Triphenylphosphine on:
[Wikipedia]
[Google]
[Amazon]
Triphenylphosphine (IUPAC name: triphenylphosphane) is a common organophosphorus compound with the formula P(C6H5)3 and often abbreviated to P Ph3 or Ph3P. It is widely used in the synthesis of organic and organometallic compounds. PPh3 exists as relatively air stable, colorless crystals at room temperature. It dissolves in non-polar organic solvents such as
International Chemical Safety Card 0700
Tertiary phosphines Phenyl compounds
benzene
Benzene is an organic chemical compound with the molecular formula C6H6. The benzene molecule is composed of six carbon atoms joined in a planar ring with one hydrogen atom attached to each. Because it contains only carbon and hydrogen atoms ...
and diethyl ether
Diethyl ether, or simply ether, is an organic compound in the ether class with the formula , sometimes abbreviated as (see Pseudoelement symbols). It is a colourless, highly volatile, sweet-smelling ("ethereal odour"), extremely flammable li ...
.
Preparation and structure
Triphenylphosphine can be prepared in the laboratory by treatment of phosphorus trichloride with phenylmagnesium bromide orphenyllithium
Phenyllithium or lithobenzene is an organometallic agent with the empirical formula C6H5Li. It is most commonly used as a metalating agent in organic syntheses and a substitute for Grignard reagents for introducing phenyl groups in organic synthes ...
. The industrial synthesis involves the reaction between phosphorus trichloride, chlorobenzene
Chlorobenzene is an aromatic organic compound with the chemical formula C6H5Cl. This colorless, flammable liquid is a common solvent and a widely used intermediate in the manufacture of other chemicals.
Uses
Historical
The major use of chlorob ...
, and sodium:
:PCl3 + 3 PhCl + 6 Na → PPh3 + 6 NaCl
Triphenylphosphine crystallizes in triclinic and monoclinic modification. In both cases, the molecule adopts a pyramidal structure with propeller-like arrangement of the three phenyl groups.
Principal reactions with chalcogens, halogens, and acids
Oxidation
Triphenylphosphine undergoes slow oxidation by air to givetriphenylphosphine oxide
Triphenylphosphine oxide (often abbreviated TPPO) is the organophosphorus compound with the formula OP(C6H5)3, also written as Ph3PO or PPh3O (Ph = C6H5). This colourless crystalline compound is a common but potentially useful waste product in ...
, Ph3PO:
:2 PPh3 + O2 → 2 OPPh3
This impurity can be removed by recrystallisation of PPh3 from either hot ethanol
Ethanol (abbr. EtOH; also called ethyl alcohol, grain alcohol, drinking alcohol, or simply alcohol) is an organic compound. It is an alcohol with the chemical formula . Its formula can be also written as or (an ethyl group linked to a ...
or isopropanol. This method capitalizes on the fact that OPPh3 is more polar and hence more soluble in polar solvents than PPh3.
Triphenylphosphine abstracts sulfur from polysulfide
Polysulfides are a class of chemical compounds containing chains of sulfur atoms. There are two main classes of polysulfides: inorganic and organic. Among the inorganic polysulfides, there are ones which contain anions, which have the general formu ...
compounds, episulfides, and elemental sulfur. Simple organosulfur compounds such as thiol
In organic chemistry, a thiol (; ), or thiol derivative, is any organosulfur compound of the form , where R represents an alkyl or other organic substituent. The functional group itself is referred to as either a thiol group or a sulfhydryl gro ...
s and thioether
In organic chemistry, an organic sulfide (British English sulphide) or thioether is an organosulfur functional group with the connectivity as shown on right. Like many other sulfur-containing compounds, volatile sulfides have foul odors. A su ...
s are unreactive, however. The phosphorus-containing product is triphenylphosphine sulfide
Triphenylphosphine sulfide (IUPAC name: triphenyl-''λ''5-phosphanethione) is the organophosphorus compound with the formula , usually written (where Ph = phenyl). It is a colourless solid, which is soluble in a variety of organic solvents.
Struc ...
, Ph3PS. This reaction can be employed to assay the "labile" S0 content of a sample, say vulcanized rubber. Triphenylphosphine selenide, Ph3PSe, may be easily prepared via treatment of PPh3 with red (alpha-monoclinic) Se. Salts of selenocyanate, SeCN−, are used as the Se0 source. PPh3 can also form an adduct with Te, although this adduct primarily exists as (Ph3P)2Te rather than PPh3Te.
