|
Ligand Cone Angle
In coordination chemistry, the ligand cone angle (a common example being the Tolman cone angle or ''θ'') is a measure of the steric bulk of a ligand in a transition metal coordination complex. It is defined as the solid angle formed with the metal at the vertex and the outermost edge of the van der Waals spheres of the ligand atoms at the perimeter of the cone (see figure). Tertiary phosphine ligands are commonly classified using this parameter, but the method can be applied to any ligand. The term ''cone angle'' was first introduced by Chadwick A. Tolman, a research chemist at DuPont. Tolman originally developed the method for phosphine ligands in nickel complexes, determining them from measurements of accurate physical models. Asymmetric cases The concept of cone angle is most easily visualized with symmetrical ligands, e.g. PR3. But the approach has been refined to include less symmetrical ligands of the type PRR′R″ as well as diphosphines. In such asymmetric cases, the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Trimethylphosphine
Trimethylphosphine is a neutral organophosphorus compound with the formula P(CH3)3, commonly abbreviated as PMe3. This colorless liquid has a strongly unpleasant odor, characteristic of alkylphosphines. The compound is a common ligand in coordination chemistry. Structure and bonding It is a pyramidal molecule with approximate ''C''3''v'' symmetry. The C–P–C bond angles are approximately 98.6°. The C–P–C bond angles are consistent with the notion that phosphorus predominantly uses the 3p orbitals for forming bonds and that there is little sp hybridization of the phosphorus atom. The latter is a common feature of the chemistry of phosphorus. As a result, the lone pair of trimethylphosphine has predominantly s-character as is the case for phosphine, PH3. PMe3 can be prepared by the treatment of triphenyl phosphite with methylmagnesium chloride: : 3 CH3MgCl + P(OC6H5)3 → P(CH3)3 + 3 C6H5OMgCl The synthesis is conducted in dibutyl ether, from which the more volatile PM ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Hydroformylation
Hydroformylation, also known as oxo synthesis or oxo process, is an industrial process for the production of aldehydes from alkenes. This chemical reaction entails the net addition of a formyl group (CHO) and a hydrogen atom to a carbon-carbon double bond. This process has undergone continuous growth since its invention: Production capacity reached 6.6×106 tons in 1995. It is important because aldehydes are easily converted into many secondary products. For example, the resulting aldehydes are hydrogenated to alcohols that are converted to detergents. Hydroformylation is also used in speciality chemicals, relevant to the organic synthesis of fragrances and drugs. The development of hydroformylation is one of the premier achievements of 20th-century industrial chemistry. The process entails treatment of an alkene typically with high pressures (between 10 and 100 atmospheres) of carbon monoxide and hydrogen at temperatures between 40 and 200 °C. In one variation, formal ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Journal Of The Chemical Society
The ''Journal of the Chemical Society'' was a scientific journal established by the Chemical Society in 1849 as the ''Quarterly Journal of the Chemical Society''. The first editor was Edmund Ronalds. The journal underwent several renamings, splits, and mergers throughout its history. In 1980, the Chemical Society merged with several other organizations into the Royal Society of Chemistry. The journal's continuity is found in '' Chemical Communications'', ''Dalton Transactions'', '' Faraday Transactions'', and '' Perkin Transactions'', all of which are published by the Royal Society of Chemistry. History ;''Proceedings of the Chemical Society'' * ''Memoirs of the Chemical Society of London'' (1841) * ''Proceedings of the Chemical Society of London'' (1842–1843) * ''Memoirs and Proceedings of the Chemical Society'' (1843–1848) * ''Proceedings of the Chemical Society, London'' (1885–1914) * Published as a supplement to ''Journal of the Chemical Society'' from 1914 to 1956 * '' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Homogeneous Catalysis
