|
Transglutaminase
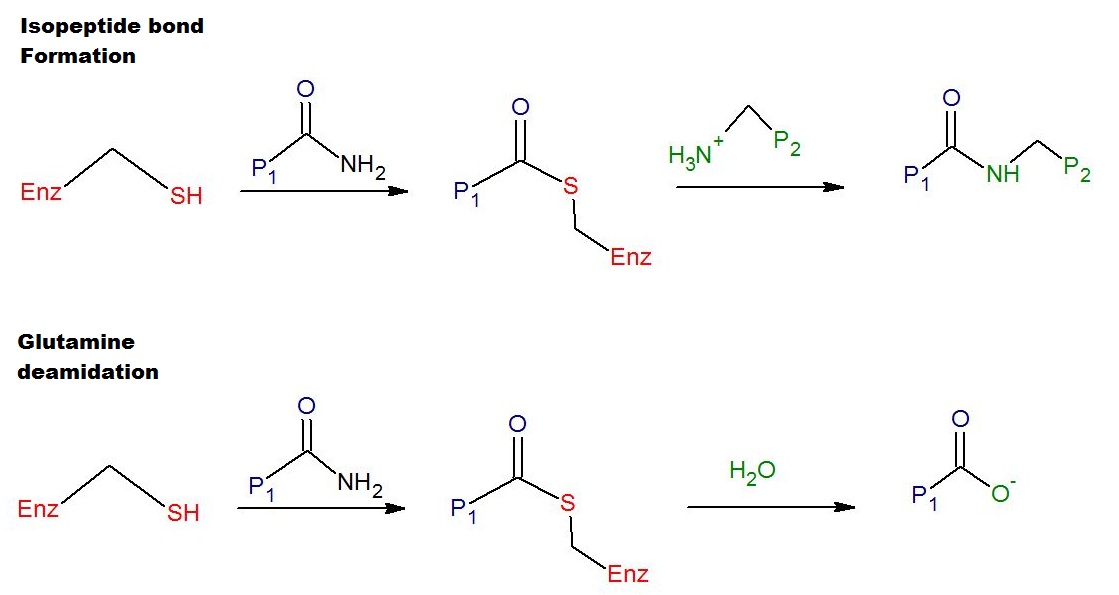
Transglutaminases are enzymes that in nature primarily catalyze the formation of an isopeptide bond between γ- carboxamide groups ( -(C=O)NH2 ) of glutamine residue side chains and the ε- amino groups ( -NH2 ) of lysine residue side chains with subsequent release of ammonia ( NH3 ). Lysine and glutamine residues must be bound to a peptide or a protein so that this cross-linking (between separate molecules) or intramolecular (within the same molecule) reaction can happen. Bonds formed by transglutaminase exhibit high resistance to proteolytic degradation (proteolysis). The reaction is : Gln-(C=O)NH2 + NH2-Lys → Gln-(C=O)NH-Lys + NH3 Transglutaminases can also join a primary amine ( RNH2 ) to the side chain carboxyamide group of a protein/peptide bound glutamine residue thus forming an isopeptide bond :Gln-(C=O)NH2 + RNH2 → Gln-(C=O)NHR + NH3 These enzymes can also deamidate glutamine residues to glutamic acid residues in the presence of wate ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Isopeptide Bond
An isopeptide bond is a type of amide bond formed between a carboxyl group of one amino acid and an amino group of another. An isopeptide bond is the linkage between the side chain amino or carboxyl group of one amino acid to the α-carboxyl, α-amino group, or the side chain of another amino acid. In a typical peptide bond, also known as eupeptide bond, the amide bond always forms between the α-carboxyl group of one amino acid and the α-amino group of the second amino acid. Isopeptide bonds are rarer than regular peptide bonds. Isopeptide bonds lead to branching in the primary sequence of a protein. Proteins formed from normal peptide bonds typically have a linear primary sequence. Amide bonds, and thus isopeptide bonds, are stabilized by resonance (electron delocalization) between the carbonyl oxygen, the carbonyl carbon, and the nitrogen atom. The bond strength of an isopeptide bond is similar to that of a peptide due to the similar bonding type. The bond strength of a pep ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Coagulation
Coagulation, also known as clotting, is the process by which blood changes from a liquid to a gel, forming a thrombus, blood clot. It results in hemostasis, the cessation of blood loss from a damaged vessel, followed by repair. The process of coagulation involves Platelet-activating factor, activation, Cell adhesion, adhesion and aggregation of platelets, as well as deposition and maturation of fibrin. Coagulation begins almost instantly after an injury to the endothelium that lines a blood vessel. Exposure of blood to the subendothelial space initiates two processes: changes in platelets, and the exposure of subendothelial Tissue factor, platelet tissue factor to coagulation factor VII, which ultimately leads to cross-linked fibrin formation. Platelets immediately form a plug at the site of injury; this is called ''primary hemostasis. Secondary hemostasis'' occurs simultaneously: additional coagulation factors beyond factor VII (#Coagulation factors, listed below) respond in a c ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Catalytic Triad
A catalytic triad is a set of three coordinated amino acid residues that can be found in the active site of some enzymes. Catalytic triads are most commonly found in hydrolase and transferase enzymes (e.g. proteases, amidases, esterases, aminoacylase, acylases, lipases and β-lactamases). An acid-base (chemistry), base-nucleophile triad is a common motif for generating a nucleophilic residue for covalent catalysis. The residues form a charge-relay network to polarise and activate the nucleophile, which attacks the Substrate (chemistry), substrate, forming a covalent intermediate which is then hydrolysed to release the Product (chemistry), product and regenerate free enzyme. The nucleophile is most commonly a serine or cysteine, but occasionally threonine or even selenocysteine. The Protein tertiary structure, 3D structure of the enzyme brings together the triad residues in a precise orientation, even though they may be far apart in the sequence (Protein primary structure, primary ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Factor XIII