Aryl azides
In chemistry, azide is a linear, polyatomic anion with the formula and structure . It is the conjugate base of hydrazoic acid . Organic azides are organic compounds with the formula , containing the azide functional group. The dominant applicat ...
react with PPh3 to give phosphanimines, analogues of OPPh3, via the Staudinger reaction. Illustrative is the preparation of triphenylphosphine phenylimide:
:PPh3 + PhN3 → PhNPPh3 + N2
The phosphanimine can be hydrolyzed to the amine. Typically the intermediate phosphanimine is not isolated.
:PPh3 + RN3 + H2O → OPPh3 + N2 + RNH2
Chlorination
Cl2 adds to PPh3 to givetriphenylphosphine dichloride
Triphenylphosphine dichloride, Ph3PCl2, is a chlorinating agent widely used in organic chemistry. Applications include the conversion of alcohols and ethers to alkyl chlorides, the cleavage of epoxides to vicinal dichlorides and the chlorination ...
( Ph3Cll), which exists as the moisture-sensitive phosphonium halide. This reagent is used to convert alcohols to alkyl chlorides in organic synthesis. Bis(triphenylphosphine)iminium chloride (PPN+Cl−, formula C6H5)3P)2Nl is prepared from triphenylphosphine dichloride:
:2 Ph3PCl2 + NH2OH·HCl + Ph3P → Cl + 4HCl + Ph3PO
Protonation
PPh3 is a weak base. It forms isolable triphenylphosphonium salts with strong acids such as HBr:Hercouet, A.; LeCorre, M. (1988) Triphenylphosphonium bromide: A convenient and quantitative source of gaseous hydrogen bromide. Synthesis, 157–158 :P(C6H5)3 + HBr → P(C6H5)3sup>+Br−Organic reactions
PPh3 is widely used in organic synthesis. The properties that guide its usage are its nucleophilicity and its reducing character. The nucleophilicity of PPh3 is indicated by its reactivity toward electrophilic alkenes, such as Michael-acceptors, and alkyl halides. It is also used in the synthesis of biaryl compounds, such as theSuzuki reaction
The Suzuki reaction is an organic reaction, classified as a cross-coupling reaction, where the coupling partners are a boronic acid and an organohalide and the catalyst is a palladium, palladium(0) complex. It was first published in 1979 by Akira ...
.
Quaternization
PPh3 combines withalkyl halide
The haloalkanes (also known as halogenoalkanes or alkyl halides) are alkanes containing one or more halogen substituents. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely us ...
s to give phosphonium salt
In polyatomic cations with the chemical formula (where R is a hydrogen or an alkyl, aryl, or halide group). These cations have tetrahedral structures. The salts are generally colorless or take the color of the anions.
Types of phosphonium c ...
s. This quaternization
In organic chemistry, the Menshutkin reaction converts a tertiary amine into a quaternary ammonium salt by reaction with an alkyl halide. Similar reactions occur when tertiary phosphines are treated with alkyl halides.
The reaction is the met ...
reaction is particularly fast for benzylic and allylic halides:
:PPh3 + CH3I → H3PPh3sup>+I−
These salts, which can often be isolated as crystalline solids, react with strong bases to form ylides, which are reagents in the Wittig reaction
The Wittig reaction or Wittig olefination is a chemical reaction of an aldehyde or ketone with a triphenyl phosphonium ylide called a Wittig reagent. Wittig reactions are most commonly used to convert aldehydes and ketones to alkenes. Mos ...
s.
Aryl halides will quaternize PPh3 to give tetraphenylphosphonium
Tetraphenylphosphonium chloride is the chemical compound with the formula (C6H5)4PCl, abbreviated Ph4PCl or PPh4Cl. Tetraphenylphosphonium and especially tetraphenylarsonium salts were formerly of interest in gravimetric analysis of perchlorate an ...
salts:
:PPh3 + PhBr → Ph4r
The reaction however requires elevated temperatures and metal catalysts.
Mitsunobu reaction
In theMitsunobu reaction
The Mitsunobu reaction is an organic reaction that converts an alcohol into a variety of functional groups, such as an ester, using triphenylphosphine and an azodicarboxylate such as diethyl azodicarboxylate (DEAD) or diisopropyl azodicarboxylat ...
, a mixture of triphenylphosphine and diisopropyl azodicarboxylate ("DIAD", or its diethyl analogue, DEAD
Death is the irreversible cessation of all biological functions that sustain an organism. For organisms with a brain, death can also be defined as the irreversible cessation of functioning of the whole brain, including brainstem, and brain ...
) converts an alcohol and a carboxylic acid to an ester. DIAD is reduced as it serves as the hydrogen acceptor, and the PPh3 is oxidized to OPPh3.