In chemistry, homogeneous catalysis is catalysis by a soluble catalyst in a solution. Homogeneous catalysis refers to reactions where the catalyst is in the same phase as the reactants, principally in solution. In contrast, heterogeneous catalysis describes processes where the catalysts and substrate are in distinct phases, typically solid-gas, respectively. The term is used almost exclusively to describe solutions and implies catalysis by organometallic compounds. Homogeneous catalysis is an established technology that continues to evolve. An illustrative major application is the production of acetic acid. Enzymes are examples of homogeneous catalysts. Examples Acid catalysis The proton is a pervasive homogeneous catalyst because water is the most common solvent. Water forms protons by the process of self-ionization of water. In an illustrative case, acids accelerate (catalyze) the hydrolysis of esters: :CH3CO2CH3 + H2O CH3CO2H + CH3OH At neutral pH, aqueous solutions of mo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Journal Of Computational Chemistry
The ''Journal of Computational Chemistry'' is a peer-reviewed scientific journal published since 1980 by John Wiley & Sons. It covers research, contemporary developments in theory and methodology, and applications in all areas of computational chemistry, including ab initio quantum chemistry methods and semiempirical methods, density functional theory, molecular mechanics, molecular dynamics, statistical mechanics, cheminformatics, biomolecular structure prediction, molecular design, and bioinformatics. According to the ''Journal Citation Reports'', the journal has a 2020 impact factor The impact factor (IF) or journal impact factor (JIF) of an academic journal is a scientometric index calculated by Clarivate that reflects the yearly mean number of citations of articles published in the last two years in a given journal, as ... of 3.376, ranking it 80th out of 179 journals in the category "Chemistry, Multidisciplinary". References External links * {{Official websi ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mesityl
Mesitylene or 1,3,5-trimethylbenzene is a derivative of benzene with three methyl substituents positioned symmetrically around the ring. The other two isomeric trimethylbenzenes are 1,2,4-trimethylbenzene (pseudocumene) and 1,2,3-trimethylbenzene (hemimellitene). All three compounds have the formula C6H3(CH3)3, which is commonly abbreviated C6H3Me3. Mesitylene is a colorless liquid with sweet aromatic odor. It is a component of coal tar, which is its traditional source. It is a precursor to diverse fine chemicals. The mesityl group (Mes) is a substituent with the formula C6H2Me3 and is found in various other compounds. Preparation Mesitylene is prepared by transalkylation of xylene over solid acid catalyst:Karl Griesbaum, Arno Behr, Dieter Biedenkapp, Heinz-Werner Voges, Dorothea Garbe, Christian Paetz, Gerd Collin, Dieter Mayer, Hartmut Höke “Hydrocarbons” in Ullmann's Encyclopedia of Industrial Chemistry 2002 Wiley-VCH, Weinheim. . :2 C6H4(CH3)2 ⇌ C6H3(CH ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
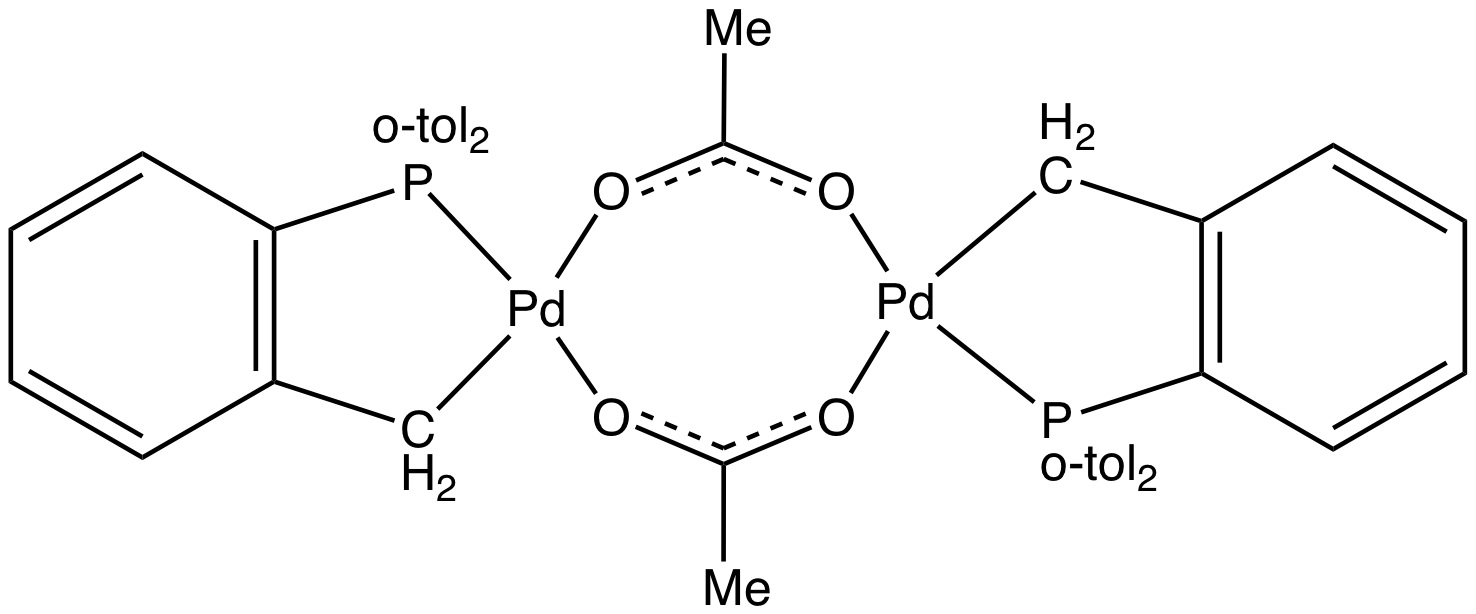
Tris(o-tolyl)phosphine