Factor XIII, or fibrin stabilizing factor, is a plasma protein and zymogen. It is activated by thrombin to factor XIIIa which crosslinks fibrin in coagulation. Deficiency of XIII worsens clot stability and increases bleeding tendency. Human XIII is a heterotetramer. It consists of 2 enzymatic A peptides and 2 non-enzymatic B peptides. XIIIa is a dimer of activated A peptides. Function Within blood, thrombins cleave fibrinogens to fibrins during coagulation and a fibrin-based blood clot forms. Factor XIII is a transglutaminase that circulates in human blood as a heterotetramer of two A and two B subunits. Factor XIII binds to the clot via their B units. In the presence of fibrins, thrombin efficiently cleaves the R37– G38 peptide bond of each A unit within a XIII tetramer. A units release their N-terminal activation peptides. Both of the non- covalently bound B units are now able to dissociate from the tetramer with the help of calcium ions (Ca2+) in the blood ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Enzyme
An enzyme () is a protein that acts as a biological catalyst by accelerating chemical reactions. The molecules upon which enzymes may act are called substrate (chemistry), substrates, and the enzyme converts the substrates into different molecules known as product (chemistry), products. Almost all metabolism, metabolic processes in the cell (biology), cell need enzyme catalysis in order to occur at rates fast enough to sustain life. Metabolic pathways depend upon enzymes to catalyze individual steps. The study of enzymes is called ''enzymology'' and the field of pseudoenzyme, pseudoenzyme analysis recognizes that during evolution, some enzymes have lost the ability to carry out biological catalysis, which is often reflected in their amino acid sequences and unusual 'pseudocatalytic' properties. Enzymes are known to catalyze more than 5,000 biochemical reaction types. Other biocatalysts include Ribozyme, catalytic RNA molecules, also called ribozymes. They are sometimes descr ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Streptomyces Mobaraensis
''Streptomyces'', from στρεπτός (''streptós''), meaning "twisted", and μύκης (''múkés''), meaning "fungus", is the largest genus of Actinomycetota, and the type genus of the family Streptomycetaceae. Over 700 species of ''Streptomyces'' bacteria have been described. As with the other Actinomycetota, streptomycetes are gram-positive, and have very large genomes with high GC content. Found predominantly in soil and decaying vegetation, most streptomycetes produce spores, and are noted for their distinct "earthy" odor that results from production of a volatile metabolite, geosmin. Different strains of the same species may colonize very diverse environments. Streptomycetes are characterised by a complex secondary metabolism. Between 5-23% (average: 12%) of the protein-coding genes of each ''Streptomyces'' species are implicated in secondary metabolism. Streptomycetes produce over two-thirds of the clinically useful antibiotics of natural origin (e.g., neomycin, s ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Chromosome
A chromosome is a package of DNA containing part or all of the genetic material of an organism. In most chromosomes, the very long thin DNA fibers are coated with nucleosome-forming packaging proteins; in eukaryotic cells, the most important of these proteins are the histones. Aided by chaperone proteins, the histones bind to and condense the DNA molecule to maintain its integrity. These eukaryotic chromosomes display a complex three-dimensional structure that has a significant role in transcriptional regulation. Normally, chromosomes are visible under a light microscope only during the metaphase of cell division, where all chromosomes are aligned in the center of the cell in their condensed form. Before this stage occurs, each chromosome is duplicated ( S phase), and the two copies are joined by a centromere—resulting in either an X-shaped structure if the centromere is located equatorially, or a two-armed structure if the centromere is located distally; the jo ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Protein 4
Proteins are large biomolecules and macromolecules that comprise one or more long chains of amino acid residues. Proteins perform a vast array of functions within organisms, including catalysing metabolic reactions, DNA replication, responding to stimuli, providing structure to cells and organisms, and transporting molecules from one location to another. Proteins differ from one another primarily in their sequence of amino acids, which is dictated by the nucleotide sequence of their genes, and which usually results in protein folding into a specific 3D structure that determines its activity. A linear chain of amino acid residues is called a polypeptide. A protein contains at least one long polypeptide. Short polypeptides, containing less than 20–30 residues, are rarely considered to be proteins and are commonly called peptides. The individual amino acid residues are bonded together by peptide bonds and adjacent amino acid residues. The sequence of amino acid residues i ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Papain-like Protease
Papain-like proteases (or papain-like (cysteine) peptidases; abbreviated PLP or PLCP) are a large protein family of cysteine protease enzymes that share structural and enzymatic properties with the group's namesake member, papain. They are found in all domains of life. In animals, the group is often known as cysteine cathepsins or, in older literature, lysosomal peptidases. In the MEROPS protease enzyme classification system, papain-like proteases form Clan CA. Papain-like proteases share a common catalytic dyad active site featuring a cysteine amino acid residue that acts as a nucleophile. The human genome encodes eleven cysteine cathepsins which have a broad range of physiological functions. In some parasites papain-like proteases have roles in host invasion, such as cruzipain from ''Trypanosoma cruzi''. In plants, they are involved in host defense and in development. Studies of papain-like proteases from prokaryotes have lagged their eukaryotic counterparts. In cellular org ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Immunoglobulin
An antibody (Ab) or immunoglobulin (Ig) is a large, Y-shaped protein belonging to the immunoglobulin superfamily which is used by the immune system to identify and neutralize antigens such as pathogenic bacteria, bacteria and viruses, including those that cause disease. Each individual antibody recognizes one or more specific antigens, and antigens of virtually any size and chemical composition can be recognized. Antigen literally means "antibody generator", as it is the presence of an antigen that drives the formation of an antigen-specific antibody. Each of the branching chains comprising the "Y" of an antibody contains a paratope that specifically binds to one particular epitope on an antigen, allowing the two molecules to bind together with precision. Using this mechanism, antibodies can effectively "tag" the antigen (or a microbe or an infected cell bearing such an antigen) for attack by cells of the immune system, or can neutralize it directly (for example, by blocking a p ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |
Transamidation
Transamidation is a chemical reaction in which an amide reacts with an amine to generate a new amide: :RC(O)NR'2 + HNR"2 → RC(O)NR"2 + HNR'2 The reaction is typically very slow, but it can be accelerated with Lewis acid and organometallic catalysts. Primary amides (RC(O)NH2) are more amenable to this reaction. Ureas In contrast to the reluctance of amides as substrates, urea is more susceptible to this exchange process. Transamidation is practiced, sometimes even on an industrial scale, to prepare a variety of N-substituted ureas: :(H2N)2CO + R2NH → (R2N)(H2N)CO + NH3 :(R2N)(H2N)CO + R2NH → (R2N)2CO + NH3 Methylurea, precursor to theobromine, is produced from methylamine and urea. Phenylurea is produced similarly but from anilinium chloride:{{cite journal , title=Arylureas II. Urea Method p-Ethoxyphenylurea , journal=Org. Synth. , year=1951 , volume=31 , page=11 , doi=10.15227/orgsyn.031.0011 :(H2N)2CO + 6H5NH3l → (C6H5(H)N)(H2N)CO + NH4C ... [...More Info...] [...Related Items...] OR: [Wikipedia] [Google] [Baidu] |