|
Zinc Iodide
Zinc iodide is the inorganic compound with the formula ZnI2. It exists both in anhydrous form and as a dihydrate. Both are white and readily absorb water from the atmosphere. It has no major application. Preparation It can be prepared by the direct reaction of zinc and iodine in water or in refluxing ether. or by treating zinc with iodine in aqueous solution: : Zn + I2 → ZnI2 Structure as solid, gas, and in solution The structure of solid ZnI2 is unusual relative to the dichloride. While zinc centers are tetrahedrally coordinated, as in ZnCl2, groups of four of these tetrahedra share three vertices to form “super-tetrahedra” of composition , which are linked by their vertices to form a three-dimensional structure. These "super-tetrahedra" are similar to the P4O10 structure. Molecular ZnI2 is linear as predicted by VSEPR theory with a Zn-I bond length of 238 pm. In aqueous solution the following have been detected: Zn(H2O)62+, nI(H2O)5sup>+, tetrahedral ZnI2(H2O)2, Zn ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Pearson Symbol
The Pearson symbol, or Pearson notation, is used in crystallography as a means of describing a crystal structure, and was originated by W. B. Pearson. The symbol is made up of two letters followed by a number. For example: * Diamond structure, ''cF''8 * Rutile structure, ''tP''6 The two (italicised) letters specify the Bravais lattice. The lower-case letter specifies the crystal family, and the upper-case letter the centering type. The number at the end of the Pearson symbol gives the number of the atoms in the conventional unit cell.Nomenclature of Inorganic Chemistry IUPAC Recommendations 2005 IR-3.4.4, pp. 49–51; IR-11.5, pp. 241–242. |
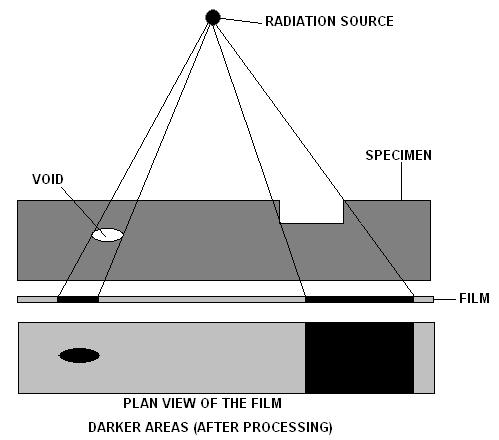
Industrial Radiography
Industrial radiography is a modality of non-destructive testing that uses ionizing radiation to inspect materials and components with the objective of locating and quantifying defects and degradation in material properties that would lead to the failure of engineering structures. It plays an important role in the science and technology needed to ensure product quality and reliability. In Australia, industrial radiographic non-destructive testing is colloquially referred to as "bombing" a component with a "bomb". Industrial Radiography uses either X-rays, produced with X-ray generators, or gamma rays generated by the natural radioactivity of sealed radionuclide sources. Neutrons can also be used. After crossing the specimen, photons are captured by a detector, such as a silver halide film, a phosphor plate, flat panel detector or CdTe detector. The examination can be performed in static 2D (named radiography), in real time 2D (fluoroscopy), or in 3D after image reconstructio ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Iodides
An iodide ion is the ion I−. Compounds with iodine in formal oxidation state −1 are called iodides. In everyday life, iodide is most commonly encountered as a component of iodized salt, which many governments mandate. Worldwide, iodine deficiency affects two billion people and is the leading preventable cause of intellectual disability. Structure and characteristics of inorganic iodides Iodide is one of the largest monatomic anions. It is assigned a radius of around 206 picometers. For comparison, the lighter halides are considerably smaller: bromide (196 pm), chloride (181 pm), and fluoride (133 pm). In part because of its size, iodide forms relatively weak bonds with most elements. Most iodide salts are soluble in water, but often less so than the related chlorides and bromides. Iodide, being large, is less hydrophilic compared to the smaller anions. One consequence of this is that sodium iodide is highly soluble in acetone, whereas sodium chloride is ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
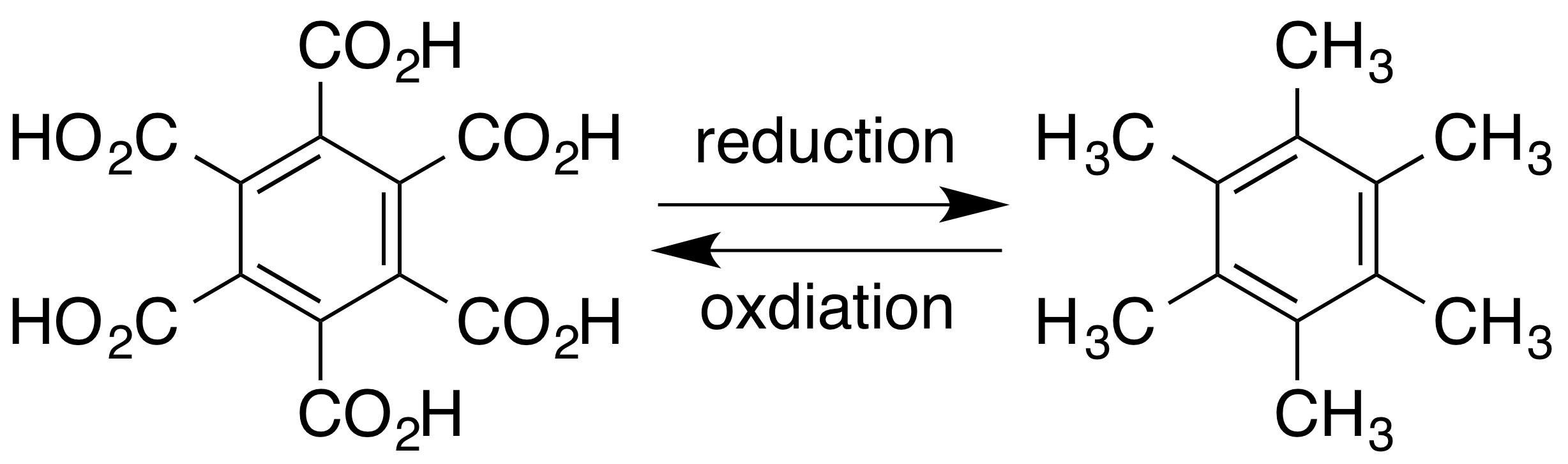
Hexamethylbenzene