Tris(''o''-tolyl)phosphine is an organophosphorus compound with the formula P(C6H4CH3)3. It is a white, water-insoluble solid that is soluble in organic solvents. In solution it slowly converts to the phosphine oxide. As a phosphine ligand, it has a wide cone angle of 194°. Consequently, it tends to cyclometalate when treated with metal halides and metal acetates. Complexes of this ligand are common in homogeneous catalysis. {{clear-left See also *Triphenylphosphine Triphenylphosphine (IUPAC name: triphenylphosphane) is a common organophosphorus compound with the formula P(C6H5)3 and often abbreviated to P Ph3 or Ph3P. It is widely used in the synthesis of organic and organometallic compounds. PPh3 exists ... References Tertiary phosphines 2-Tolyl compounds ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Tricyclohexylphosphine
Tricyclohexylphosphine is the tertiary phosphine with the formula P( C6H11)3. Commonly used as a ligand in organometallic chemistry, it is often abbreviated to PCy3, where Cy stands for cyclohexyl. It is characterized by both high basicity (p''K''a = 9.7) and a large ligand cone angle (170°).{{cite journal , last1 = Immirzi , first1 = A. , last2 = Musco , first2 = A. , title = A Method to Measure the Size of Phosphorus Ligands in Coordination Complexes , doi = 10.1016/S0020-1693(00)95635-4 , year = 1977 , journal = Inorg. Chim. Acta , volume = 25 , pages = L41–42 Important complexes containing P(Cy)3 ligands include the 2005 Nobel Prize-winning Grubbs' catalyst and the homogeneous hydrogenation Hydrogenation is a chemical reaction between molecular hydrogen (H2) and another compound or element, usually in the presence of a catalyst such as nickel, palladium or platinum. The process is commonly employed to reduce or saturate organic ... catalyst Crabtree' ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triphenylphosphine
Triphenylphosphine (IUPAC name: triphenylphosphane) is a common organophosphorus compound with the formula P(C6H5)3 and often abbreviated to P Ph3 or Ph3P. It is widely used in the synthesis of organic and organometallic compounds. PPh3 exists as relatively air stable, colorless crystals at room temperature. It dissolves in non-polar organic solvents such as benzene and diethyl ether. Preparation and structure Triphenylphosphine can be prepared in the laboratory by treatment of phosphorus trichloride with phenylmagnesium bromide or phenyllithium. The industrial synthesis involves the reaction between phosphorus trichloride, chlorobenzene, and sodium: :PCl3 + 3 PhCl + 6 Na → PPh3 + 6 NaCl Triphenylphosphine crystallizes in triclinic and monoclinic modification. In both cases, the molecule adopts a pyramidal structure with propeller-like arrangement of the three phenyl groups. Principal reactions with chalcogens, halogens, and acids Oxidation Triphenylphosphine undergoes ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triethylphosphine
Triethylphosphine is the organophosphorus compound with the formula P(CH2CH3)3, commonly abbreviated as PEt3. It is a colorless liquid with an unpleasant odor characteristic of alkylphosphines. The compound is a common ligand in organometallic chemistry, such as in auranofin. Structure and simple reactions It is a pyramidal molecule with approximate ''C''3''v'' symmetry. PEt3 is usually prepared using Grignard reagents: : 3 CH3CH2MgCl + P(OC6H5)3 → P(CH2CH3)3 + 3 C6H5OMgCl PEt3 reacts with strong acids to give salts PEt3.Annette Schier and Hubert Schmidbaur "P-Donor Ligands" in Encyclopedia of Inorganic Chemistry 2006, Wiley-VCH, Weinheim. This reaction is reversible. Similarly, it is also easily alkylated to give phosphonium derivatives. PEt3 is easily oxidised to the phosphine oxide with oxygen. Coordination chemistry Triethylphosphine is a highly basic ligand that forms coordination complexes with many metals. As a ligand, triethylphosphine's Tolman cone angle ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |




molybdenum_tricarbonyl.png)