Appel reaction
In theAppel reaction
The Appel reaction is an organic reaction that converts an alcohol into an alkyl chloride using triphenylphosphine and carbon tetrachloride. The use of carbon tetrabromide or bromine as a halide source will yield alkyl bromides, whereas using carbo ...
, a mixture of PPh3 and CX4 (X = Cl, Br) is used to convert alcohols to alkyl halides. Triphenylphosphine oxide
Triphenylphosphine oxide (often abbreviated TPPO) is the organophosphorus compound with the formula OP(C6H5)3, also written as Ph3PO or PPh3O (Ph = C6H5). This colourless crystalline compound is a common but potentially useful waste product in ...
(OPPh3) is a byproduct.
:PPh3 + CBr4 + RCH2OH → OPPh3 + RCH2Br + HCBr3
This reaction commences with nucleophilic attack of PPh3 on CBr4, an extension of the quaternization reaction listed above.
Deoxygenation
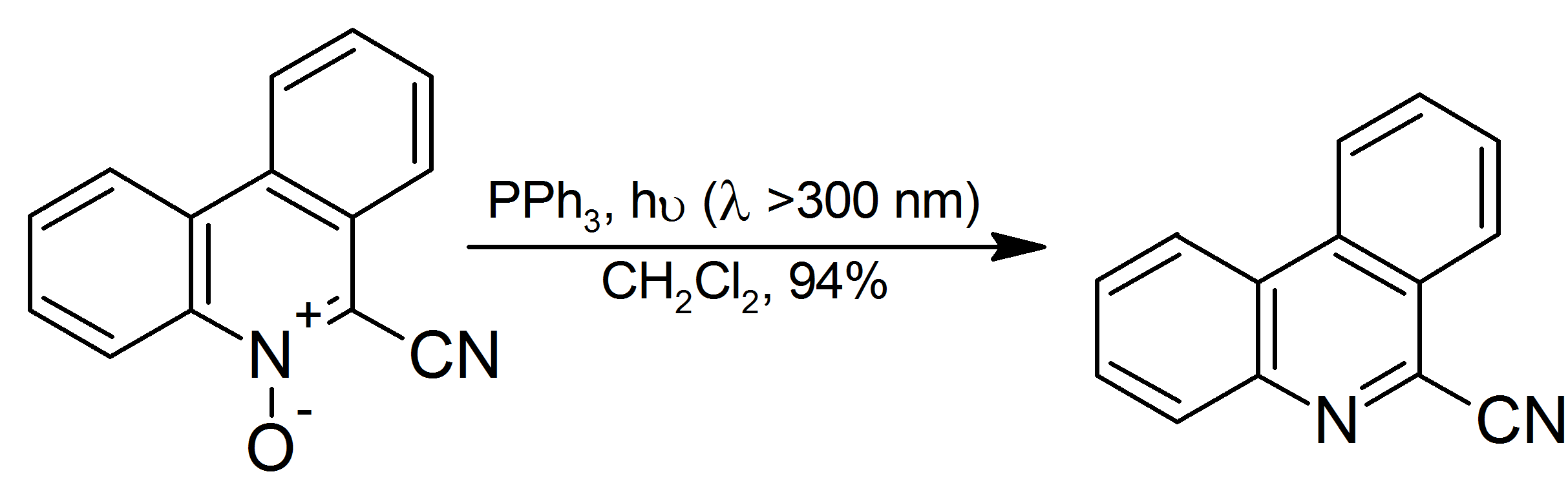
The easy oxygenation of PPh3 is exploited in its use to deoxygenate organic peroxides, which generally occurs with retention of configuration: :PPh3 + RO2H → OPPh3 + ROH (R = alkyl) It is also used for the decomposition of organicozonide
Ozonide is the polyatomic anion . Cyclic organic compounds formed by the addition of ozone () to an alkene are also called ozonides.
Ionic ozonides
Inorganic ozonides are dark red salts. The anion has the bent shape of the ozone molecule.
Ino ...
s to ketones and aldehydes, although dimethyl sulfide is more popular for the reaction as the side product, dimethyl sulfoxide is more readily separated from the reaction mixture than triphenylphosphine oxide. Aromatic ''N''-oxides are reduced to the corresponding amine in high yield at room temperature with irradiation:
:
Sulfonation
Sulfonation Aromatic sulfonation is an organic reaction in which a hydrogen atom on an arene is replaced by a sulfonic acid functional group in an electrophilic aromatic substitution. Aryl sulfonic acids are used as detergents, dye, and drugs.
Stoichiometry a ...
of PPh3 gives tris(3-sulfophenyl)phosphine, P(C6H4-3-SO3−)3 ( TPPTS), usually isolated as the trisodium salt. In contrast to PPh3, TPPTS is water-soluble, as are its metal derivatives. Rhodium complexes of TPPTS are used in certain industrial hydroformylation reactions.
Reduction to diphenylphosphide
Lithium
Lithium (from el, λίθος, lithos, lit=stone) is a chemical element with the symbol Li and atomic number 3. It is a soft, silvery-white alkali metal. Under standard conditions, it is the least dense metal and the least dense solid ...
in THF as well as Na or K react with PPh3 to give Ph2PM (M = Li, Na, K). These salts are versatile precursors to tertiary phosphines. For example, 1,2-dibromoethane
1,2-Dibromoethane, also known as ethylene dibromide (EDB), is an organobromine compound with the chemical formula . Although trace amounts occur naturally in the ocean, where it is formed probably by algae and kelp, it is mainly synthetic. It is ...
and Ph2PM react to give Ph2PCH2CH2PPh2. Weak acids such ammonium chloride, convert Ph2PM (M = Li, Na, K) into diphenylphosphine
Diphenylphosphine, also known as diphenylphosphane, is an organophosphorus compound with the formula (C6H5)2PH. This foul-smelling, colorless liquid is easily oxidized in air. It is a precursor to organophosphorus ligands for use as catalysts.
Syn ...
:
:(C6H5)2PM + H2O → (C6H5)2PH + MOH
Transition metal complexes
Triphenylphosphine binds well to mosttransition metal
In chemistry, a transition metal (or transition element) is a chemical element in the d-block of the periodic table (groups 3 to 12), though the elements of group 12 (and less often group 3) are sometimes excluded. They are the elements that ca ...
s, especially those in the middle and late transition metals of groups 7–10. In terms of steric bulk, PPh3 has a Tolman cone angle
In coordination chemistry, the ligand cone angle (a common example being the Tolman cone angle or ''θ'') is a measure of the steric bulk of a ligand in a transition metal coordination complex. It is defined as the solid angle formed with the me ...
of 145°, which is intermediate between those of P(C6H11)3 (170°) and P(CH3)3 (115°). In an early application in homogeneous catalysis
In chemistry, homogeneous catalysis is catalysis by a soluble catalyst in a solution. Homogeneous catalysis refers to reactions where the catalyst is in the same phase as the reactants, principally in solution. In contrast, heterogeneous catalysi ...
, NiBr2(PPh3)2 was used by Walter Reppe
Walter Julius Reppe (29 July 1892 in Göringen – 26 July 1969 in Heidelberg) was a German chemist. He is notable for his contributions to the chemistry of acetylene.
Education and career
Walter Reppe began his study of the natural sciences ...
for the synthesis of acrylate esters from alkynes, carbon monoxide
Carbon monoxide (chemical formula CO) is a colorless, poisonous, odorless, tasteless, flammable gas that is slightly less dense than air. Carbon monoxide consists of one carbon atom and one oxygen atom connected by a triple bond. It is the simple ...
, and alcohols.*{{cite journal , title = Cyclisierende Polymerisation von Acetylen. III Benzol, Benzolderivate und hydroaromatische Verbindungen , author1=Reppe, W. , author2=Schweckendiek, W. J. , journal = Justus Liebigs Annalen der Chemie , volume = 560 , issue = 1 , pages = 104–116 , year = 1948 , doi = 10.1002/jlac.19485600104 The use of PPh3 was popularized by its use in the hydroformylation catalyst RhH(PPh3)3(CO).
Polymer-anchored PPh3 derivatives
Polymeric analogues of PPh3 are known whereby polystyrene is modified with PPh2 groups at the para position. Such polymers can be employed in many of the applications used for PPh3 with the advantage that the polymer, being insoluble, can be separated from products by simple filtration of reaction slurries. Such polymers are prepared via treatment of 4-lithiophenyl-substituted polystyrene withchlorodiphenylphosphine
Chlorodiphenylphosphine is an organophosphorus compound with the formula (C6H5)2PCl, abbreviated Ph2PCl. It is a colourless oily liquid with a pungent odor that is often described as being garlic-like and detectable even in the ppb range. It is u ...
(PPh2Cl).
See also
* Tris(o-tolyl)phosphine * Decyl(triphenyl)phosphoniumReferences
External links
International Chemical Safety Card 0700
Tertiary phosphines Phenyl compounds