6H6. The benzene molecule is composed of six carbon atoms joined in a planar ring with one hydrogen atom attached to each. Because it contains only carbon and hydrogen atoms ...
, . The benzenium ion can be isolated as a stable compound when benzene is protonated by the carborane superacid
Carborane acids (X, Y, Z = H, Alk, F, Cl, Br, CF3) are a class of superacids, some of which are estimated to be at least one million times stronger than 100% pure sulfuric acid in terms of their Hammett acidity function values (''H''0 ≤ –18) ...
, H(CB11H(CH3)5Br6). The benzenium salt is crystalline with thermal stability up to 150 °C. Bond length
In molecular geometry, bond length or bond distance is defined as the average distance between nuclei of two bonded atoms in a molecule. It is a transferable property of a bond between atoms of fixed types, relatively independent of the rest of ...
s deduced from X-ray crystallography
X-ray crystallography is the experimental science determining the atomic and molecular structure of a crystal, in which the crystalline structure causes a beam of incident X-rays to diffract into many specific directions. By measuring the angles ...
are consistent with a cyclohexadienyl cation structure.
Acylium ions
An acylium ion is a cation with the formula RCO+. The structure is described as R−C≡O+ or R−=O. It is the synthetic and reactive equivalent of an acyl carbocation, but the actual structure has the oxygen and carbon linked by a triple bond. Such species are common reactive intermediates, for example, in the Friedel−Crafts acylations also in many other organic reactions such as the Hayashi rearrangement. Salts containing acylium ions can be generated by removal of the halide fromacyl halide
In organic chemistry, an acyl halide (also known as an acid halide) is a chemical compound derived from an oxoacid by replacing a hydroxyl group () with a halide group (, where X is a halogen).
If the acid is a carboxylic acid (), the compoun ...
s:
:RCOCl + SbCl5 → RCO+
The C–O distance in these cations is near 1.1 ångström
The angstromEntry "angstrom" in the Oxford online dictionary. Retrieved on 2019-03-02 from https://en.oxforddictionaries.com/definition/angstrom.Entry "angstrom" in the Merriam-Webster online dictionary. Retrieved on 2019-03-02 from https://www.m ...
s, even shorter than that in carbon monoxide
Carbon monoxide (chemical formula CO) is a colorless, poisonous, odorless, tasteless, flammable gas that is slightly less dense than air. Carbon monoxide consists of one carbon atom and one oxygen atom connected by a triple bond. It is the simple ...
. Acylium cations are characteristic fragments observed in EI-mass spectra
A mass spectrum is a histogram plot of intensity vs. ''mass-to-charge ratio'' (''m/z'') in a chemical sample, usually acquired using an instrument called a ''mass spectrometer''. Not all mass spectra of a given substance are the same; for example ...
of ketones.
See also
*Borenium ion
In chemistry, a boranylium ion is an inorganic cation with the chemical formula , where R represents a non-specific substituent. Being electron-deficient, boranylium ions form adducts with Lewis bases. Boranylium ions have historical names that de ...
* Nitrenium ion
A nitrenium ion (also called: aminylium ion or imidonium ion (obsolete)) in organic chemistry is a reactive intermediate based on nitrogen with both an electron lone pair and a positive charge and with two substituents (). Nitrenium ions are isoel ...
References
{{reflist Cations Reactive intermediates Carbocations