Hexamethylbenzene, also known as mellitene, is a hydrocarbon with the molecular formula C12H18 and the condensed structural formula C6(CH3)6. It is an aromatic compound and a derivative of benzene, where benzene's six hydrogen atoms have each been replaced by a methyl group. In 1929 Kathleen Lonsdale reported the crystal structure of hexamethylbenzene, demonstrating that the central ring is hexagonal and flat and thereby ending an ongoing debate about the physical parameters of the benzene system. This was a historically significant result, both for the field of X-ray crystallography and for understanding aromaticity. The compound can be prepared by reacting phenol with methanol at elevated temperatures over a suitable solid catalyst such as alumina. The mechanism of the process has been studied extensively, with several intermediates having been identified. Alkyne trimerisation of dimethylacetylene also yields hexamethylbenzene in the presence of a suitable catalyst. Hexa ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Triptane
Triptane, or 2,2,3-trimethylbutane, is an organic chemical compound with the molecular formula C7 H16 or (H3C-)3C-C(-CH3)2H. It is therefore an alkane, specifically the most compact and heavily branched of the heptane isomers, the only one with a butane (C4) backbone. Triptane is commonly used as an anti-knock additive in aviation fuels. See also *List of gasoline additives Petrol additives increase petrol's octane rating or act as corrosion inhibitors or lubricants, thus allowing the use of higher compression ratios for greater efficiency and power. Types of additives include metal deactivators, corrosion inhibitors ... References Hydrocarbons Alkanes {{Hydrocarbon-stub ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Methanol
Methanol (also called methyl alcohol and wood spirit, amongst other names) is an organic chemical and the simplest aliphatic alcohol, with the formula C H3 O H (a methyl group linked to a hydroxyl group, often abbreviated as MeOH). It is a light, volatile, colourless, flammable liquid with a distinctive alcoholic odour similar to that of ethanol (potable alcohol). A polar solvent, methanol acquired the name wood alcohol because it was once produced chiefly by the destructive distillation of wood. Today, methanol is mainly produced industrially by hydrogenation of carbon monoxide. Methanol consists of a methyl group linked to a polar hydroxyl group. With more than 20 million tons produced annually, it is used as a precursor to other commodity chemicals, including formaldehyde, acetic acid, methyl tert-butyl ether, methyl benzoate, anisole, peroxyacids, as well as a host of more specialised chemicals. Occurrence Small amounts of methanol are present in normal, healthy ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Osmium Tetroxide
Osmium tetroxide (also osmium(VIII) oxide) is the chemical compound with the formula OsO4. The compound is noteworthy for its many uses, despite its toxicity and the rarity of osmium. It also has a number of unusual properties, one being that the solid is volatile. The compound is colourless, but most samples appear yellow. This is most likely due to the presence of the impurity OsO2, which is yellow-brown in colour. In biology, its property of binding to lipids has made it a widely-used stain in electron microscopy. Physical properties Osmium(VIII) oxide forms monoclinic crystals. It has a characteristic acrid chlorine-like odor. The element name osmium is derived from ''osme'', Greek for ''odor''. OsO4 is volatile: it sublimes at room temperature. It is soluble in a wide range of organic solvents. It is also moderately soluble in water, with which it reacts reversibly to form osmic acid (see below). ''Pure'' osmium(VIII) oxide is probably colourless and it has been sug ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrode
An electrode is an electrical conductor used to make contact with a nonmetallic part of a circuit (e.g. a semiconductor, an electrolyte, a vacuum or air). Electrodes are essential parts of batteries that can consist of a variety of materials depending on the type of battery. The electrophore, invented by Johan Wilcke, was an early version of an electrode used to study static electricity. Anode and cathode in electrochemical cells Electrodes are an essential part of any battery. The first electrochemical battery made was devised by Alessandro Volta and was aptly named the Voltaic cell. This battery consisted of a stack of copper and zinc electrodes separated by brine-soaked paper disks. Due to fluctuation in the voltage provided by the voltaic cell it wasn't very practical. The first practical battery was invented in 1839 and named the Daniell cell after John Frederic Daniell. Still making use of the zinc–copper electrode combination. Since then many more batteries ha ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Zinc Bromide
Zinc bromide ( Zn Br2) is an inorganic compound with the chemical formula Zn Br2. It is a colourless salt that shares many properties with zinc chloride (ZnCl2), namely a high solubility in water forming acidic solutions, and good solubility in organic solvents. It is hygroscopic and forms a dihydrate ZnBr2·2H2O. Production ZnBr2 · 2H2O is prepared by treating zinc oxide or zinc metal with hydrobromic acid. : ZnO + 2HBr + H2O → ZnBr2·2H2O : Zn + 2HBr → ZnBr2 + H2 The anhydrous material can be produced by dehydration of the dihydrate with hot CO2 or by reaction of zinc metal and bromine. Sublimation in a stream of hydrogen bromide also gives the anhydrous derivative. Structure ZnBr2 crystallizes in the same structure as ZnI2: four tetrahedral Zn centers share three vertices to form “super-tetrahedra” of nominal composition 2−, which are linked by their vertices to form a three-dimensional structure. The dihydrate ZnBr2 · 2H2O can be described as ( n(H2O ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Electrochemical Cell
An electrochemical cell is a device capable of either generating electrical energy from chemical reactions or using electrical energy to cause chemical reactions. The electrochemical cells which generate an electric current are called voltaic or galvanic cells and those that generate chemical reactions, via electrolysis for example, are called electrolytic cells. A common example of a galvanic cell is a standard 1.5 volt cell meant for consumer use. A Battery (electricity), ''battery'' consists of one or more cells, connected in Series and parallel circuits, parallel, series or series-and-parallel pattern. Electrolytic cell An electrolytic cell is an electrochemical cell that drives a non-spontaneous redox reaction through the application of electrical energy. They are often used to decompose chemical compounds, in a process called electrolysis—the Greek word lysis means ''to break up''. Important examples of electrolysis are the decomposition of water into hydrogen and ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Patent
A patent is a type of intellectual property that gives its owner the legal right to exclude others from making, using, or selling an invention for a limited period of time in exchange for publishing an enabling disclosure of the invention."A patent is not the grant of a right to make or use or sell. It does not, directly or indirectly, imply any such right. It grants only the right to exclude others. The supposition that a right to make is created by the patent grant is obviously inconsistent with the established distinctions between generic and specific patents, and with the well-known fact that a very considerable portion of the patents granted are in a field covered by a former relatively generic or basic patent, are tributary to such earlier patent, and cannot be practiced unless by license thereunder." – ''Herman v. Youngstown Car Mfg. Co.'', 191 F. 579, 584–85, 112 CCA 185 (6th Cir. 1911) In most countries, patent rights fall under private law and the patent holder ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Opacity (optics)
Opacity is the measure of impenetrability to electromagnetic or other kinds of radiation, especially visible light. In radiative transfer, it describes the absorption and scattering of radiation in a medium, such as a plasma, dielectric, shielding material, glass, etc. An opaque object is neither transparent (allowing all light to pass through) nor translucent (allowing some light to pass through). When light strikes an interface between two substances, in general some may be reflected, some absorbed, some scattered, and the rest transmitted (also see refraction). Reflection can be diffuse, for example light reflecting off a white wall, or specular, for example light reflecting off a mirror. An opaque substance transmits no light, and therefore reflects, scatters, or absorbs all of it. Both mirrors and carbon black are opaque. Opacity depends on the frequency of the light being considered. For instance, some kinds of glass, while transparent in the visual range, are lar ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |