|
Gas Law
The laws describing the behaviour of gases under fixed pressure, volume, amount of gas, and absolute temperature conditions are called gas laws. The basic gas laws were discovered by the end of the 18th century when scientists found out that relationships between pressure, volume and temperature of a sample of gas could be obtained which would hold to approximation for all gases. The combination of several empirical gas laws led to the development of the ideal gas law. The ideal gas law was later found to be consistent with atomic and kinetic theory. History In 1643, the Italian physicist and mathematician, Evangelista Torricelli, who for a few months had acted as Galileo Galilei's secretary, conducted a celebrated experiment in Florence. He demonstrated that a column of mercury in an inverted tube can be supported by the pressure of air outside of the tube, with the creation of a small section of vacuum above the mercury. This experiment essentially paved the way towards the ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Boltzmann Constant
The Boltzmann constant ( or ) is the proportionality factor that relates the average relative thermal energy of particles in a ideal gas, gas with the thermodynamic temperature of the gas. It occurs in the definitions of the kelvin (K) and the molar gas constant, in Planck's law of black-body radiation and Boltzmann's entropy formula, and is used in calculating Johnson–Nyquist noise, thermal noise in resistors. The Boltzmann constant has Dimensional analysis, dimensions of energy divided by temperature, the same as entropy and heat capacity. It is named after the Austrian scientist Ludwig Boltzmann. As part of the 2019 revision of the SI, the Boltzmann constant is one of the seven "Physical constant, defining constants" that have been defined so as to have exact finite decimal values in SI units. They are used in various combinations to define the seven SI base units. The Boltzmann constant is defined to be exactly joules per kelvin, with the effect of defining the SI unit ke ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
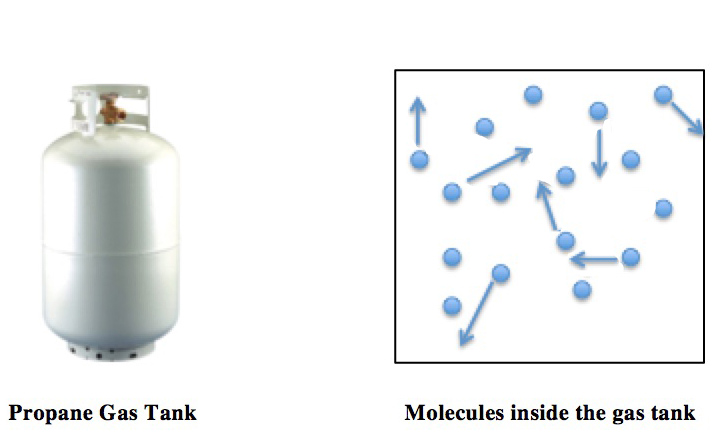
Ideal Gas
An ideal gas is a theoretical gas composed of many randomly moving point particles that are not subject to interparticle interactions. The ideal gas concept is useful because it obeys the ideal gas law, a simplified equation of state, and is amenable to analysis under statistical mechanics. The requirement of zero interaction can often be relaxed if, for example, the interaction is perfectly elastic or regarded as point-like collisions. Under various conditions of temperature and pressure, many real gases behave qualitatively like an ideal gas where the gas molecules (or atoms for monatomic gas) play the role of the ideal particles. Many gases such as nitrogen, oxygen, hydrogen, noble gases, some heavier gases like carbon dioxide and mixtures such as air, can be treated as ideal gases within reasonable tolerances over a considerable parameter range around standard temperature and pressure. Generally, a gas behaves more like an ideal gas at higher temperature and lower ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Ideal Gas Law
The ideal gas law, also called the general gas equation, is the equation of state of a hypothetical ideal gas. It is a good approximation of the behavior of many gases under many conditions, although it has several limitations. It was first stated by Benoît Paul Émile Clapeyron in 1834 as a combination of the empirical Boyle's law, Charles's law, Avogadro's law, and Gay-Lussac's law. The ideal gas law is often written in an empirical form: pV = nRT where p, V and T are the pressure, volume and Thermodynamic temperature, temperature respectively; n is the amount of substance; and R is the ideal gas constant. It can also be derived from the microscopic kinetic theory of gases, kinetic theory, as was achieved (independently) by August Krönig in 1856 and Rudolf Clausius in 1857. Equation The state function, state of an amount of gas is determined by its pressure, volume, and temperature. The modern form of the equation relates these simply in two main forms. The temperature us ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Mass
Mass is an Intrinsic and extrinsic properties, intrinsic property of a physical body, body. It was traditionally believed to be related to the physical quantity, quantity of matter in a body, until the discovery of the atom and particle physics. It was found that different atoms and different elementary particle, elementary particles, theoretically with the same amount of matter, have nonetheless different masses. Mass in modern physics has multiple Mass in special relativity, definitions which are conceptually distinct, but physically equivalent. Mass can be experimentally defined as a measure (mathematics), measure of the body's inertia, meaning the resistance to acceleration (change of velocity) when a net force is applied. The object's mass also determines the Force, strength of its gravitational attraction to other bodies. The SI base unit of mass is the kilogram (kg). In physics, mass is Mass versus weight, not the same as weight, even though mass is often determined by ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Graham's Law
Graham's law of effusion (also called Graham's law of diffusion) was formulated by Scottish physical chemist Thomas Graham in 1848. Keith J. Laidler and John M. Meiser, ''Physical Chemistry'' (Benjamin/Cummings 1982), pp. 18–19 Graham found experimentally that the rate of effusion of a gas is inversely proportional to the square root of the molar mass of its particles. This formula is stated as: :=\sqrt, where: :Rate1 is the rate of effusion for the first gas. (volume or number of moles per unit time). :Rate2 is the rate of effusion for the second gas. :''M1'' is the molar mass of gas 1 :''M2'' is the molar mass of gas 2. Graham's law states that the rate of diffusion or of effusion of a gas is inversely proportional to the square root of its molecular weight. Thus, if the molecular weight of one gas is four times that of another, it would diffuse through a porous plug or escape through a small pinhole in a vessel at half the rate of the other (heavier gases diffuse mor ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Real Gas
Real gases are non-ideal gases whose molecules occupy space and have interactions; consequently, they do not adhere to the ideal gas law. To understand the behaviour of real gases, the following must be taken into account: * compressibility effects; *variable specific heat capacity; *van der Waals forces; *non-equilibrium thermodynamic effects; *issues with molecular dissociation and elementary reactions with variable composition For most applications, such a detailed analysis is unnecessary, and the ideal gas approximation can be used with reasonable accuracy. On the other hand, real-gas models have to be used near the condensation point of gases, near critical points, at very high pressures, to explain the Joule–Thomson effect, and in other less usual cases. The deviation from ideality can be described by the compressibility factor Z. Models Van der Waals model Real gases are often modeled by taking into account their molar weight and molar volume RT = \left(p + \frac\r ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Universal Gas Constant
The molar gas constant (also known as the gas constant, universal gas constant, or ideal gas constant) is denoted by the symbol or . It is the molar equivalent to the Boltzmann constant, expressed in units of energy per temperature, temperature increment per amount of substance, rather than energy per temperature increment per ''particle''. The constant is also a combination of the constants from Boyle's law, Charles's law, Avogadro's law, and Gay-Lussac's law. It is a physical constant that is featured in many fundamental equations in the physical sciences, such as the ideal gas law, the Arrhenius equation, and the Nernst equation. The gas constant is the constant of proportionality that relates the energy scale in physics to the temperature scale and the scale used for amount of substance. Thus, the value of the gas constant ultimately derives from historical decisions and accidents in the setting of units of energy, temperature and amount of substance. The Boltzmann constan ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Avogadro's Law
Avogadro's law (sometimes referred to as Avogadro's hypothesis or Avogadro's principle) or Avogadro-Ampère's hypothesis is an experimental gas law relating the volume of a gas to the amount of substance of gas present. The law is a specific case of the ideal gas law. A modern statement is: Avogadro's law states that "equal volumes of all gases, at the same temperature and pressure, have the same number of molecules." For a given mass of an ideal gas, the volume and amount (moles) of the gas are directly proportional if the temperature and pressure are constant. The law is named after Amedeo Avogadro who, in 1812, hypothesized that two given samples of an ideal gas, of the same volume and at the same temperature and pressure, contain the same number of molecules. As an example, equal volumes of gaseous hydrogen and nitrogen contain the same number of molecules when they are at the same temperature and pressure, and display ideal gas behavior. In practice, real gases show sm ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Combined Gas Law
The ideal gas law, also called the general gas equation, is the equation of state of a hypothetical ideal gas. It is a good approximation of the behavior of many gases under many conditions, although it has several limitations. It was first stated by Benoît Paul Émile Clapeyron in 1834 as a combination of the empirical Boyle's law, Charles's law, Avogadro's law, and Gay-Lussac's law. The ideal gas law is often written in an empirical form: pV = nRT where p, V and T are the pressure, volume and temperature respectively; n is the amount of substance; and R is the ideal gas constant. It can also be derived from the microscopic kinetic theory, as was achieved (independently) by August Krönig in 1856 and Rudolf Clausius in 1857. Equation The state of an amount of gas is determined by its pressure, volume, and temperature. The modern form of the equation relates these simply in two main forms. The temperature used in the equation of state is an absolute temperature: the a ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Standard Temperature And Pressure
Standard temperature and pressure (STP) or standard conditions for temperature and pressure are various standard sets of conditions for experimental measurements used to allow comparisons to be made between different sets of data. The most used standards are those of the International Union of Pure and Applied Chemistry (IUPAC) and the National Institute of Standards and Technology (NIST), although these are not universally accepted. Other organizations have established a variety of other definitions. In industry and commerce, the standard conditions for temperature and pressure are often necessary for expressing the volumes of gases and liquids and related quantities such as the rate of volumetric flow (the volumes of gases vary significantly with temperature and pressure): standard cubic meters per second (Sm3/s), and normal cubic meters per second (Nm3/s). Many technical publications (books, journals, advertisements for equipment and machinery) simply state "standard cond ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Molar Volume
In chemistry and related fields, the molar volume, symbol ''V''m, or \tilde V of a substance is the ratio of the volume (''V'') occupied by a substance to the amount of substance (''n''), usually at a given temperature and pressure. It is also equal to the molar mass (''M'') divided by the mass density (''ρ''): V_ = \frac = \frac The molar volume has the SI unit of cubic metres per mole (m3/mol), although it is more typical to use the units cubic decimetres per mole (dm3/mol) for gases, and cubic centimetres per mole (cm3/mol) for liquids and solids. Definition The molar volume of a substance ''i'' is defined as its molar mass divided by its density ''ρ''''i''0: V_ = For an ideal mixture containing ''N'' components, the molar volume of the mixture is the weighted sum of the molar volumes of its individual components. For a real mixture the molar volume cannot be calculated without knowing the density: V_ = \frac There are many liquid–liquid mixtures, for instance mix ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Encyclopædia Britannica
The is a general knowledge, general-knowledge English-language encyclopaedia. It has been published by Encyclopædia Britannica, Inc. since 1768, although the company has changed ownership seven times. The 2010 version of the 15th edition, which spans 32 volumes and 32,640 pages, was the last printed edition. Since 2016, it has been published exclusively as an online encyclopedia, online encyclopaedia. Printed for 244 years, the ''Britannica'' was the longest-running in-print encyclopaedia in the English language. It was first published between 1768 and 1771 in Edinburgh, Scotland, in three volumes. The encyclopaedia grew in size; the second edition was 10 volumes, and by its fourth edition (1801–1810), it had expanded to 20 volumes. Its rising stature as a scholarly work helped recruit eminent contributors, and the 9th (1875–1889) and Encyclopædia Britannica Eleventh Edition, 11th editions (1911) are landmark encyclopaedias for scholarship and literary ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |